Abstract
Gefitinib is a first generation, reversible epidermal growth factor receptor (EGFR) tyrosine kinase inhibitor (TKI), and EGFR TKIs, such as gefitinib and erlotinib, have yielded dramatic and durable responses in approximately 75% of of non-small cell lung cancer (NSCLC) patients whose tumor has an activating EGFR mutation. EGFR mutations are found in approximately 10%-15% of lung cancers in Caucasians, and they are more frequent in female patients, patients with adenocarcinoma, patients who are never-smokers or have a history of light-smoking, and patients with Asian ethnicity. Recent phase III trials comparing gefitinib with standard chemotherapy have demonstrated a similar survival benefit of gefitinib in patients with NSCLC and improved quality of life both in a first-line setting (IPASS; comparing gefitinib with carboplatin/paclitaxel) and a previously-treated setting (INTEREST; comparing gefitinib with docetaxel). Subset analyses of the data obtained in these studies showed that in patients with EGFR-mutant NSCLC gefitinib yielded a higher response rate, longer progression-free survival, and similar overall survival than standard cytotoxic chemotherapy did. The toxicity of EGFR TKIs is generally milder than that of standard cytotoxic chemotherapy. This review focuses on gefitinib, and issues in the management of EGFR-mutant NSCLC are discussed.
Introduction
Lung cancer is the leading cause of cancer-related deaths worldwide. 1 Non-small cell lung cancer (NSCLC) accounts for approximately 85% of cases of lung cancer, and more than half of all lung cancer patients have advanced disease at the time of diagnosis. 2 Platinum-based combination chemotherapy is currently the standard option for treating patients with advanced or metastatic NSCLC, and it provides a modest survival benefit.3–5 The NSCLC Meta-Analyses Collaborative Group recently updated their initial analyses published in 1995 7 and demonstrated that the addition of chemotherapy to supportive care improved the survival rate at 1 year only slightly, from 20% to 29%. 8 Thus, there is an urgent need for a new treatment modality.
The epidermal growth factor receptor (EGFR) is a member of the HER/ErbB family of transmembrane receptors, which form dimers upon ligand binding, thereby activating the intracellular tyrosine kinase domain and triggering downstream effector pathways that lead to cellular proliferation, angiogenesis, and metastasis.9–11 Because of the importance of the EGFR in tumor biology, an EGFR-targeted treatment strategy that includes a EGFR tyrosine kinase inhibitor (TKI) has been developed. 12 Gefitinib was the first agent in this class that was introduced in clinical practice. In the initial phase II trials gefitinib demonstrated clinical activity and yielded a response rate of approximately 12%-18% in NSCLC patients who had previously been treated with cytotoxic chemotherapy.13,14 Based on the results of these trials, on July 5, 2002, before approval in any other country, the regulatory agency in Japan, the Ministry of Health, Labour and Welfare (MHLW) approved gefitinib for the treatment of NSCLC via an accelerated approval process. The United States (US) Food and Drug Administration (FDA) later approved gefitinib on May 5, 2003. However, a subsequent phase III trial (IRESSA Survival Evaluation in Lung Cancer; ISEL) failed to demonstrate any statistically significant survival benefit of gefitinib over placebo, although a subset of Asian patients and a subset of never smokers seemed to benefit from gefitinib. 15 In response to the results of the ISEL trial, AstraZeneca temporarily withdrew authorization application of gefitinib from the European Medicines Agency (EMEA), and the FDA restricted the use of gefitinib to patients who were enrolled in clinical trials or deriving clinical benefit from ongoing treatment.
Although no clinical benefit of gefitinib for unselected NSCLC patients was demonstrated in early trials, the results of several studies have suggested activity of gefitinib in selected populations, such as patients with Asian ethnicity, never-smokers, patients with adenocarcinoma, and women.13,14,16,17 In 2004, activating mutations in the EGFR gene were discovered and found to be associated with a dramatic and durable response to gefitinib in NSCLC patients.18–20 Following this discovery, several prospective phase II studies demonstrated a high response rate to gefitinib of approximately 60%-80% in patients with EGFR mutations.21–30 Moreover, a recent phase III trial that evaluated gefitinib monotherapy as first-line treatment in comparison with one of the standard platinum-based chemotherapies, paclitaxel and carboplatin (IRESSA Pan-ASia Study; IPASS), showed superior progression-free survival, a higher objective response rate, better tolerability, and a significant quality of life benefit in the gefinitib group. 31 Subset analyses suggested that EGFR mutations were both a predictive factor and prognostic factor in NSCLC patients treated with gefitinib. Moreover, a phase III INTEREST trial comparing gefitinib with docetaxel in patients with previously-treated NSCLC showed equivalent efficacy in terms of overall survival. 32 Based on the results of these trials, the European Commission has granted marketing authorization for use of gefitinib across all lines of therapy to treat patients with locally advanced or metastatic NSCLC harboring EGFR mutations. We are now confronting a new era and need to be concerned about individualized treatment options in patients with EGFR-mutant NSCLC.
Mechanism of Action, Metabolism, and Pharmacokinetic Profile
The mechanism of the clinical antitumor action of gefitinib has not been fully characterized. Gefitinib is an orally bioavailable synthetic anilinoquinazoline that reversibly inhibits intracellular phosphorylation of the EGFR tyrosine kinase.33–36 EGFR is a 170-kDa (1186 amino acid) membrane-bound protein, and it is expressed on the surface of the cancer cells in approximately 40%-80% of NSCLC.
Gefitinib is orally available as brown film-coated tablets for once daily administration. Gefitinib is slowly absorbed after oral administration with mean bioavailability of 60%, and the plasma Cmax is reached 3-7 hours after dosing. 37 Bioavailability is not significantly altered by food intake. 38 In vitro binding of gefitinib to human plasma proteins (serum albumin and alpha 1-acid glycoprotein) is 90% and is independent of the drug concentration. Gefitinib is primarily metabolised by CYP3A439,40 and excreted in the feces (86%). 38 Renal elimination of gefitinib and its metabolites accounts for less than 4% of the administered dose. 38 The elimination half-life is about 48 hours. 41 Steady-state plasma concentrations are achieved within 10 days. 38 Drugs that cause a significant, sustained elevation of gastric juice pH (e.g. histamine H2 receptor antagonists) may reduce the plasma concentration of gefitinib.
Clinical Studies
Phase I studies
The optimal dose and schedule of gefitinib administration have been evaluated in five phase I trials, the first in healthy volunteers, and the other four in patients with solid tumors.41–45 Intermittent and daily dose schedules ranging from 50 mg to 1000 mg per day were assessed. Adverse events were generally mild, i.e. grade 1-2 according to the Common Toxicity Criteria of the National Cancer Institute, reversible, and mainly consisted of an acne-like rash, diarrhea, nausea, anorexia, vomiting, and asthenia. The dose-limiting toxicity was diarrhea, which occurred at doses ranging from 700 mg/day to 1000 mg/day, and the maximum tolerated dose (MTD) was identified as 700 mg/day. Moreover, doses >600 mg/day required dose reduction in many patients. Gefitinib doses of 150 mg/day or more were associated with target inhibition, and doses of 150-800 mg/day were associated with a tumor response. Based on these findings, two doses, 250 mg/day and 500 mg/day, were selected for investigation in phase II and III trials. Both doses were significantly below the MTD, in contrast to the usual doses selected for cytotoxic agents, which are near the MTD.
Phase II studies of gefitinib in unselected patients (Table 1)
Phase II trials of gefitinib in unselected patients.
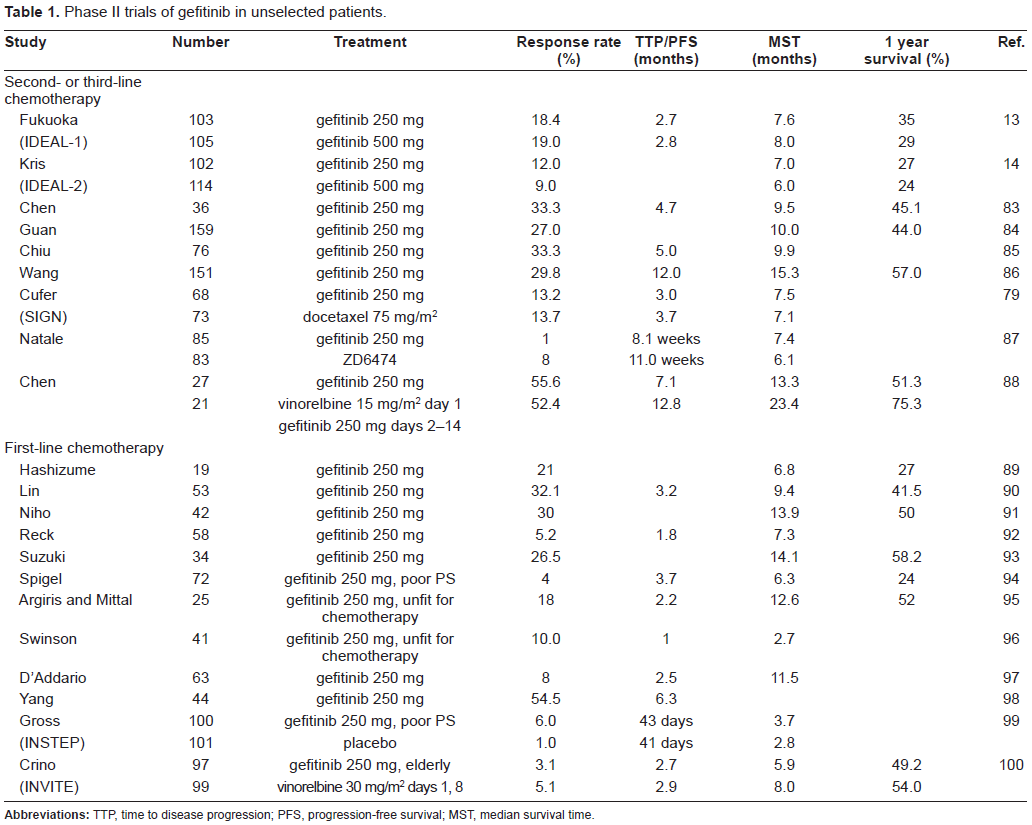
Two randomized, double-blind, parallel-group, multicenter phase II studies (Iressa Dose Evaluation in Advanced Lung Cancer; IDEAL-1 and -2) investigated the activity of gefitinib in patients with advanced NSCLC.13,14 The IDEAL-1 trial randomly assigned 210 patients with advanced NSCLC in Europe, Australia, Japan, and South Africa who had received one or two chemotherapy regimens, at least one of which had contained platinum, to receive either gefitinib 250 mg/day or gefitinib 500 mg/day, and no significant difference in response rate to gefitinib was observed between the 250 mg/day group and 500 mg/day group (18.4% versus 19.0%). The IDEAL-2 trial was performed in 30 centers in the US. A total of 221 patients who had received at least two chemotherapy regimens were randomly assigned to receive either gefitinib 250 mg/day or gefitinib 500 mg/day. The same as in IDEAL-1, no significant difference in response rate to gefitinib was found between the 250 mg/day group and the 500 mg/day group (12% versus 9%). The 250 mg/day dose of gefitinib was chosen for subsequent trials based on the tolerability and efficacy data observed in the IDEAL trials. Other major phase II studies are summarized in Table 1.
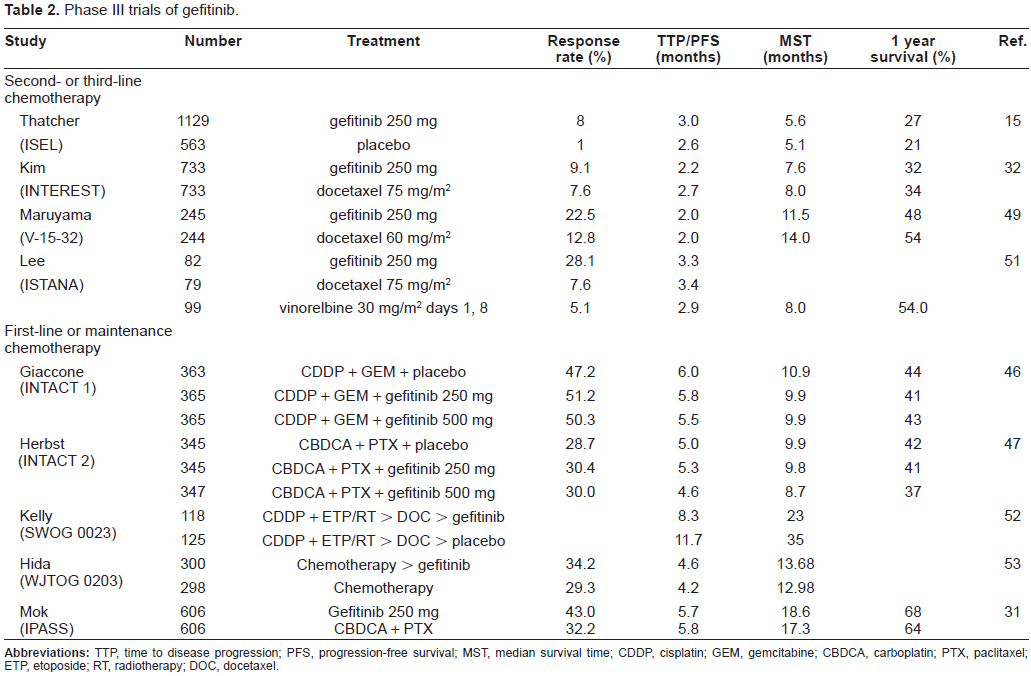
Phase III studies (Table 2)
Phase III trials of gefitinib.
Combination of gefitinib with cytotoxic chemotherapy
The IRESSA NSCLC Trial Assessing Combination Treatment (INTACT)-1 and -2 studies were randomized, double-blind, placebo-controlled studies that investigated gefitinib (250 or 500 mg/day) in combination with standard platinum-based first-line chemotherapy.46,47 In INTACT 1, which was conducted on 1093 patients in Europe, North America, South America, South Africa, and Asia, gefitinib was combined with gemcitabine and cisplatin. In INTACT 2, conducted mainly in the US, gefitinib was combined with paclitaxel and carboplatin. Neither the 250 mg/day nor the 500 mg/day dose of gefitinib in combination with cytotoxic combination chemotherapy increased overall survival compared with placebo. The subset analysis of tumor samples from INTACT 1 and 2 showed that the group of patients whose tumor had an EGFR mutation had a higher response rate than the group without a mutation, but the difference did not reach statistical significance (72% versus 55%). 48 Overall survival, however, was unaffected by the addition of gefitinib to chemotherapy in patients whose tumor had an EGFR mutation (HR 1.77; 95% CI 0.50-6.23).
Monotherapy as second- or third-line therapy
The Iressa Survival Evaluation in Lung Cancer (ISEL) was a double-blind, placebo-controlled, multicenter phase III trial that compared overall survival of gefitinib monotherapy as second- or third-line treatment for patients with locally advanced or metastatic NSCLC. 15 A total of 1692 patients were enrolled from 210 centers in 28 countries across Europe, Asia, the US, South America, Australia, and Canada, and 1129 of the patients were assigned to gefitinib and 563 to placebo. The objective response rate was significantly higher in the gefitinib arm (8% versus 1%). However, median survival was 5.6 months and 1-year survival 27% in the gefitinib group, compared to 5.1 months and 21%, respectively, in the placebo group, and neither difference was statistically significant. Pre-planned subgroup analyses showed longer survival times in the subsets of the gefitinib patients with Asian ethnicity and who were never-smokers.
Three phase III trials have investigated gefitinib versus docetaxel, a standard second-line chemotherapy, in previously-treated NSCLC patients. INTEREST (Iressa in NSCLC Trial Evaluating Response and Survival versus Taxotere), the largest of the phase III trials, compared gefitinib (250 mg/day) with docetaxel (75 mg/m2 every 3 weeks) in 1466 patients from 24 countries, 32 and non-inferiority of gefitinib in comparison with docetaxel in terms of overall survival in all patients, which was the primary endpoint, was demonstrated (HR 1.02; 96% CI, 0.905-1.150). However, superiority of gefitinib in terms of overall survival was not demonstrated in the 174 patients with a high EGFR-gene-copy number. Surprisingly, exploratory analyses showed that the overall survival benefit of gefitinib was not restricted to certain subgroups. No clinical factors or biomarkers appeared to be predictive of a greater survival-prolonging effect of gefitinib over docetaxel. Patients with adenocarcinoma, patients of Asian origin, females, and never-smokers had similarly long survival times whether on gefitinib and docetaxel. Among the patients whose tumor had a EGFR mutation the gefitinib group had a significantly longer progression-free survival than the docetaxel group (median progression-free survival 7.0 months versus 4.1 months, p = 0.0012); but overall survival was similar (14.2 months versus 16.6 months, p = 0.6043). The V-15-32 study compared gefitinib 250 mg daily with docetaxel 60 mg/m2 every 3 weeks, and was a smaller phase III trial involving 490 Japanese patients with previously-treated NSCLC. 49 It demonstrated similar activity by gefitinib and docetaxel, but the trial did not achieve the primary endpoint of non-inferiority (HR 1.01; 95% CI 0.80-1.27; P = 0.94). There was a subsequent high gefitinib prescription in the docetaxel arm (53%), whereas only 36% of the patients in the gefitinib arm received cytotoxic chemotherapy that included docetaxel. Thus, the post-study treatment may have impacted the assessment of overall survival. Another possibility is that the diverse patient population that included both patients with and without EGFR mutations influenced the overall results. A recent review addressed differences between EGFR TKIs, including gefitinib, and cytotoxic agents. 50 The authors pointed out that cytotoxic agents displayed broader anticancer spectra than molecularly-targeted agents did, but that molecularly-targeted agents could produce a profound response. They speculated that the survival of patients treated with EGFR-TKI might not improve even if the response rate was twice that of the response rate to cytotoxic agents according to the results of V-15-32 trial. The third trial, Iressa as Second Line Therapy in Advanced NSCLC-Asia (ISTANA), was a phase III trial that compared gefitinib (250 mg daily) with docetaxel (75 mg/m2 every 3 weeks) in 161 Korean patients with advanced NSCLC. 51 Progression-free survival was significantly longer in the gefitinib group than in the docetaxel group (HR 0.73; 90% CI 0.53-1.00; P = 0.00441).
Maintenance and adjuvant therapy
Maintenance gefitinib therapy was assessed in two phase III trials. In the first of the two trials, Southwest Oncology Group (SWOG) 0023 trial, assessed gefitinib maintenance in comparison with placebo after definitive radiotherapy plus concurrent etoposide and cisplatin with consolidation docetaxel, but it closed early after 575 patients of the planned 840 had been accrued. 52 An unplanned interim analysis after the results of the ISEL trial 15 showed that gefitinib maintenance was unlikely to improve survival at all. Disappointingly, overall survival was shorter in the gefinitib group, in which median survival was 19 months, compared with 26 months in the placebo group. In the second of the two trials, the West Japan Thoracic Oncology Group (WJTOG) 0203 investigated gefitinib maintenance after platinum-based combination chemotherapy. 53 The improvement in overall survival, the primary endpoint, did not reach the level of statistical significance, although there was a trend toward longer survival in the gefitinib arm (HR 0.86; 95% CI 0.72-1.03; P = 0.10). The pre-planned subgroup analysis showed longer overall survival for Gefitinib in patients with adenocarcinoma (HR 0.79; 95% CI 0.65-0.98; P = 0.03).
The National Cancer Institute of Canada BR19 study was conducted to compare gefitinib as adjuvant therapy for resected early NSCLC to placebo. However, after the results of the ISEL trial became available, this trial was also closed early, and the results have still not been made available.
Trials of gefitinib therapy in selected populations of NSCLC patients
EGFR mutations
In April 2004, two groups of investigators reported finding that activating EGFR mutations were present in a subset of NSCLC and that they had a high response rate to gefitinib.18,19 EGFR mutations are predominantly found in patients with adenocarcinoma, patients of Asian origin, females, and never-smokers. Based on this finding, several trials have investigated the efficacy of gefitinib in groups of patients who were highly selected for the presence of activating EGFR mutations. The I-CAMP combined these seven prospective studies conducted in Japan to evaluate gefitinib in patients with EGFR-mutant NSCLC. 21 The data obtained in a total of 148 patients were analyzed, and the response rate, median progressionfree survival, and overall survival were 76.4%, 9.7 months, and 24.3 months, respectively. Sequist and colleague reported the multicenter iTARGET study conducted in the US. 29 In that study 31 patients were treated with gefitinib, and the response rate was 55%. Yang and colleague reported a high response rate (84%) in EGFR-mutant NSCLC patients with an exon 19 deletion and exon 21 L858R, but the response rate was poor (17%) in the patients whose tumor had other EGFR mutations. 30 A meta-analysis based on a comprehensive EGFR somatic mutation database confirmed that EGFR mutations were predictive of response to gefitinib (RR 6.38, 95% CI 4.68-7.70) in patients with advanced NSCLC. 54
Other clinocopathologic factors
Clinocopathologic factors have also been considered in patient selection. The ONCOBELL trial selected patients who were never-smokers or had evidence of a high gene copy by FISH and were phospho-Akt (P-Akt) positive, and the patients received gefitinib monotherapy. 55 Forty-two patients were enrolled, and the response rate was 48%. EGFR mutations were detected in 24 patients (67%), and their response rate was 63%. Patients with bronchioloalveolar carcinoma were selected in a phase II trial performed by SWOG, and the response rate was 17% in previously untreated patients and 9% in previously treated patients. 56 A phase II trial conducted in Korea investigated the efficacy of gefitinib in 72 chemotherapy-naive never-smokers with adenocarcinoma, and the objective response rate was 55%. 57
The results of an open-label, randomized phase III trial (Iressa Pan ASian Study; IPASS) that compared gefitinib with paclitaxel and carboplatin as first-line chemotherapy in patients with adenocarcinoma who were never- or light-smokers were recently reported. 31 The primary endpoint, non-inferiority of progression-free survival of gefitinib in compari son with paclitaxel/carboplatin, was demonstrated, and progression-free survival was longer in gefitinib (HR 0.74; 95% CI 0.65-0.85; P < 0.0001). However, the Kaplan-Meier plots of the gefitinib group and paclitaxel/carboplatin group “crossed”, with the progression-free survival rate being better in the paclitaxel/carboplatin group in the first 6 months and better in the gefitinib group in the remaining 16 months, and thus the results need to be interpreted carefully. Subgroup analyses revealed better progression-free survival in NSCLC patients with EGFR mutations who were treated with gefitinib than in those treated with paclitaxel/carboplatin, whereas gefitinib was inferior to paclitaxel/carboplatin in patients without EGFR mutations.
Safety
Gefitinib is generally well tolerated. The most common adverse events reported at the recommended 250 mg daily dose have been diarrhea, rash, acne, dry skin, nausea, and vomiting. 58 Diarrhea was the dose-limiting toxicity in phase I trials. Myelosuppression, neurotoxicity, and nephrotoxicity are typically observed with conventional cytotoxic chemotherapy but not with gefitinib. In the ISEL study, 15 the most common adverse events in the group treated with gefitinib were rash and diarrhea. The overall frequency of grade 3/4 adverse events was 30% in the gefitinib group as opposed to 7% in the placebo group. The data from the ISEL and IDEAL-1 trials showed similar adverse event profiles in Asian and non-Asian patients,13,15 but in the ISEL study the frequency of grade 3/4 toxicity was slightly higher in the Asian patients (43% versus 30%).
The most troublesome adverse event that has occurred in patients treated with gefitinib has been interstitial lung disease (ILD).59–66 A higher incidence of ILD has been reported in patients treated with gefitinib in Japan than in other countries. The worldwide incidence of ILD in patients treated with gefitinib is approximately 1%-2%, and in approximately one third of the patients it has been fatal.15,32 The INTEREST study reported ILD in 1.4% of the patients who received gefitinib, compared with 1.1% of the patients who received docetaxel. 32 In the ISEL study, 1% of the patients who received gefitinib developed ILD, 15 and ISTANA, a phase III trial conducted in Korea, reported a slightly higher incidence of 3.7% among patients who received gefitinib. 51 In a Taiwanese study that evaluated gefitinib as first-line chemotherapy, the incidence of ILD was 0.9%. 67 In Japanese case series the incidence of ILD has been relatively high. The West Japan Thoracic Oncology Group retrospectively analyzed 1976 patients treated with gefitinib and found that the incidence of ILD was 3.2%. 62 Another multicenter prospective study conducted in Japan reported gefitinib-related ILD in 193 (5.8%) of 3322 cases. 68 A nested case-control study conducted in Japan by an academic team in collaboration with AstraZeneca reported an observed incidence rate of ILD per 1000 person-weeks over 12 weeks of 4.5 for gefitinib and 1.7 for chemotherapy. 60 The corresponding observed, naive, cumulative incidence rates at the end of the 12-week follow-up period were 4.0% and 2.1%, respectively. The risk factors that have been identified for ILD are male gender, prior history of smoking, advanced age, poor performance status, and preexisting pulmonary fibrosis, which probably has the greatest impact on the development of ILD. These risk factors do not apply to women, adenocarcinoma patients, or never-smokers, all of which are groups that are more likely to benefit from gefitinib. However, I-CAMP reported an incidence of ILD of 2.7% in patients with a EGFR mutation. 21 The reason for the difference in the incidence of ILD between Japanese and non-Japanese patients is unclear.
Efficacy
EGFR mutations and the efficacy of gefitinib (Table 3)
Gefitinib in selected patients.
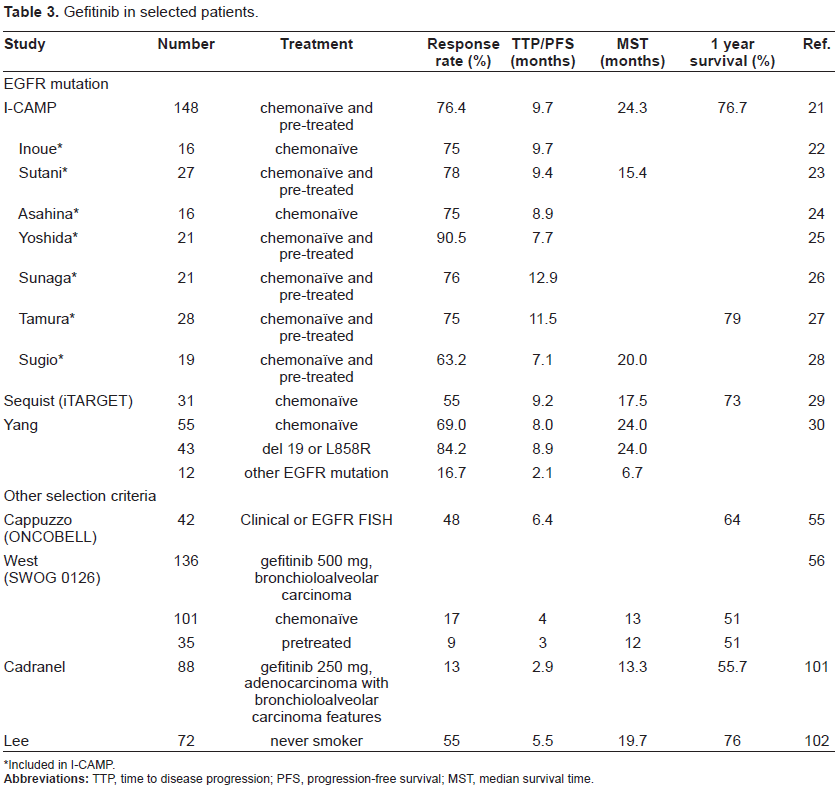
Included in I-CAMP.
Gefitinib has yielded a response rate of approximately 75%, a progression-free survival time of 10 months, and an overall survival time of 24 months. 21 The results of two retrospective studies indicated that the presence of exon 19 deletions is associated with better survival than the presence of L858R mutations is.69,70 Gefitinib is currently available in second- or third-line settings in most countries; however, the subset analyses in IPASS indicated that gefitinib is active and one of the options for first-line chemotherapy in patients whose tumor had an EGFR mutation. 31 Accordingly, EMEA has recommended approval of gefitinib in all lines of therapy for patients with locally advanced or metastatic NSCLC whose tumor has an activating EGFR mutation.
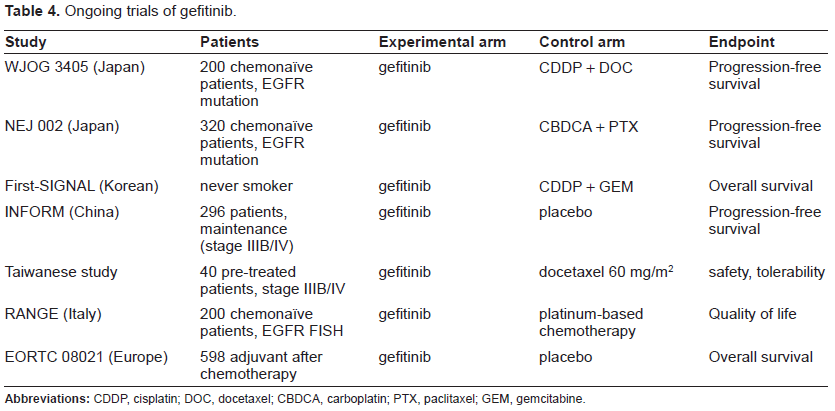
When to prescribe gefitinib is an important issue. A study from Taiwan referred to this issue and compared the outcomes of different lines of gefitinib treatment in patients with exon 19 deletions or L858R. 71 They suggested that the response rate to gefitinib of chemonaive patients is higher than that of previously-treated patients; however, there is no difference in terms of overall survival. The results of I-CAMP suggested that gefitinib produces longer progression-free survival in a first-line setting than in a second-line or subsequent treatment setting. 21 These findings should be validated, and the role of gefitinib as first-line chemotherapy in patients with EGFR mutations should be clarified by two ongoing randomized phase III trials comparing gefitinib with standard chemotherapy (Table 4).
Ongoing trials of gefitinib.
The response to gefitinib of patients without activating EGFR mutations is poor. In the exploratory analysis of the results of the IDEAL studies, the response rate of patients whose tumor lacked activating EGFR mutations was 10%, 48 and in the IPASS study the response rate of such patients was only 1%. 31 One of the reasons for the difference in response rate among these trials may be technical issues related to the detection of EGFR mutations, and a discrepancy between the sensitivity of methods of detecting EGFR mutations has recently been demonstrated. 72 The IPASS study used the scorpion ARMS method to detect EGFR mutations, and it is considered more sensitive.
Mechanism of secondary resistance
Even patients whose tumor harbors activating EGFR mutations and who experience an initial dramatic response always develop resistance to gefitinib, but the mechanism responsible for the development of resistance to gefitinib has not been fully characterised. The T790M mutation in exon 20 was found in approximately 50% of NSCLCs with acquired resistance to gefitinib,73,74 and exploratory analyses have shown that seven out of 14 Japanese patients and 14 out of 24 Taiwanese patients had the T790M mutation.75,76 Since codon 790 is involved in the binding site to EGFR-TKIs, substitution of threonine by the bulkier methionine is suspected of causing steric hindrance to TKI binding. Another mechanism that has been proposed for the development of resistance to gefitinib is MET amplification. 77 MET is a receptor for hepatocyte growth factor/scatter factor, and overexpression, amplification, and mutation of the MET gene have reported in various human cancers, including in NSCLC. MET amplification was detected in four of 18 (22%) NSCLCs that developed resistance to gefitinib or erlotinib.
Patient preference
The evidence suggests that in unselected NSCLC patients 32 second-line gefitinib therapy results in a survival rate that is similar to the rate obtained with standard second-line chemotherapy.32,49,51 In EGFR mutant NSCLC, gefitinib therapy yields longer progression-free survival than standard second-line chemotherapy and a higher response rate.31,78 One of the potential benefits of gefitinib over cytotoxic chemotherapy is its mild toxicity. The relatively mild myelosuppression, alopecia, gastrointestinal toxicity, and asthenia caused by gefitinib results in better quality of life (QOL). In the SIGN trial, 79 patients treated with gefitinib showed better improvement in symptoms as assessed by lung cancer symptom (LCS; 37% versus 26%) and QOL as assessed by the Functional Assessment of Cancer Therapy-Lung (FACT-L; 34% versus 26%) than patients treated with docetaxel. In addition, in the INTEREST trial patients treated with gefitinib showed a significantly better improvement in QOL as measured by the FACT-L score. 32 Symptom improvement rates were also better in the gefitinib arm than in the docetaxel arm, but the difference was not statistically significant. Gefitinib also improved aspects of QOL over docetaxel in the V-15-32 study. 80 On the other hand, skin rash, diarrhea, and, rarely, ILD especially in Japanese patients may be troublesome. Gefitinib is available in an oral dosage form, which makes administration convenient for patients.
Gefitinib showed activity even in patients with poor PS if their tumor had an EGFR mutation. According to a recent phase II study in a first-line chemotherapy setting, gefitinib yielded a median progression-free survival of 6.5 months, median survival of 17.8 months, and 1-year survival rate of 63% in patients whose tumor had EGFR mutations who did not have indications for cytotoxic chemotherapy because of poor PS. 81 The authors of that study concluded that testing for EGFR mutations as biomarkers should be recommended in this patient population. Gefitinib is an option for patients with poor PS and EGFR mutations even in a first-line setting.
Conclusions
Gefitinib represents a significant advance in the treatment of advanced and metastatic NSCLC. However, considerable issues remain to be resolved. Although gefitinib has shown activity against recurrent or relapsed NSCLC, is the survival benefit limited to certain populations? Recent trials clearly show that gefitinib induces a greater tumor response in patients with activating EGFR mutations.18,19,21,29,30 However, a subset analysis of the results of the INTEREST study showed that the survival benefit of gefitinib is not limited to certain subgroups. 78 Similar findings were obtained in a randomized phase III study that compared another EGFR tyrosine kinase inhibitor, erlotinib, with placebo in patients with recurrent NSCLC (BR.21 study). 82 Erlotinib showed similar activity over the subgroups in terms of overall survival, but the response rates were higher in patients with adenocarcinoma, female patients and patients whose tumors were found to be positive for EGFR expression by immunochemistry. The difference between the efficacy of gefitinib and erlotinib against NSCLC remains to be resolved.
Gefitinib has been demonstrated to yield a higher response rate and better progression-free survival in patients whose tumor has activating EGFR mutations than standard cytotoxic chemotherapy does. Gefitinib should be considered for the treatment of NSCLC patients with activating EGFR mutations, although the optimal timing of gefitinib therapy has not been determined. Gefitinib's toxicity is relatively mild except for ILD, which is uncommon. Gefitinib is a standard second-line chemotherapy, and could be served as one of the options for first-line chemotherapy in patients with activating EGFR mutations based on IPASS trial. Two randomized phase III trials designed to assess gefitinib as first-line therapy for NSCLC patients whose tumor has an EGFR mutation are currently underway in Japan.
Disclosures
There was no source of support. The authors have no conflicts of interest to declare.
