Abstract
Breast cancer remains the second leading cause of malignancy-related death in women in the United States, regardless of advances in novel therapeutic agents. High priority should be emphasized in research aimed at the study of pharmacological and natural compounds that may potentially prevent the development of breast cancer in susceptible patients. Among the known selective estrogen receptor modulators with proven chemopreventive effects, raloxifene has been studied in a number of clinical trials evaluating this drug for the prevention of osteoporosis and coronary heart disease. The MORE and CORE trials had as a primary end point the efficacy of raloxifene in the treatment of women with osteoporosis. These studies showed that raloxifene reduced the risk of invasive breast cancer in postmenopausal women. However, the STAR trial showed no significant difference between raloxifene and tamoxifen recipients in the incidence of invasive breast cancer in postmenopausal women at high risk of invasive breast cancer. This review focuses on the chemopreventive properties of raloxifene and the clinical trials that have proven its efficacy as a chemopreventive agent in invasive breast cancer.
Introduction
Breast cancer is the most common malignancy among women in almost all of Europe, in North America, in much of Latin America, and in Australia. 1 In many of these populations, it is the leading cause of death for women between the ages of 35 and 54 years, and among women younger than 50 years it is almost as common as all cancers combined in men. 2 Breast cancer affects an estimated 1.2 million women worldwide each year and it accounts for over 410,712 deaths. 3 In 2008, 182,460 new cases of invasive breast cancer and over 40,410 deaths occurred among women in the United States. 4 Nonetheless, breast cancer incidence have reached a plateau in recent years after having a consistent 2%-3% increase in the last three decades.5,6 Perhaps these changes could be attributed to the early detection by mammography and a reduction in hormone replacement therapy. With the advent of new treatment modalities and early detection, there has been a 13% increase in breast cancer survival over the last three decades.5,7,8 Despite these improvements, breast cancer still remains a major cause of morbidity and mortality; therefore, high priority should be given to research aimed at the study of pharmacological and natural agents that could potentially prevent the development of breast cancer in susceptible patients.
Estrogen in Breast Cancer
The link between ovarian function and breast cancer has long been established. In 1896, Beatson demonstrated that in some patients with metastatic breast cancer (MBC) the disease could be palliated by performing bilateral oophorectomy. 9 Ovarian hormones are crucial in the regulation of breast cancer cells. Their actions include major mechanisms, such as DNA synthesis, gene expression, cell division and differentiation, progression through cell cycle phases, growth factor synthesis, apoptosis, modulation of cyclic adenosine monophosphate dependent pathways and stimulation of certain enzymes, such as cathepsin D and plasminogen activator.10,11 Epidemiologic studies have clearly demonstrated that early menopause, whether natural or artificial (bilateral oophorectomy), substantially reduces breast cancer risk. Menopause before the age of 35 years is associated with a 60%-70% reduction in breast cancer risk. 12
The discovery of the estrogen receptor (ER) by Jensen and Jacobson provided a way to predict hormone responsiveness of a patient's tumor to endocrine ablation.13,14 During the 1950s, research focused on contraception discovered antiestrogens.15,16 Subsequently, several studies with the use of antiestrogens led to the development of tamoxifen which became the first antiestrogen approved for the treatment of advanced breast cancer in postmenopausal women. 17 The term “antiestrogen” is not an accurate one; these medications have estrogen-like properties in bone and lipid levels as well as an estrogen antagonist function in uterus and breast. Hence, the name selective estrogen receptor modulator (SERM) is more suitable.
Several risk factors for the development of breast cancer; age, family history, prior history of breast cancer, early menarche, late menopause, age at first pregnancy, oral contraceptives, postmenopausal hormone replacement, diet, and alcohol have been documented. 18 The lifetime risk for women in the United States of America (USA) is 13% with a 3.3% risk of dying from the disease. 19 Despite the well-known risk factors, estimating an individual's risk is complicated because a risk factor may not be constant throughout a women's life. The modified Gail model which is based on age, race, age at menarche, age at first live birth or nuliparity, the number of first degree relatives with breast cancer and the number and histology of previous breast biopsies is the most well-known and recommended method for assessing breast cancer risk. 20 This model calculates the percentage risk of developing breast cancer in the next 5 years and it also provides and estimate of lifetime breast cancer risk. The women whose 5 year predicted risk is equal or greater than 1.66% are considered to be at high risk for developing invasive breast cancer.
In addition to having monthly breast self-examinations and yearly mammography for early detection of breast cancers, only three other prophylactic options have proven to be effective in the prevention of breast cancer for high-risk women: 1) bilateral mastectomies, 20 2) bilateral oophorectomy, 21 or 3) pharmacologic agents such as tamoxifen and/or raloxifene (EVISTA®, Eli Lilly, Indianopolis, IN, USA). 23 All three reduce the risk yet do not eliminate it entirely.
A chemopreventive agent targets the very substrates of the carcinogenesis process by either preventing the development of a premalignant lesion, preventing a premalignant lesion from developing into an invasive cancer or causing a premalignant lesion to reverse toward a normal state. The benefits of raloxifene, a second generation SERM, as breast cancer chemoprevention were identified while conducting studies on women with osteoporosis.23,24 It has been studied and used for the last couple of decades as chemoprophylaxis for women with high risks of developing breast cancer.25–28
Pharmacodynamic and Pharmacokinetic Properties
Raloxifene, a benzotiopene SERM, has shown to inhibit estrogen-stimulated growth of mammary cancer in preclinical studies. Raloxifene competes with estrogen at ER and acts as an estrogen agonist in bone, and an estrogen antagonist in uterine and breast tissues.29,30 Raloxifene is administered orally and is rapidly absorbed. Approximately 60% of an oral dose is absorbed; however, raloxifene undergoes extensive first-pass glucuronidation and enterohepatic cycling and the absolute bioavailability of the drug is 2%. 30 Peak plasma concentrations of raloxifene and its metabolites are reached approximately 6 hours after oral administration. 31 The recommended dosage of raloxifene is 60 mg once daily. 29 Raloxifene is extensively distributed in the body and its two monoglucuronide metabolites are >95% bound to plasma proteins. 32 Since no other metabolites have been detected, it seems that the drug is not metabolized by the cytochrome P450 system. 33 The excretion of raloxifene occurs primarily in the feces and <0.2% as unchanged drug in the urine. 34
It has not been seen that age, race or alcohol consumption affects raloxifene pharmacokinetics. 34 The drug has good overall tolerability; adverse effects such as hot flashes, peripheral edema, endometrial cavity fluid, leg cramps, and gall bladder disease were experienced more frequently in patients taking raloxifene than in the placebo group.25,35 In addition, raloxifene is associated with rare but serious complications which include deep venous thrombosis and pulmonary embolism. 24 Raloxifene should not be used in women at increased risk of venous thrombosis such as women who smoke, those who have had a prior pulmonary embolism, venous thromboembolism, stroke, transient ischemic attacks and or uncontrolled atrial fibrillation. In addition, raloxifene should be discontinued at least 72 hours prior to prolonged immobilization (e.g. surgery).
Raloxifene and its Impact on Breast Cancer Prevention
During the past decade, a number of clinical trials have been conducted to assess the benefit of raloxifene on osteoporosis and fracture (Table 1). The Multiple Outcomes of Raloxifene Evaluation (MORE) trial 24 randomized 7,704 postmenopausal female patients (average age, 65.5 years) who had osteoporosis and had no history of cancer of the breast or endometrium to receive either raloxifene at a dose of 60 or 120 mg oral daily or placebo. The primary endpoint was bone events; secondary endpoint included the report of breast cancer incidence. Within a median follow up of 40 months, 13 cases of invasive breast cancer were confirmed in the 5,129 women assigned to take raloxifene and 27 cases in the 2,576 women assigned to take placebo. Thus, a 76% risk reduction of invasive breast cancer was reported in those patients taking raloxifene; risk reduction was similar in both groups taking raloxifene (60 mg and 120 mg). This trial provided evidence that a median of 40 months of treatment with raloxifene decreases the risk of newly diagnosed breast cancer in postmenopausal women who have osteoporosis and who have no prior history of breast cancer.
Phase III trials of raloxifene with reduction of invasive breast cancer as an endpoint.
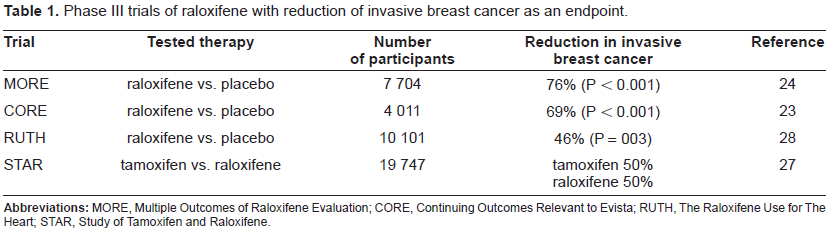
The Continuing Outcomes Relevant to Evista (CORE) trial examined the effect of 4 additional years of raloxifene (60 mg oral daily) therapy on the incidence of invasive breast cancer in women in the MORE trial who agreed to continue therapy. 23 After 4 years of participation in this trial. The risk of invasive breast cancer was reduced by 69%. No increase in the risk of endometrial cancer was observed with raloxifene in either the MORE or CORE trials, but both studies showed a significant increase in the risk of thromboembolic disease similar to the NSABP P-1 study.
The Raloxifene Use for The Heart (RUTH) trial randomized 10,101 postmenopausal women (mean age, 67.5 years) with CHD or multiple risk factors for CHD to take raloxifene 60 mg oral daily versus placebo and participants were followed for a median period of 5.6 years. 28 The study showed no difference between the two study arms in regard with the cardiac primary outcomes. In terms of breast cancer, a 46% risk reduction for invasive breast cancer was documented with raloxifene. The incidence of strokes between raloxifene and placebo groups were not different, however, there was a 49% increase in fatal strokes in women taking raloxifene. A 44% increase in venous thromboembolism was noted in the raloxifene group when compared with the placebo group. These results raised the possibility that protective effects of raloxifene such as breast cancer reduction were not large enough to balance the impact of the serious venous and arterial thromboembolic adverse events. It should be taken into consideration that the RUTH trial was focused on elderly women with a high risk for CHD events, and therefore, its result should not be automatically generalized to the entire postmenopausal population.
Tamoxifen vs. raloxifene
The NSABP Study of Tamoxifen and Raloxifene (STAR) trial was launched to directly compare tamoxifen with raloxifene in a population of women at increased risk for breast cancer. 27 It evaluated 19,747 postmenopausal women over the age of 35 (defined as: 1- history of at least 12 months without spontaneous menstrual bleeding or 2- a documented hysterectomy and bilateral salpingo-oophorectomy (BSO) or 3- age 55 years or older with a hysterectomy with or without oophorectomy or 4- age younger than 55 years, either with a hysterectomy without oophorectomy or with unknown ovary status, and with a documented level of follicle-stimulating hormone confirming elevation in the postmenopausal range) with a 5-year predicted breast cancer risk of at least 1.66% based on the Gail model, 20 and women with LCIS. Patients were randomized to receive tamoxifen 20 mg oral daily or raloxifene 60 mg oral daily. In 2006, after a median follow-up of 3.9 years no difference was found in the incidence of invasive breast cancer in the patients treated with raloxifene or tamoxifen; both decreased the incidence of invasive breast cancer by 50%. However, as observed in the CORE trial, raloxifene did not decrease the incidence of non-invasive carcinoma, while tamoxifen decreased the incidence in half. 27 Raloxifene demonstrated a significant lower incidence of vasomotor symptoms, leg cramps, gynecologic and bladder symptoms in comparison with those patients who were taking tamoxifen. Moreover, patients taking raloxifene experienced a decreased incidence of cataracts and endometrial hyperplasia in comparison to the tamoxifen group. 35 The risk of serious complications such as thromboembolic events were also significantly less in the raloxifene group. In terms of endometrial cancer, the incidence was 38% lower in the raloxifene group than in the tamoxifen group. The number of strokes was similar in both groups, but the incidence of thromboembolic disease was statistically significantly less in the raloxifene group.
Conclusion
Although outcomes for breast cancer patients has improved over the past several decades due to earlier diagnosis and improved adjuvant treatments, the best approach will continue to be prevention, targeting the disease at the initial stages of development. Despite the case that more than one-half million women in the USA take raloxifene for osteoporosis, we believe that this drug is still underutilized since only a subset of eligible women receives it. The stigma of “cancer drugs” and the increased risk of serious adverse events might be the reasons behind their limited acceptance.
The choice of chemoprevention agent must consider a risk versus benefit estimation for each patient. Since raloxifene has proven to reduce the risk of invasive breast cancer, it is recommended for postmenopausal women at high-risk of invasive breast cancer. Lack of data regarding safety and efficacy of raloxifene in premenopausal women has precluded its use in this specific population. In addition, the MORE and CORE trials showed the benefit of raloxifene as breast cancer chemoprevention over an 8 year span, but the optimal duration of chemoprevention is not known.
Disclosures
Edgardo Santos has served on a Speaker's Bureau for Eli Lilly and Company, Genentech, Inc., Millenium Pharmaceuticals, Inc., and sanofi-aventis U.S. Stefan Gluck has acted as a consultant for and has received honoraria and grants form Sanofi-Aventis, Roche, Eli Lilly, Genentech, GlaxoSmithKline, Novartis, Pfizer, Merck, Abraxis and AstraZeneca.
