Abstract
Multiple sclerosis (MS) is considered an autoimmune disease causing demyelination in the central nervous system (CNS) that subsequently leads to axonal damage and neurological impairment. Currently available first line therapies are based on immunomodulation with beta-interferons or glatirameracetate. However, these treatments are only partially effective, thus, more powerful therapies represent an unmet need in MS. Natalizumab is a monoclonal antibody targeting the α4β1 integrin that has been shown to be crucial in the process of transmigration of immunocompetent cells across the blood-brain-barrier (BBB) into the CNS. Two phase III trials have demonstrated clinical and paraclinical efficacy of natalizumab and recent data suggest that many patients that have failed on a first-line disease modifying drug (DMD) benefit from a treatment with natalizumab. Unfortunately, since the licensing of natalizumab in 2006 there have been 75 cases of progressive multifocal leukoencephalopathy (PML) reported. This rare, but potentially fatal infection of the brain by JC-virus restricts the use of natalizumab. Currently there are attempts to define algorithms based on the identification of risk factors for the development of PML to achieve a better safety management for MS patients treated with this monoclonal antibody.
Introduction
Multiple sclerosis (MS) is the most common cause of non-traumatic neurological deficits in early adulthood. It affects approximately 2.5 million individuals worldwide with a peak age of onset in the third and fourth decade.1,2 MS is thought to be a chronic inflammatory autoimmune disease of the central nervous system (CNS) that leads to immune mediated demyelination and subsequent axonal degeneration. 3 The clinical presentation varies considerably between individuals. The most common presentation is a relapsing-remitting course (RRMS), often converting into a secondary progressive phase after 10–15 years. The currently available treatments achieve their best results during the relapsing-remitting phase and target the immune reaction.1,2 Traditional first-line therapies comprise of different interferon-beta (IFNβ) preparations and glatirameracetate (GA). However, these therapies reduce the annualised relapse rate by only approximately 30%4–6 which is clearly not sufficient and thus the search for novel, more effective treatments is warranted. Although there are no comparative studies with IFNβ or GA, the licensing of natalizumab for the treatment of RRMS in 2006 was a first step towards the development of a more specific immune therapy for patients with relapsing forms of MS.
What is natalizumab?
Natalizumab is a humanised IgG4 monoclonal antibody (mAb) against the α4 integrin, the first licensed mAb for the treatment of relapsing-remitting multiple sclerosis (RRMS). The development of such a drug took its beginning in the landmark publication by Georges Köhler and Cesar Milstein in 1975 that described the generation of mAb 7 and was granted in 1984 with the nobel prize. They fused an antibody producing B cell line with an immortalised myeloma cell line thus generating an immortalised cell line that produced a specific antibody derived from one clone. This allowed the production of a nearly unlimited amount of this mAb. Since the specific B cell clone is usually generated by immunisation of mice with the respective antigen, most mAb are of mouse origin. Application of such a mAb may evoke in more than 50% of patients an immune reaction due to the formation of human anti-mouse antibodies (HAMA), thus limiting its therapeutic application.8,9 Molecular cloning techniques allow the generation of less antigenic mAb, so colled chimeric or humanised antibodies, where the murine antigen binding site is fused with a human constant region or only the antigen recognising amino acid residues, the three hypervariable complementary-determining regions (CDRs), are left from the murine origin. 10 Natalizmab is such a humanised antibody where the murine protein content is reduced to 5%–10%, 11 thus minimising the antigenicity of the mAb. Even fully human mAb can be generated with novel techniques. 12
Natalizumab–-Mode of Action
Natalizumab binds to the α4 subunit of the very late antigen-4 (VLA-4), an α4β1 integrin (CD49d/CD29) that is known to be crucial for the transmigration of immune cells across the blood-brain-barrier (BBB).13,14 VLA-4 is expressed primarily on T cells and monocytes and to a lesser extent on granulocytes. 15 It has been shown experimentally that the blockade of VLA-4 by natalizumab inhibits the interaction with VCAM-1 and thus prevents the infiltration of encephalitogenic T cells into the CNS16,17 (Fig. 1). Thus, inhibition of leukocyte migration and extravasation is believed to be the main mode of action although additional mechanisms might modulate the therapeutic and adverse effects. For instance, natalizumab seems to have a direct effect on gene expression relevant for function and differentiation of T cells, B cells, neutrophils, and erythrocytes. 18 Furthermore, within the CNS natalizumab may block the interaction of α4 integrins with extracellular matrix proteins thus inhibiting the migration of immune cells within the CNS parenchyma.
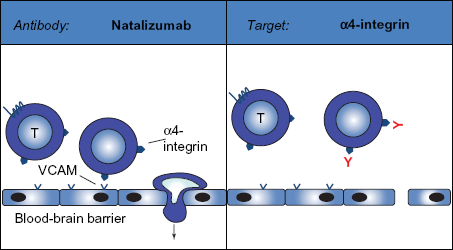
Natalizumab interferes with the α4 integrin, which is critically involved in the process of cell migration across the blood-brain barrier (BBB). By blocking the interaction between VLA-4 and VCAM natalizumab inhibits the transmigration of immunocompetent cells, especially of T cells, out of the blood vessel into the central nervous system.
Natalizumab–-Efficacy Data
After first successful phase II and phase IIb trials,19,20 natalizumab was tested in two large multicenter randomized controlled phase III studies.21,22 In the AFFIRM trial a total of 942 patients received either natalizumab 300 mg intravenously or placebo every 4 weeks for two years. 21 In summary, treatment with natalizumab resulted in a relapse rate reduction of 68% which correlated to a number needed to treat of 1.8. 76% of natalizumab treated but only 53% of placebo treated patients remained relapse free. Confirmed disablility progression, as measured by the Expanded Disabilitiy Status Scale (EDSS), was reduced by natalizumab treatment by 42% (17% confirmed progression in the treatment group vs. 29% in the placebo group). Moreover, more than 37% of patients in the natalizumab group remained free of disease activity as defined by the absence of relapses, sustained disease progression, Gd-enhancing lesions, and new or enlarging lesions, while only 7% in the placebo group remained free of this combined disease activity measures. 23 These clinical data were also reflected by MRI results with an over 90% reduction in gadolinium (Gd)-enhancing lesions, well in line with the previous results from the phase IIb study. Positive effects of natalizumab treatment were also reported for quality of life assessments, requirement for concomittant glucocorticosteroid pulse treatments, or MS related hospitalizations. 24 Recent data suggest that some patients even experience an improvement of their disability as measured by the EDSS. 25
At the same time as the AFFIRM trial was conducted, a second phase III study, the SENTINEL trial, included 1171 patients pretreated with interferon beta1a (IFNβ1a) that had at least one relapse under this treatment. Natalizumab was administered in combination with IFNβ1a and compared to IFNβ1a treatment alone. 22 By and large the results corroborated the data from the AFFIRM study. Natalizumab treatment resulted in a relapse rate reduction of 53% in comparison to treatment with IFNβ1a alone. Similarly, MRI data showed a reduction of 89% of Gd-enhancing lesions and 83% reduction of new or enlarging T2 lesions over two years. Since there were two cases of progressive multifocal leukoencephalopathy (PML) as severe side effects in the SENTINEL trial plus an additional case in a study in Crohn's disease, natalizumab was licensed as monotherapy only for patients who failed on immunomodulatory therapy (IFNβ or GA) as well as for patients with highly active RRMS based on a retrospective assessment of the trial data. Highly active RRMS was defined by the authorities (FDA and EMEA) as at least two severe relapses in the previous year and signs of disease activity on MRI. This restricted approval was introduced as a consequence to the PML cases for risk-benefit considerations (see below).
Natalizumab Safety Concerns
Overall, natalizumab was well tolerated in both large phase III trials. Side effects included headaches and fatigue on infusion days. Allergic reactions occured in about 4% of natalizumab treated patients. Most allergic reactions occurred after the second or third infusion with urticaria, headache, flush, and hypotonia. 26 However, beside this type I hypersensitivity reaction also delayed type III reactions have been observed.27–29 The occurence of allergic reactions is linked to the presence of anti-natalizumab antibodies. In the AFFIRM trial 68% of the patients with infusion related reactions were positive for anti-natalizumab antibodies. 30 Approximately 10% of natalizumab treated patients develop transiently anti-natalizumab antibodies that persist in 6%. Antibodies usually develop within the first 12 weeks of treatment.21,22 Thus, it is recommended to test all patients with infusion related reactions and to perform a routine test after 6 to 9 month of treatment. In patients tested postitive for anti-natalizumab antibodies further infusions should be halted and retesting performed after 4–6 weeks. Patients persistently positive for anti-natalizumab antibodies should not be exposed again to natalizumab due to an increased risk for allergic reactions and the neutralising effect on natalizumab efficacy.
Elevations of liver enzymes and bilirubin were observed in 0.1% of natalizumab treated patients. However, in nearly all cases this side effect was clinically not relevant.
As already mentioned, there were two cases of PML in the SENTINEL trial and another case in a study of natalizumab in Crohn's disease.31–33 Since these patients had concomittant treatment with IFNβ or previous immunosuppressive treatment it was originally suspected that PML occurs in combination therapy. This is the reason why the approval is restriced to monotherapy. Unfortunately, since the licensing of natalizumab 75 more cases have been reported up to Nov. 2nd 2010 (data on file Biogen Idec, http://www.tysabri.de). PML is currently the most important side effect that limits the use of natalizumab and thus requires a stringent pharmacovigilance program and close clinical monitoring of every patient. Despite these cases the risk-benefit-ratio is still considered to be favourable for natalizumab. 34
Progressive multifocal leukoencephalopathy (PML)
PML is a viral opportunistic infection of the brain caused by reactivation of the JC virus (JCV) leading to demyelination. 35 JCV is a polyomavirus belonging to the same family as BK- and SV-40 virus. 36 Latent JCV infection is often found in uroepithelial cells in the kidney and in the bone marrow37,38 Reactivation usually occurs in individuals with suppressed cellular immunity, e.g. in HIV-positive patients, patients with lymphoproliferative disorders, or immunosuppressed transplant recipients. It is believed that upon reactivation JCV is transported to the CNS via either B cells or as free virus. 35 In the CNS oligodendrocytes and other cells are infected by JCV leading to multifocal demyelination. In case of persisting immune deficiency this demyelination may be fatal. As mentioned above there have been 75 PML cases since the licensing of natalizumab in 2006. With more than 75000 patients treated with natalizumab the incidence of PML is 0.96 per 1000 (95% confidence intervall 0.76–1.21). While there were only very few cases during the first year of treatment the risk seems to increase after two years of treatment. Therefore, a renewed written consent has to be given by the patient when treatment is given for more than two years. Analysis of the first 28 cases with PML 39 under natalizumab treatment has revealed that the risk of PML is increased when patients where pretreated with immunosuppressants, mostly mitoxantrone. Of the 75 PML patients 15 (20%) had died, which is considerably lower compared to PML associated with other diseases like HIV or lymphoproliferative diseases. Of the surviving patients it is currently not possible to estimate the risk for permanent disability due to the PML, however, approximately 50% of the patients experience a severe disability (Karnofsky score <40) six month after diagnosis of PML (source: www.tysabri.de). Thus, PML remains a severe and potentially fatal side effect. Clinical alertness for symptoms of PML has to be kept high for both the treating physician as well as relatives and friends. Despite the lower incidence of PML during the first year of treatment there should always be a high alertness for symptoms since the early diagnosis seems to be crucial for a favourable outcome. Typical clinical symptoms of PML are cognitive impairments, change in personality, aphasia, hemianopsia, and epileptic seizures that are all less common as symptoms of MS. However, typical symptoms of an MS relapse, e.g. hemiparesis, may also be the first sign of PML. In case of a clinical suspicion of PML natalizumab has to be stopped, and a thorough work-up including brain MRI and a spinal tap for JCV PCR in the CSF have to be performed. In some cases even brain biopsy is required to secure the diagnosis of PML. Once PML is diagnosed natalizumab has to be stopped. The restoration of the immune system is the only proven therapy for PML.40,41 Therefore, removal of natalizumab by plasma exchange (PE) is suggested as primary therapy. 42 Unfortunately, the quick restoration of the immune system bears the risk of development of an immune reconstitution inflammatory syndrome (IRIS), an overshooting immune reaction in the brain. This is associated with unexplained clinical worsening that can include cerebral edema and enlarging inflammatory lesions with Gd-enhancement on MRI despite the improvement of the immune system. IRIS may be life threatening and requires intensive care treatment with high dose steroids. So far, all patients with natalizumab-associated PML developed IRIS regardless of the use of PE. The syndrome occurred days to weeks after PE while it occurred later in patients that did not receive PE. 39
Other treatments for PML may include mefloquin that has been shown in vitro to inhibit JCV replication. 43 Furthermore, it was shown in vitro that JCV enters cells via the serotonin receptor 5-HT(2A)R that may be blocked by mirtazapine. 44 Both substances have been utilised in patients with PML,45,39 however, there are no controlled studies and there is no evidence that these treatments have an impact on survival. Indeed, some of the fatal natalizumab related cases were even treated with these substances. 39
Currently it is assumed that PML develops due to a reactivation of JCV. Either there is persisting virus in the CNS or a temporary viremia is responsible in the setting of immunosuppression. Recent work has indicated that while the prevalence of anti-JCV antibodies in natalizumab treated patients was 53.6%, all of the 17 available pre-PML sera from patients that later developed PML were positive for anti-JCV antibodies. 46 This supports the assumption that development of PML is due to a reactivation rather than a primary infection with JCV. It is currently not clear if measurement of anti-JCV antibodies prior to therapy may help to assess the risk of development of PML. It has to be kept in mind that there is a rate of 2.5% of false negative tests with ELISA testing for anti-JCV antibodies, 46 possibly limiting the informative value of such an investigation. It is clear that measurement of JCV DNA in either serum or urine does not help to stratify for patients at higher risk for the development of PML.47,48
Conclusion
The administration of natalizumab is an effective treatment for patients with RRMS. Its mechanism of action has been specifically designed to interfere with the migration of inflammatory cells via the BBB into the CNS parenchyma. Due to the development of PML as a rare but severe and life treatening side effect its approval is restricted to patients who are either very active or who fail on other disease modifying treatments. The risk for the development of PML is currently approximately 1:1000 for a treatment duration of two years. There is evidence that this risk is increasing with longer treatment duration and thus the indication has to be reassessed after two years of treatment. Furthermore, prior immunosuppressive treatment seems to increase the risk for PML in patients treated with natalizumab. On the other hand, no prior treatment with immunosuppression translates into a lower PML risk. Current research avenues focus on the identification of risk parameters for the development of PML in order to develop future guidelines for the management and risk minimisation for patients with active RRMS.
Disclosures
This manuscript has been read and approved by all authors. This paper is unique and not under consideration by any other publication and has not been published elsewhere. MS has received lecture fees and travel support for attending meetings from Bayer, Baxter, BiogenIdec, MerckSerono, Sanofi-Aventis, Talecris and research support from Bayer, BiogenIdec, MerckSerono, and Teva. BCK has received honoraria for lecturing, travel expenses for attending meetings, and financial support for research from Baxter, Bayer Schering, Biogen Idec, Merck Serono, Novartis, Roche, Sanofi Aventis, Talecris and TEVA Neurosciences. The authors confirm that they have permission to reproduce any copyrighted material.
