Abstract
Therapies targeting epidermal growth factor receptor (EGFR) are a promising recent development in the treatment of metastatic colorectal cancer (mCRC). Panitumumab is a fully human anti-EGFR monoclonal antibody that competitively inhibits the binding of all known EGFR ligands, including epidermal growth factor and transforming growth factor alpha, to cells expressing EGFR. In patients with mCRC, panitumumab monotherapy has resulted in favorable clinical responses, including increases in objective response rate, stable disease rate, and progression-free survival. Panitumumab has also shown promising antitumor activity in combination with selected chemotherapy regimens. Responses and improvements in progression-free survival associated with panitumumab monotherapy in patients with mCRC appear to be confined to patients whose tumors express wild-type KRAS. Therapy with panitumumab is generally well tolerated; the most common adverse events observed include skin-related toxicities, gastrointestinal toxicities, and hypomagnesemia. Infusion reactions are rare, and the agent has low immunogenicity.
Keywords
Introduction
Standard-of-care therapeutic options for patients with advanced or metastatic colorectal cancer (mCRC) include a variety of chemotherapy regimens using 1 or more of the following active drugs: 5-fluorouracil (5-FU), leucovorin (LV), capecitabine, irinotecan, and oxaliplatin. 1 Despite the availability of these therapeutic options, colorectal cancer was estimated to have caused approximately 50,000 deaths in the United States in 2008. 2 Recent efforts to improve treatment of mCRC have focused on the development of therapies targeting tumor growth factors and their associated receptors and signaling pathways.3,4
The epidermal growth factor receptor (EGFR) is a clinically validated therapeutic target in mCRC. The EGFR (also known as HER) has many known ligands, which include epidermal growth factor, transforming growth factor alpha, epiregulin, β-cellulin, heparin-binding EGF, and amphiregulin. 5 Ligand binding induces receptor homodimerization or heterodimerization with other HER family members, recruitment of adapter proteins, and subsequent activation of a variety of signaling pathways including the Ras/Raf/mitogen-activated protein kinase (MAPK) pathway, the signal transducer and activator of transcription (STAT), and the phosphatidylinositol 3-kinase (PI3K)/AKT pathway. 5 Activation of intracellular effectors of these pathways leads to changes in transcription of numerous different genes involved in processes such as cell division, adhesion, migration, differentiation, and apoptosis. Dysregulation of these signaling pathways can ultimately result in phenotypic changes including neoplastic cell growth, survival, and metastatic spread. Expression of EGF and EGFR is frequently observed in colorectal tumors,6,7 and overexpression of EGFR has been associated with advanced tumor stages. 8 These observations suggest that dysregulation of EGFR signaling may play a significant role in the neoplastic proliferation of cells in colorectal tumors.7,9 Results of studies investigating EGFR overexpression as a prognostic indicator have been mixed. Higher grades of EGFR expression, 10 stronger membranous staining for EGFR, 11 the extent of positive immunohistochemical staining for EGFR, 12 and intensity of EGFR staining in deep tumor regions 6 have been associated with reduced patient survival time in some studies. These findings provide support for the potential utility of anti-EGFR agents as adjuvant therapy for mCRC. 10 However, other studies have not found an association between survival and EGFR expression. 8 Most studies have shown limited overexpression of other HER family members (such as HER2 and HER3),13–17 which supports the use of anti-EGFR agents in the treatment of mCRC.
Panitumumab is a fully human immunoglobulin (Ig) G2 kappa monoclonal antibody directed against the EGFR. It was originally produced using XenoMouse® technology. 18 Mice were immunized with the human cervical epidermal carcinoma cell line A431, which expresses high levels of EGFR, and panitumumab was selected during screening of anti-EGFR monoclonal antibodies based on its high affinity and specificity. 19 Because panitumumab is a fully human IgG2 antibody, it was anticipated that it would have a relatively long half-life, low rate of infusion reactions, and minimal immunogenicity with repeated administration. 18 Panitumumab is approved in the United States as a single agent for the treatment of EGFR-expressing mCRC that is refractory to chemotherapy, 20 and is approved in Europe in patients with EGFR-expressing mCRC with nonmutated (wild-type) KRAS after failure of chemotherapy. 21 In addition to panitumumab, cetuximab, a chimeric IgG1 monoclonal antibody directed against the EGFR, has been shown to be effective in the treatment of mCRC.22–24
Mechanism of Action and Pharmacodynamics of Panitumumab
Panitumumab binds to domain III of the EGFR, which is the ligand-binding domain, with high affinity (Kd, 5 × 10-11 mol/L), competitively blocking the binding of all known EGFR ligands to carcinoma cell lines that express EGFR.19,25 In addition, panitumumab treatment results in rapid internalization and downregulation of EGFR expression. 26 Furthermore, panitumumab inhibits cellular proliferation (potentially as a consequence of cell cycle arrest) and apoptosis and inhibits the production of proangiogenic factors such as vascular endothelial growth factor (VEGF) and interleukin 8 by tumor cells.27–29 As a result, panitumumab may inhibit tumor growth and progression directly by blocking tumor cell proliferation and indirectly by inhibiting tumor angiogenesis.
Panitumumab has also been shown to prevent tumor formation and to cause regression of established tumors in an in vivo tumor xenograft model. 19 In a murine minimum-residual-disease model, panitumumab treatment for 3 weeks completely prevented the formation of A431 tumor xenografts for 8 months, whereas mice treated with either saline or control antibodies developed tumors within 10 days. Similarly, in mice with established A431 xenografts, treatment with panitumumab for 3 weeks not only inhibited growth of the xenografts but also caused complete eradication of tumors for up to 250 days following treatment. 19 Subsequent analysis demonstrated that tumor inhibition in the A431 model was associated with plasma exposure levels of panitumumab that are similar to those seen in patients. 30 In addition, a significant reduction in phosphorylated EGFR levels in the tumors and a significant reduction in tumor markers of proliferation and MAPK signaling were observed. Similarly, inhibition of tumor growth has been observed in a variety of other mouse xenograft models, including models of colon cancer.31,32 Interestingly, the ability of panitumumab to inhibit A431 tumor xenograft growth in athymic nude mice appears to be dependent on the cell-surface expression of a threshold level of EGFR because panitumumab significantly inhibits the growth of tumors expressing high levels of EGFR per cell but not growth of tumors with lower EGFR expression. 32 However, EGFR expression, as measured by immunohistochemistry, has not been shown to correlate with activity among patients in clinical trials. 33
Pharmacokinetics of Panitumumab
Pharmacokinetic analysis of data from patients in a phase 1 study of panitumumab in metastatic renal cell carcinoma established that panitumumab concentrations increase nonlinearly in a greater-than-dose-proportional manner at doses up to 2 mg/kg. 34 The nonlinear increases in panitumumab concentration may have resulted from the gradual depletion (saturation) of available EGFR as a consequence of panitumumab binding and subsequent EGFR internalization. 34 At doses higher than 2 mg/kg, panitumumab (similar to other IgG antibodies) may have been cleared by the reticuloendothelial system via Fc receptor–expressing cells.34,35
Although the indicated dose of panitumumab is 6 mg/kg given once every 2 weeks as a 1-hour intravenous infusion, 20 other doses and schedules have been clinically tested. Doses of 9 mg/kg given every 3 weeks and infusion times of 30 minutes (following a well-tolerated first infusion of 60 min) for the 6-mg/kg dose have demonstrated similar pharmacokinetic properties and overall safety as the recommended dose. 36 These dose schedules had similar adverse event profiles, with the exception of greater incidence of treatment-related grade 3 adverse events with the 9-mg/kg dose than the 6-mg/kg dose; none of the treatment-related adverse events were more severe than grade 3 in any of the dose groups. 36 At the 6-mg/kg dose given every 2 weeks, steady-state concentrations are achieved by the third infusion of panitumumab,20,37 The mean area under the concentration versus time curve was 1306 μg·d/mL, the mean clearance was 4.9 mL/kg/d, and the mean elimination half-life was approximately 7.5 days. 20 There are no reported effects of age, sex, race, mild to moderate renal or hepatic dysfunction, or EGFR membrane-staining intensity on panitumumab pharmacokinetics. 20
Efficacy of Panitumumab in Metastatic Colorectal Cancer
Panitumumab as a monotherapy for metastatic colorectal cancer
In phase 1 and phase 2 clinical trials, treatment with panitumumab as a monotherapy for mCRC has been associated with favorable clinical responses.37–41 In a phase 1 open-label study of panitumumab monotherapy for patients with EGFR-expressing tumors, 39 of 96 patients enrolled had advanced treatment-refractory colorectal cancer. Of these, 5 (13%) had a partial response with a median response duration of 32 weeks (95% CI, 28-32 wk) and 9 (23%) had stable disease. 37 In an open-label phase 2 study in 148 patients with EGFR-expressing mCRC that had progressed despite chemotherapy, the overall response rate among patients receiving panitumumab (2.5 mg/kg once weekly for 8 wk) was 9% (95% CI, 5%-15%) and 29% of patients achieved stable disease. 40 Median progression-free survival (PFS) was 14 weeks (95% CI, 8-16 wk), and the median overall survival was 8.6 months (95% CI, 5.9-9.8 mo). 40 Response rates of 4% to 13% have been observed in other phase 2 trials.38,41
The US Food and Drug Administration approval of panitumumab as a monotherapy in mCRC followed a phase 3 trial in 463 patients with chemotherapy-refractory mCRC. 42 Patients who received panitumumab plus best supportive care (BSC) had significantly longer PFS (median 8.0 vs. 7.3 wk, respectively; hazard ratio [HR], 0.54; 95% CI, 0.44-0.66; stratified log-rank test P < 0.0001; Fig. 1) and a greater objective response rate (10% vs. 0%, respectively) compared with patients who received BSC alone. 42 Overall survival was similar among patients receiving panitumumab plus BSC and those receiving BSC alone (HR, 1.00; 95% CI, 0.82-1.22). However, it should be noted that patients in the BSC arm who had disease progression were eligible to receive panitumumab in a crossover study. Among patients who crossed over and received panitumumab (n = 176; 76% of BSC arm), 11% had an objective response and the median PFS was 9.4 weeks (95% CI, 8.0-13.4). 43 This efficacy of panitumumab among patients who crossed over likely explains the observed lack of overall survival benefit with panitumumab in this study.
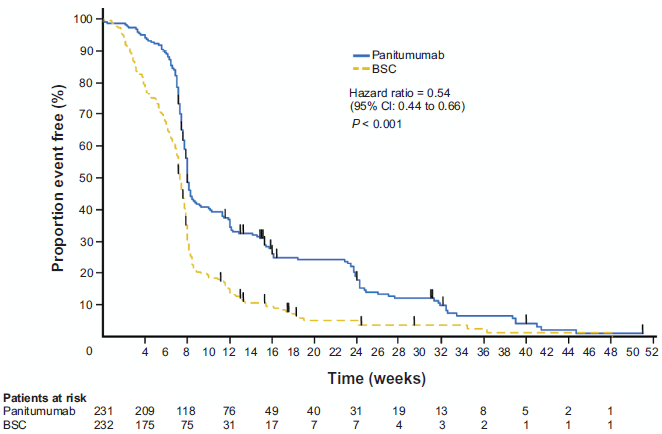
Progression-free survival among patients treated with panitumumab plus best supportive care (BSC) and patients receiving BSC alone. Median progression-free survival in the panitumumab group was 8.0 weeks (95% CI, 7.9-8.4) compared with 7.3 weeks (95% CI, 7.1-7.7) among patients in the BSC group. Adapted with permission from Van Cutsem E, Peeters M, Siena S, et al. 2007. Open-label phase III trial of panitumumab plus best supportive care compared with best supportive care alone in patients with chemotherapy-refractory metastatic colorectal cancer. Journal of Clinical Oncology. 25(13):1658-4l.
Predictors of response to panitumumab
Defining the correlates of responsiveness to panitumumab may help identify patients most likely to benefit from panitumumab treatment. Somewhat surprisingly, no association between EGFR tumor membrane expression and clinical response to anti-EGFR antibodies has been observed.33,39,44 Consequently, a number of clinical studies were conducted to further investigate the association between the level of EGFR expression and clinical benefit in patients with mCRC.38–41,43 The results of these studies showed that EGFR protein expression–-assessed by the proportion of tumor cells staining positive for EGFR on the membrane or by EGFR staining intensity per cell–-does not correlate with response to panitumumab. In a phase 2 study that evaluated the antitumor activity of panitumumab in patients with chemotherapy-refractory mCRC with low (1%-9%) or negative (<1%) tumor cell membrane EGFR expression, partial response rates of 5% and 4%, respectively, were observed. 39 In two further phase 2 studies in patients with tumors with ≥ 10% 1+ EGFR immunohistochemical membrane staining, response rates of 8% to 9% were observed.38,40 Moreover, among patients with ≥ 10% EGFR staining, there was no significant difference in clinical response when patients were stratified by “low” (2+ or 3+ EGFR staining intensity in <10% tumor cells) versus “high” (2+ or 3+ EGFR staining intensity in >10% tumor cells) EGFR membrane staining intensity. 40 As with panitumumab, clinical response to cetuximab does not appear to correlate with immunohistochemical EGFR protein expression.22,45
Some studies have suggested a genetic basis for the antitumor activity of monoclonal antibodies to EGFR, including increased EGFR gene copy number, which has been shown to correlate with better response to panitumumab and cetuximab.46,47 However, not all studies have consistently demonstrated the utility of this biomarker in mCRC.48–51 Studies suggest that assessment of EGFR copy number has the potential to help predict which patients are most likely to benefit from treatment with panitumumab, but clinical trials with prospectively defined scoring parameters are still needed to determine the utility of increased gene copy number as a biomarker. 52
Recent retrospective analyses of clinical trial data have demonstrated that mutations in the gene KRAS are predictive of lack of response to panitumumab among patients with mCRC. The KRAS gene encodes a signaling protein involved in the regulation of cell growth and proliferation, and oncogenic activating mutations of this gene have been implicated in the malignant transformation of tumor cells. 53 The presence of KRAS mutations is associated with a lack of response to panitumumab compared with wild-type KRAS (Table 1).54–56 Freeman et al examined the association of KRAS mutational status with response to panitumumab monotherapy in a retrospective analysis of data from 62 patients enrolled in 3 phase 2 studies that investigated panitumumab as a second-line therapy for mCRC. 56 Mutations in KRAS were observed in 24 (38.7%) of the patients; 38 (61.3%) had wild-type KRAS. Following panitumumab treatment, a partial response was seen in 10.5% of patients with wild-type KRAS, but in none of the patients with mutant KRAS. Likewise, stable disease was seen in 52.6% and 20.8% of patients with wild-type and mutant KRAS, respectively. PFS also favored the patients with wild-type KRAS versus mutant KRAS (HR, 0.4; 95% CI, 0.2-0.7; P = 0.002). 56 Similar results were observed in an analysis of data from a phase 3 trial that investigated panitumumab plus BSC or BSC alone in patients with chemotherapy-refractory mCRC. 54 Of the 427 patients included in the analysis, 184 (43%) of these patients had KRAS mutations. The response rate to panitumumab was 17% among patients with wild-type KRAS and 0% among patients with mutant KRAS. Moreover, among patients with wild-type KRAS who received panitumumab plus BSC, median PFS was 12.3 weeks, compared with 7.3 weeks among those who received BSC alone (Fig. 2; HR = 0.45; 95% CI: 0.34-0.59). Among patients with mutations in KRAS, median PFS was similar among those who received panitumumab plus BSC and those who received BSC alone (Fig. 2; 7.4 vs. 7.3 wk; HR = 0.99; 95% CI: 0.73-1.36; P < 0.0001). These results suggest that the antineoplastic activity of panitumumab in mCRC is mediated by inhibition of the RAS/RAF/MAPK signaling pathway and that KRAS mutations can interfere with this inhibition. 54 Several studies have also demonstrated that KRAS mutations predict a lack of response to cetuximab monotherapy among patients with mCRC.57–60 In the recent multicenter phase 3 CRYSTAL study, in which patients with mCRC received cetuximab plus infusional 5-FU, leucovorin, and irinotecan (FOLFIRI) or FOLFIRI alone as first-line therapy, patients with KRAS mutations had a significantly reduced response rate following treatment with cetuximab plus FOLFIRI compared with patients who received FOLFIRI alone. 24 However, although there was a trend toward improved PFS and overall survival among wild-type KRAS patients who received cetuximab, this was not statistically significant.
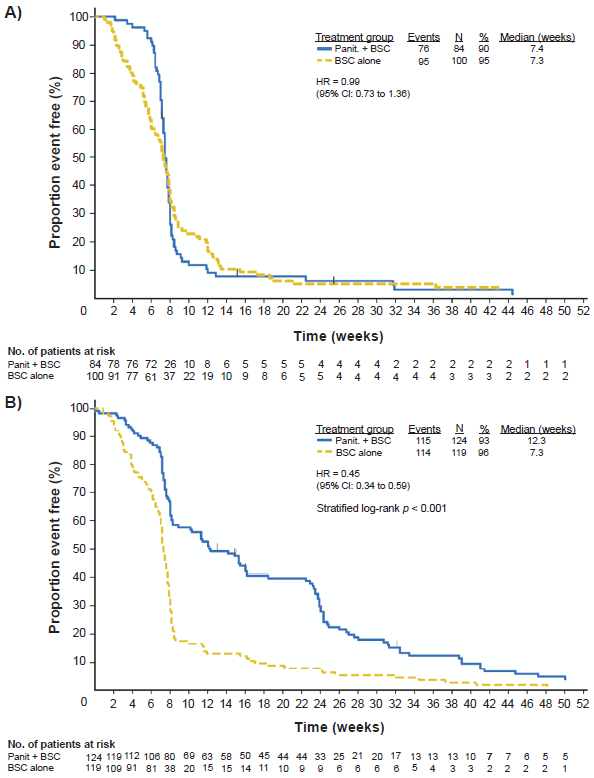
Progression-free survival among patients receiving panitumumab with (A) mutant KRAS and (B) wild-type KRAS. Hazard ratios (HRs) are shown for panitumumab (panit.) vs. best supportive care (BSC) adjusted for randomization factors. Adapted with permission from Amado RG, Wolf M, Peeters M, et al. 2008b. Wild-type KRAS is required for panitumumab efficacy in patients with metastatic colorectal cancer. Journal of Clinical Oncology. 26(10):1626-34.
Efficacy of panitumumab monotherapy among patients with wild-type or mutant KRAS.*
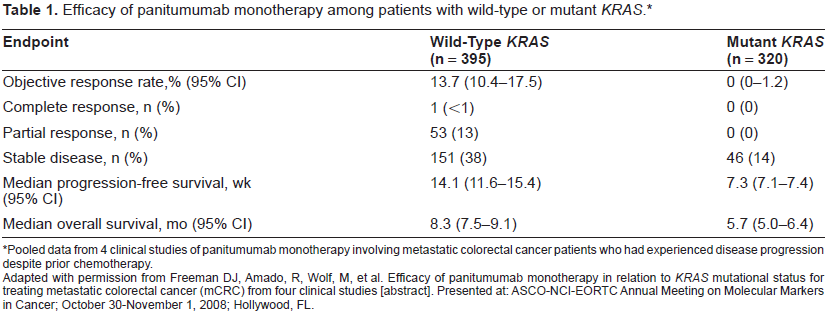
Pooled data from 4 clinical studies of panitumumab monotherapy involving metastatic colorectal cancer patients who had experienced disease progression despite prior chemotherapy.
Adapted with permission from Freeman DJ, Amado, R, Wolf, M, et al. Efficacy of panitumumab monotherapy in relation to KRAS mutational status for treating metastatic colorectal cancer (mCRC) from four clinical studies [abstract]. Presented at: ASCO-NCI-EORTC Annual Meeting on Molecular Markers in Cancer; October 30-November 1, 2008; Hollywood, FL.
Recent studies have focused on the investigation of the role of other mutations that might predict response to anti-EGFR monoclonal antibodies. Other genes that encode downstream effectors of the EGFR, including BRAF and PIK3CA, have been evaluated in a subset of patients from phase 2 and phase 3 studies.56,61 Among 79 patients with wild-type KRAS who received treatment with either panitumumab or the anti-EGFR monoclonal antibody cetuximab, 0 of 11 patients (0%) with a mutation in BRAF had a response during therapy, whereas 22 of 68 patients (32%) with wild-type BRAF had a response. 61 Furthermore, mutations in BRAF were associated with significantly shorter PFS and overall survival. Finally, a recent study demonstrated an association between mutations in PIK3CA and poor response to cetuximab and panitumumab. 62 However, a subsequent study found no association between PIK3CA mutations and response to cetuximab. 63 Moreover, because mutations in PIK3CA occur at a low prevalence and often occur in tumors that also harbor KRAS mutations, it is unclear whether their analysis will provide additional clinical utility as a biomarker.50,56,64,65 Clearly, further research in prospective sample sets is required to validate mutations in BRAF and PIK3CA as predictive biomarkers in mCRC.
Combination therapies with panitumumab for metastatic colorectal cancer
Panitumumab has shown encouraging antitumor activity when administered in combination with chemotherapy. In a small phase 2 study that investigated panitumumab plus FOLFIRI as first-line therapy for mCRC (n = 24), 33% of patients who received this combination had a partial response and 46% had stable disease. 66 With this combination, median PFS was 10.9 months and overall survival was 22.5 months. In a separate treatment group in the same study, the combination of panitumumab plus bolus 5-FU and irinotecan (n = 19) was associated with a partial response rate of 47%, median PFS was 5.6 months, and median overall survival was 17 months. However, this combination of panitumumab plus bolus 5-FU and irinotecan was associated with a high incidence of grade 3/4 diarrhea and was poorly tolerated. 66 The Panitumumab Regimen Evaluation in Colorectal Cancer to Estimate Primary Response to Treatment (PRECEPT) trial is an ongoing phase 2 trial that will assess the effect of KRAS status on response to panitumumab plus FOLFIRI. 67 This study is fully enrolled with 102 patients with available KRAS mutational status. Early results showed that numerical differences in favor of patients with wild-type KRAS were observed for PFS and overall survival. 67
Additional phase 3 trials of panitumumab in combination with chemotherapy for early lines of therapy for mCRC are ongoing68–70 and will provide additional information on the safety and efficacy of these treatment regimens. The PRIME (Panitumumab Randomized Trial In Combination With Chemotherapy for Metastatic Colorectal Cancer to Determine Efficacy) study includes patients with mCRC that have not previously received treatment and investigates the safety and efficacy of panitumumab (6 mg/kg once every 2 weeks) in combination with FOLFOX compared with FOLFOX alone.68,70 The 20050181 study is assessing the safety and efficacy of panitumumab (6.0 mg/kg once every 2 weeks) plus FOLFIRI compared with FOLFIRI as a second-line treatment for mCRC. 69 Initial safety data from the pooled patient population have recently been reported (see Safety and Tolerability section). Both the PRIME and 20050181 trials have been amended to analyze outcomes according to tumor KRAS status.
The phase 3 Panitumumab Advanced Colorectal Cancer Evaluation (PACCE) study assessed the efficacy of panitumumab in combination with the anti-VEGF antibody bevacizumab and either oxaliplatinor irinotecan-based chemotherapy as first-line therapy for mCRC. 71 The combination was associated with poor responses and exacerbated toxicity in both cohorts. Addition of panitumumab to the treatment regimen was associated with reduced PFS in both the oxaliplatin (10.0 vs. 11.4 mo) and irinotecan (10.1 vs. 11.7 mo) cohorts. Addition of panitumumab was associated with shorter PFS regardless of KRAS status (wild type or mutant). Furthermore, patients receiving panitumumab in addition to bevacizumab and chemotherapy experienced a higher incidence of grade ≥3 adverse events than patients receiving bevacizumab and chemotherapy. Similar results were observed in the CAIRO2 trial, a phase 3 study of bevacizumab, capecitabine, and oxaliplatin with or without cetuximab as a first-line treatment of mCRC in 736 patients. 72 The addition of cetuximab to the treatment regimen reduced the median PFS (9.4 vs. 10.7 mo without cetuximab; P = 0.01) and was associated with an increased incidence of grade 3 or 4 adverse events. 72 Addition of cetuximab was associated with shorter PFS in patients with mutant KRAS as compared with patients with wild-type KRAS and those not receiving cetuximab with mutant KRAS (8.1 vs. 10.5 mo). Among patients with wild-type KRAS, there was no significant difference in PFS with or without cetuximab. 72 The negative effect of the combination of bevacizumab, capecitabine, oxaliplatin, and cetuximab on PFS appeared to be isolated to the mutant KRAS group. These results with cetuximab suggest that the diminished responses and exacerbated toxicities seen in PACCE are a class effect of anti-EGFR antibodies rather than being specific to panitumumab. Moreover, because there is no clear biologic hypothesis based on KRAS status to explain outcomes, there is no clear explanation for the variability in results observed between PACCE and CAIRO2.
Safety and Tolerability of Panitumumab in Colorectal Cancer
In a pooled safety analysis (N = 920) of 10 trials of panitumumab as monotherapy for mCRC, the most frequently occurring adverse events were mild to moderate and included skin-related toxicities, gastrointestinal toxicities, and hypomagnesemia (Table 2). 73 The incidence of adverse events of any grade was 92% and the incidence of grade 3 toxicities was 13%. Overall, 12% of patients receiving panitumumab discontinued because of any toxicity and 2% discontinued because of skin-related adverse events.
Panitumumab Combined Safety Analysis Across 10 Clinical Trials (N = 920).
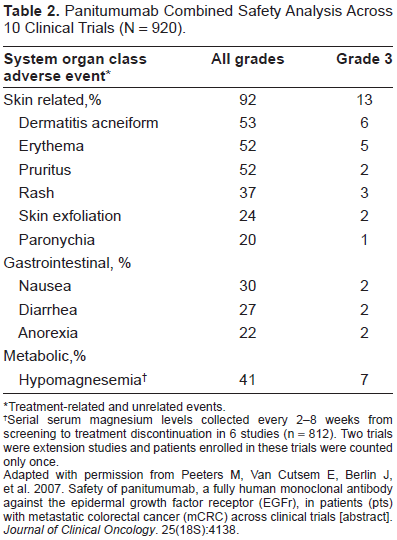
Treatment-related and unrelated events.
Serial serum magnesium levels collected every 2-8 weeks from screening to treatment discontinuation in 6 studies (n = 812). Two trials were extension studies and patients enrolled in these trials were counted only once.
Adapted with permission from Peeters M, Van Cutsem E, Berlin J, et al. 2007. Safety of panitumumab, a fully human monoclonal antibody against the epidermal growth factor receptor (EGFr), in patients (pts) with metastatic colorectal cancer (mCRC) across clinical trials [abstract]. Journal of Clinical Oncology. 25(18S):4138.
Although EGFR inhibitors are well tolerated in general, skin toxicity is a very common adverse event associated with this entire class of agents. 74 The adverse effects of EGFR inhibitors on skin include a pruritic acneiform eruption, xerosis, and telangiectasia. 74 Following panitumumab administration, skin toxicity occurs in a dose-dependent manner with virtually 100% incidence at doses ≥2.0 mg/kg once weekly, although it is generally well tolerated. 34 The appearance of skin toxicity in panitumumab-treated mCRC patients has been associated with improvement in the objective response rate, PFS, and overall survival.34,75,76 Furthermore, among patients receiving panitumumab, severe skin toxicity has been associated with better health-related quality of life (HRQoL) and improved symptoms, as compared with patients who experience minimal skin toxicity. 76 Effective management of skin toxicity is important to avoid any compromise of therapeutic compliance due to discomfort or cosmetic issues. 74 The Skin Toxicity Evaluation Protocol With Panitumumab (STEPP) study found that preemptive treatment of skin toxicity associated with panitumumab in combination with chemotherapy was associated with improved patient-reported quality of life (QoL) and reduced the incidence of grade ≥2 skin toxicity by >50% compared with reactive treatment of skin toxicity. 77
Infusion reactions are rare, and anti-panitumumab antibody formation occurs in <5% of panitumumab-treated patients.43,73,78 The rate of anti-panitumumab antibody formation appears to be dependent at least in part on the assay used. In one study, anti-panitumumab antibodies were identified in 0.3% of patients when measured by ELISA and in 4.2% of patients when measured by a Biacore assay. 78 Pooled data from 920 patients across 10 clinical trials indicated that grade 3 infusion reactions occurred in 0.4% of patients. 73 Additionally, the occurrence of persistent anti-panitumumab antibodies after dosing was seen in 4.6% of patients. Importantly, the development of anti-panitumumab antibodies does not appear to affect panitumumab pharmacokinetics or cause additional toxicity. 20
Panitumumab is also well tolerated when used in combination with chemotherapy. In a phase 2 trial, 24 patients with mCRC were treated with panitumumab in combination with infusional FOLFIRI. 66 All patients experienced skin-related toxicity (13% had grade 3 events; none had events grade ≥4), and grade 3/4 diarrhea occurred in 25% of patients. The incidence of diarrhea with this combination therapy was similar to that seen in other studies with FOLFIRI alone. In contrast, as described above, panitumumab in combination with bolus 5-FU and irinotecan (n = 19) was poorly tolerated. 66 Interim safety data from the phase 3 PRIME and 181 studies have yielded similar results.69,70 In a pooled analysis of data from 1097 patients from all arms of the 20050181 study, the most frequently occurring adverse events were skin and subcutaneous events (63%; grade 3/4, 15%), diarrhea (56%; grade 3/4, 10%), nausea (43%; grade 3/4, 2%), and fatigue (30%; grade 3/4, 5%). 69 In a pooled analysis of 903 patients from the PRIME study, the most frequently occurring adverse events were skin and subcutaneous events (56%; grade 3/4, 11%), diarrhea (47%; grade 3/4, 11%), neutropenia (44%, grade 3/4, 28%), and nausea (42%; grade 3/4, 3%). 70
Interestingly, some patients with mCRC who have experienced hypersensitivity or severe or life-threatening infusion reactions to cetuximab have been able to tolerate panitumumab without experiencing any hypersensitivity or infusion reactions.79,80 This suggests that some patients who have experienced infusion reactions with cetuximab may safely receive panitumumab, although this hypothesis has not been formally evaluated in the clinical setting.
Patient-Reported Outcomes with Panitumumab
Patient-reported outcomes, including HRQoL and symptoms, are important considerations, particularly for patients with progressive disease that is refractory to chemotherapy. Disease progression occurs as a result of tumor growth and is usually associated with progression of symptoms and diminished QoL. Results from a phase 3 study demonstrated that, following panitumumab treatment for mCRC, lack of disease progression is also associated with improved symptom scores and higher HRQoL.81,82 The achievement of at least stable disease among all patients (those treated with panitumumab or with BSC) at week 8 was associated with clinically and statistically significant improvements in disease-related symptoms.81,82 Furthermore, among patients treated with panitumumab, lack of tumor progression at 8 weeks was associated with better HRQoL score. No difference in HRQoL was observed among those with stable disease or better who were receiving BSC. The association between lack of progression of disease and improvement in HRQoL may be attributable to a greater reduction in tumor burden with panitumumab during stable disease 82 and demonstrates that delaying disease progression with panitumumab therapy is associated with clinical benefits beyond a longer progression-free survival time. Significant improvements in symptoms and HRQoL with panitumumab versus BSC were observed only in chemotherapy-refractory patients with wild-type KRAS and not in patients with mutant-type KRAS, 83 supporting the usefulness of KRAS mutational status as a biomarker for patients likely to realize clinical benefit from panitumumab therapy.
Conclusions: Role of Panitumumab in Therapy
In summary, data have shown that panitumumab is well tolerated and has antitumor activity as a monotherapy in patients with refractory mCRC. Panitumumab is approved for patients with refractory mCRC in the United States and Australia, and in Europe and Canada for these same patients with tumors expressing wild-type KRAS. Furthermore, use of panitumumab as a therapy after first or second progression for mCRC patients with wild-type KRAS who are unable to tolerate cetuximab plus irinotecan is included in current treatment guidelines. 1
KRAS mutations have been identified as a significant predictor of lack of response to panitumumab monotherapy for mCRC. Therefore, treatment with panitumumab may be appropriate for patients with wild-type KRAS. In the future, screening of patients for KRAS mutations may play an important role in determining the treatment approach for patients with mCRC. For patients without KRAS mutations, screening for mutations in other genes, including downstream effectors of the MAPK and PI3K signaling pathways, may potentially help determine the most appropriate approach for treatment of mCRC with panitumumab, although this will require further clinical validation.
Based on the results of recent clinical studies, panitumumab appears to have antitumor activity and acceptable toxicity in combination with irinotecan-based chemotherapy. Currently, the role of panitumumab in combination with various chemotherapy regimens for mCRC remains to be determined. Two large ongoing phase 3 studies will provide additional information on the utility of panitumumab in combination with FOLFOX and FOLFIRI chemotherapy for earlier lines of treatment of mCRC. Furthermore, these studies will investigate the role of KRAS and other molecular markers as predictors of response. Preliminary safety data from these studies suggest that treatment with panitumumab in combination with chemotherapy is well tolerated.
In addition to its use in mCRC, panitumumab is also being investigated as therapy for advanced head and neck cancer. An initial study suggested that the combination of panitumumab and chemoradiotherapy is safe and has promising efficacy in patients with stage III or IV head and neck cancer. 84 A number of phase 2 studies and a pivotal phase 3 study have been initiated to investigate the safety and efficacy of panitumumab in combination with a variety of chemotherapy and/or radiotherapy regimens for the treatment of locally advanced, metastatic, or recurrent head and neck cancer. Data from all of these key studies are highly anticipated and will be available soon. These will provide definitive evidence for the role of panitumumab in early lines of treatment for mCRC and in head and neck cancer.
Disclosure
The author is an employee and stock holder of Amgen Inc.
Footnotes
Acknowledgment
The author would like to thank Amy Yellen-Shaw, PhD, Benjamin Scott, PhD, and Margit Rezabek, DVM, PhD (Complete Healthcare Communications, Inc.), whose work was funded by Amgen Inc., for assistance in the preparation of this manuscript.
