Abstract
Ritonavir-boosted lopinavir (LPV/r) is a boosted protease inhibitor (PI) combination used as an alternative agent in the management of the human immunodeficiency virus (HIV). HIV treatment consists of combinations of classes of medications with complementing mechanisms of action. Protease inhibitor-based therapy is one of the mainstays of therapy. Ritonavir-boosted lopinavir can be dosed once- or twice-daily based on previous antiretroviral therapy, patient characteristics, and concomitant medications. It is currently available as an oral solution or tablet formulation and can be given without regard to food if given as a tablet. Lopinavir is extensively metabolized via the cytochrome (CYP) P450 3A4 and 3A5 hepatic isoenzymes and ritonavir is a potent inhibitor of CYP34A. Co-administration of low-dose ritonavir leads to increased plasma concentrations of lopinavir. There is potential for numerous drug-drug interactions when additional medications are administered. Clinical trials have been reported previously demonstrating the safety and efficacy of LPV/r in children, adolescents and adults and it is readily used throughout these populations. Current clinical trials are focused on examining the efficacy and toxicity of boosted-PI monotherapy as a treatment option in previously suppressed adults.
Introduction
The goal of highly active antiretroviral therapy (HAART) is to reduce the morbidity and mortality associated with the human immunodeficiency virus (HIV). The current regimens that are recommended for adult and adolescent patients include a backbone of 2 nucleos(t)ide reverse transcriptase inhibitors (NRTIs) and one of the following types of agents: non-nucleoside reverse transcriptase inhibitor (NNRTI), ritonavir-boosted protease inhibitor (PI), or integrase strand transfer inhibitor (ISTI). Lopinavir/ritonavir (LPV/r) is currently listed as an alternative PI-based regimen for adults and adolescents and the preferred regimen for pregnant women and pediatric patients.1–4 This combination is the only co-formulated ritonavir-boosted PI available and can be dosed once-daily or twice-daily based on previous antiretroviral therapy (ART), patient characteristics, and concomitant medications. 5 This review will focus on the pharmacodynamics, pharmacokinetics, efficacy, safety and place in therapy of LPV/r.
Mechanism of Action, Metabolism and Pharmacokinetic Profile
HIV-1 protease is an essential component to the development of mature, infectious HIV virions. Inhibition of HIV-1 protease prevents the cleavage of the Gag-pol polyprotein which leads to a non-infectious HIV virion. Lopinavir and ritonavir are two of the nine PI's available for use in the management of the HIV infection. When lopinavir and ritonavir are given in combination, ritonavir is utilized as a boosting agent as it inhibits the metabolism of lopinavir to its inactive metabolites. Lopinavir/ritonavir is available as an oral solution and tablet in the United States and was previously available in a capsule formulation.
Numerous pharmacokinetic studies have been completed studying the capsule formulation of LPV/r and key pharmacokinetic parameters are listed for two such studies in Table 1. The protein-binding corrected concentration of lopinavir required to inhibit the replication of wild-type HIV by 50% (EC50) is 0.07 μg/mL and was surpassed in both study populations for both dosing regimens.6,7 The tablet formulation of LPV/r has been shown to be bioequivalent to the liquid and capsule formulations in both the 400/100 mg twice-daily dosing and 800/100 mg once-daily dosing regimens. 8 A comparison of the mean serum pharmacokinetics of the soft-gelatin capsule and the tablet demonstrated Cmin values were 5.17 μg/mL and 5.64 μg/mL respectively and Cmax values were 6.79 μg/mL and 10.26 μg/mL respectively. Differences between these serum levels were not statistically significant. 8
Steady-state pharmacokinetic parameters for lopinavir.
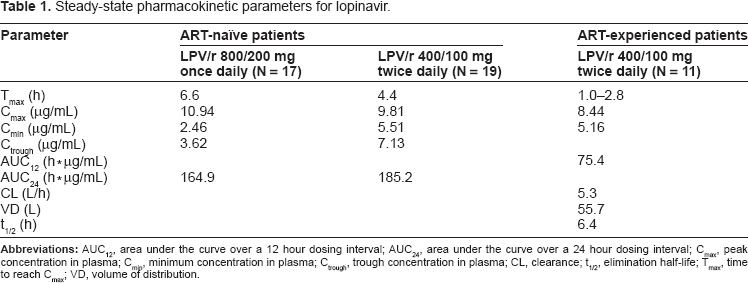
Lopinavir/ritonavir formulated as a capsule or liquid has shown increased bioavailability following a moderate to high-fat content meal and thus it is recommended in the prescribing information that these formulations be taken along with this type of a meal. 5 When evaluating the pharmacokinetic differences between the formulations in relation to meals it was found that the tablet formulation led to more consistent lopinavir and ritonavir exposure than the capsule or liquid and that ingestion of a meal did not impact the bioavailability of LPV/r to a meaningful extent. 9 Therefore, the prescribing information for the tablet formulation of LPV/r recommends that it can be taken with or without food.
Lopinavir/ritonavir is highly protein-bound at steady-state, 98%–99% bound to plasma proteins, primarily albumin and alpha1-acid glycoprotein. HIV replication occurs at the intracellular level thus the penetration of drugs such as lopinavir is an important characteristic to examine. One such study found that following the administration of 400/100 mg LPV/r capsules, the EC50 was reached in both the plasma and peripheral blood mononuclear cells with a Cmax of 8.44 μg/mL and 13.40 μg/mL, respectively. 7 Due to HIV-associated neurocognitive complications, effective distribution to the central nervous system (CNS) is also a key component to evaluate. Lopinavir/ritonavir penetrates the CNS and reaches concentrations above the EC50 by more than 5-fold. 10
Lopinavir is extensively metabolized via the cytochrome (CYP) 3A4 and 3A5 hepatic isoenzymes. Ritonavir is a potent inhibitor of CYP3A4 and thus co-administration of lopinavir and ritonavir leads to increased plasma concentrations of lopinavir. When given in concert, ritonavir is given at a lower dose than when administered as monotherapy and acts as a boosting agent, while lopinavir is the component eliciting the antiretroviral activity.11,12 Elimination of lopinavir and its metabolites is primarily via the fecal route. Following a 400/100 mg LPV/r dose, 82.6% of the dose was excreted in the feces and only 10.4% was excreted via the urine. 13
Difference in patient gender has not been found to have an effect on LPV/r concentrations. 14 Exposure to LPV/r has been found to be lower in the third trimester of pregnancy, but further research has found that the standard dosage for pregnancy results in adequate therapeutic levels for antiretroviral suppression.15,16 Administration of LPV/r oral liquid to pediatric patients results in a similar pharmacokinetic profile to that observed in adults.17,18 Due to the extensive hepatic metabolism of lopinavir, patients with hepatic impairment may have increased concentrations and caution should be used when administering LPV/r to such patients. 19 In patients with renal impairment, it is estimated that LPV/r concentrations will not be affected due to the minimal renal excretion. 20
In adult patients, the appropriate dosing regimen for LPV/r is 400/100 mg twice-daily or 800/200 mg once-daily. Once-daily dosing is acceptable only if patients have less than three of the lopinavir resistance-associated genetic mutations. The resistance substitutions include L10F/I/R/V, K20M/N/R, L24I, L33F, M36I, I47V, G48V, I54L/T/V, V82A/C/F/S/T, and I84V. The most common mutations include M46I/L, I54V, and V82A. 21 Significant more adults with ≤5 lopinavir-associated mutations at baseline achieved a virological response (>1 log10 decrease in plasma HIV RNA and or plasma RNA < 500 copies/mL) at 6 months in the expanded access programme [EAP]. 22 Lopinavir/ritonavir tablets may be administered with or without food and should be swallowed whole, not chewed, broken or crushed. Lopinavir/ritonavir capsules and oral solution must be refrigerated and taken with food, which reduces full utilization of the drug.
Clinically significant drug-drug interactions may occur when patients receive LPV/r due to CYP3A isoenzyme substrates. Therefore, physicians, pharmacists, and other care providers involved with treating HIV-1-infected patients need to collect drug history information from these patients to determine clinically significant drug drug interactions. In addition, the inhibition by ritonavir of the CYP3A and CYP2D6 enzymes may also lead to increased concentrations of other medications that are co-administered with LPV/r.11,12 The clinical impact of these drug-drug interactions varies from cautionary observation to contraindication for concomitant use. Medications that are contraindicated for use with LPV/r are detailed in Table 2. 5
Medications contraindicated for use with LPV/r.
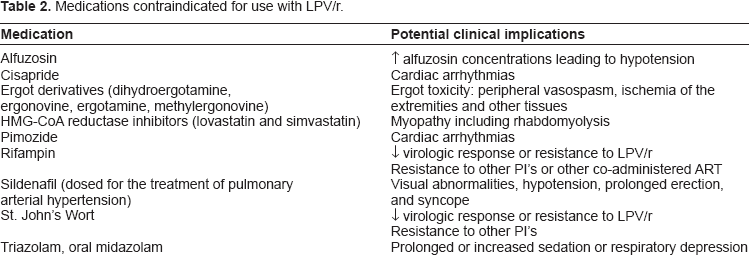
Dose adjustments and other precautionary measures need to be taken when LPV/r is co-administered with certain antiretroviral agents. Lopinavir plasma concentrations are decreased when given along with efavirenz, nevirapine, amprenavir, fosamprenavir/r, nelfinavir, and tipranavir. Lopinavir/ritonavir should not be dosed once daily when given with efavirenz, nevirapine, amprenavir or nelfinavir and the dose of LPV/r should be increased to 500/125 mg twice daily if co-administered with these agents. 5 Additionally, the oral solution should be increased to 533/133 mg twice daily if administered with these agents. 5 Increased concentrations of lopinavir can occur with administration of delavirdine and caution should be taken. Increased concentrations of tenofovir, amprenavir, indinavir, nelfinavir, saquinavir and maraviroc can occur when administered with LPV/r and the following dose adjustments should be made: decrease indinavir to 600 mg twice daily, saquinavir 1000 mg twice daily, and maraviroc 150 mg twice daily. Abacavir, zidovudine, and fosamprenavir/r concentrations can be decreased with co-administration with LPV/r however the clinical implications have not been established. Potentially clinically significant drug interactions for other classes of medications are detailed in Table 3. 5
Potentially significant drug interactions and clinical implications.

Clinical Trials
The phase II and III clinical trials that were used for FDA approval have been reported previously.23–25 A literature review was conducted to gather clinical trials that were published using PubMed from 2007 forward to summer 2010. Clinical trials were gathered from electronic citations using PubMed. Key terms that were used included “lopinavir-ritonavir, clinical trial, and humans”. The citations that were obtained from the search strategy were further clarified by only English language. The clinical trials were read and additional literature was gathered from the references of the published clinical trials if these citations were not part of the original search strategy. Currently, there are several additional trials that will be discussed including monotherapy trials to reduce pill counts for HIV-1 positive patients.
The ACTG A5073 trial was a 48-week randomized controlled trial comparing once daily versus twice daily combination antiretroviral therapy for treatment naïve patients. 26 Patients received 400 mg lopinavir and 100 mg ritonavir twice daily or 800 mg lopinavir and 200 mg ritonavir once daily. All patients received emtricitabine and stavudine or tenofovir once daily. Patients were stratified by HIV RNA level at screening into groups (<100,000 copies/ml or ≥100,000 copies/ml). The results depended on the HIV RNA level. The twice daily LPV/r regimen produced a significantly better sustained virologic response (SVR) in the higher HIV RNA group. However, adherence to prescribed regimen was significantly higher in the QD arm as compared to the BID arm (91% versus 80%, P < 0.001). Therefore, once daily therapy of LPV/r is efficacious for patients with <100,000 copies/ml and provides significantly better adherence to the prescribed regimen.
The MONARK trial was a prospective, open-label, randomized 96-week trial comparing safety and efficacy of LPV/r monotherapy with standard LPV/r plus zidovudine and lamivudine regimen as initial treatment regimen in HIV-infected patients with HIV-RNA levels <100,000 copies/ml. The primary endpoint was the proportion of patients with HIV-RNA levels below 400 copies/ml and <50 copies/ml at week 24 and week 48. A total of 83 and 53 patients were enrolled in the monotherapy or triple-drug groups. At week 48, 64% of patients in the monotherapy arm and 75% in the triple-drug arm achieved the primary endpoint. 27 This difference was not significant. Additionally, an evaluation of the impact of 48 weeks of LPV/r alone or using standard combination therapy on cell-associated HIV-DNA load was performed. 28 A total of 46 patients receiving monotherapy and 26 patients receiving triple therapy comprised the study population and HIV-DNA was quantified in whole blood using real-time PCR. Responders in the monotherapy arm had a significantly lower baseline HIV-DNA load than non-responders 2.86 versus 3.16 log copies/10 6 leucocytes, respectively (P = 0.05). Finally, resistance testing was performed at the time of initial screening and at the time of virologic failure in patients enrolled into this trial. 29 Drug resistance testing was performed for 5 of 53 patients on triple therapy and 33 of 83 on monotherapy. Major protease inhibitor (PI) resistance-associated mutations were detected in 6% of patients treated with LPV/r monotherapy by week 96 of the study. This suggests that LPV/r mono-therapy may be less than optimal therapy and lead to PI resistance-associated mutations. The long-term efficacy of LPV/r monotherapy was assessed several times.30,31 In these trials patients with HIV suppressed to <50 copies/ml for a minimum of 6 months without previous failure while receiving a PI-based plus 2 nucleosides inhibitors. Patients were randomly switched to LPV/r monotherapy or continued their current triple combination therapy. At 96-weeks, 87% remained on monotherapy with non-detectable viral load (<50 copies/ml) compared to 78% receiving triple therapy. Twelve patients (12%) in the monotherapy arm necessitated reintroduction of nucleosides. Discontinuations due to adverse events were significantly more frequent in the triple therapy group (8%) compared to the monotherapy group (0%, P < 0.01). Campos et al determined by Cox proportional hazards modeling that adherence levels and higher baseline CD4 T cell counts were associated with maintaining virologic suppression for lopinavir/ritonavir monotherapy. 32
Several studies have been reported that compare efavirenz or ritonavir-boosted other protease inhibitor with 2 nucleoside reverse transcriptase inhibitors (NRTIs) to LPV/r with 2 NRTIs.33–36 The goal of these trials is the proportion of patients with HIV-1 RNA levels <50 copies/ml at the endpoint time (48 weeks or 144 weeks). The study by Pulido et al assessed the long-term efficacy and safety of Fosamprenavir + ritonavir (FPV/r) compared to LPV/r both in combination with abacavir/lamivudine over 144 weeks. 34 Three patients experienced virologic failure from week 48 through week 144 (1 FPV/r; 2 LPV/r) and 13% (FPV/r) and 9% (LPV/r) of patients experienced diarrhea as the most common adverse event in this study extension. The Gemini study was a non-inferiority trial comparing saquinavir/ritonavir to LPV/r as initial therapy for HIV-1 adults over 48 weeks. 36 A similar proportion of patients in each group had HIV-1 RNA levels to <50 copies/ml and similar rates and severity of adverse events were noted. There was a difference in lipid levels between the two groups with LPV/r demonstrating a significantly higher change from baseline at week 48. The SWITCHMRK 1 and 2 trials were initiated as a study design to reduce the lipid abnormalities associated with regimens that included lopinavir/ritonavir. These trials switched raltegravir for LPV/r in combination antiretroviral regimens. Patients were eligible if their viral loads were below the limit of assay quantification for a minimum of 3 months while receiving a LPV/r-based regimen. A total of 707 patients were randomized and enrolled. At week 24, 84% of raltegravir patients and 91% of lopinavir/ritonavir patients had non-detectable viral loads to <50 copies/ml. Significant reductions in total cholesterol, non-HDL cholesterol, and triglycerides were reported in the raltegravir group. The study was terminated at week 24 because of lower than expected virologic efficacy in the raltegravir group. 37 Finally, the ARTEMIS trial compared darunavir/ritonavir to lopinavir/ritonavir in treatment naive HIV-1-infected patients. All patients received darunavir/ritonavir 800/100 mg once daily to lopinavir/ritonavir 800/200 mg total daily dose (once or twice daily) plus fixed dose tenofovir/emtricitabine. At 96 weeks, statistically greater percentage of patients displayed a non-detectable viral load (defined as <50 copies/ml) with darunavir/ritonavir (79%) compared to lopinavir/ritonavir (71%) P = 0.012. Additionally, fewer darunavir/ritonavir patients had grade II–IV side effects as compared to lopinavir/ritonavir. Therefore, this comparative trial showed a superior virologic response and lower side effects of darunavir/ritonavir. 38
Several trials were reported that compared the use of LPV/r with efavirenz as a nucleoside-sparing regimen.33,35,39,40 The dosing for LPV/r was 533.3/133.3 mg twice-daily along with efavirenz 600 mg daily. The proportion of patients (n = 86) with HIV-1 RNA levels <50 copies/ml at 48 weeks was 69% using an intent-to-treat analysis. 40 This lead to a comparison of efavirenz + 2 NRTIs and LPV/r + 2 NRTIs and LPV/r + efavirenz as initial treatment of HIV-1 infection. 39 Patients (n = 757) were enrolled into this open-label study. Follow-up was 96 weeks and the proportion of patients with HIV-1 RNA < 50 copies/ml was 89% in the efavirenz group, 77% in the LPV/r group and 83% in the NRTI-sparing group. This showed a statistically significant difference between the efavirenz and LPV/r groups but no difference was determined for the NRTI-sparing group. No difference in grade 3 or 4 adverse events was found in this trial (18% among groups). Virologic failure was significantly more prevalent in the NRTI-sparing group (9% of the efavirenz group, 6% in the LPV/r group and 16% in the NRTI-sparing group; P < 0.05). This led to the conclusion that virologic efficacy of the NRTI-sparing regimen was similar to that of an efavirenz regimen but associated with more drug resistance.
There are concerns about the use of boosted protease inhibitor (PI) as monotherapy for the treatment of HIV-infected patients. Several trials discussed above have documented lower efficacy with PI monotherapy as compared to PI-based combination therapy. Additionally, risks of low-level viremia are problematic as well as an increased risk for the development of resistance. The durability of the therapy is uncertain as the numbers of patients in PI monotherapy-based clinical trials have been low compared to the numbers of patients who are undergoing treatment. The level of adherence required for PI-based monotherapy is higher compared to combination therapy and therefore may only be a treatment option for those patients who are faithfully adherent in their therapy. Finally, the efficacy in reservoirs (ie, central nervous system or genital organs) is uncertain. This could lead to an increased risk of transmitting PI-resistant virus or development of HIV-associated dementia. There are several advantages associated with the use of boosted PI monotherapy. Even though the efficacy in clinical trials is less compared to combination therapy, the difference for PI-based monotherapy is small and rarely reaches statistical significance. The risk of low-level viremia has been shown to be reversible after reintroduction of NRTI therapy. It is relatively easy to identify patients that require reintroduction of NRTIs. There are on-going clinical trials of boosted PI-based monotherapy and most of the reported clinical trials have demonstrated efficacy over the course of two-years. This is encouraging for those patients that are eligible to reduce their pill burden. There is clearly an economic benefit to boosted PI monotherapy and without NRTIs, there are fewer metabolic disturbances. All of these concerns and advantages of boosted PI-based monotherapy should be part of the discussion for the HIV-infected patient when contemplating this switch to monotherapy.
Several trials have been reported for the use of LPV/r in HIV-1 infected children.18,41 Early initiation of therapy with LPV/r zidovudine and lamivudine reduced mortality by 76% and HIV progression by 75% in infants 6–12 weeks of age as part of the CHER trial. It is important to note that infants and children have rapid pharmacokinetic clearance of antiretroviral drugs. From the studies in this age group, LPV/r-based therapy in doses of 300/75 mg/m2 twice daily appear to be well tolerated and result in virologic control in approximately 80% of children.
Patient Preference
There are currently 9 available PI's for use in patients with HIV. As a class, PI's have a higher genetic barrier to resistance as compared to other antiretroviral agents and resistance to boosted-PI's is uncommon with failure. Some disadvantages seen with the use of these agents include metabolic complications such as dyslipidemia, insulin resistance, hepatoxicity, gastrointestinal side effects, and a high propensity for drug-drug interactions due to these agents being substrates and inhibitors of the CYP3A4 hepatic enzyme. If it has been determined that a PI-based antiretroviral combination would be the best choice for a given patient, various factors need to be taken into consideration when determining which PI to use. 4
Adherence to ART is crucial for suppression of viral load, thus the likelihood of adherence to the chosen therapy is an important factor to consider when selecting a regimen. As regimen complexity increases (eg, pill burden and number of times per day of medication administration) it has been shown that adherence declines. 42 Lopinavir/ritonavir is the only co-formulated ritonavir-boosted PI currently available and can be given either once- or twice-daily in treatment-naïve patients. Once-daily administration of LPV/r is not recommended in patients concurrently receiving efavirenz, nevirapine, nelfinavir or amprenavir because of the potential of decreased lopinavir concentrations. Other PI's that are available for once daily administration and are recommended as initial antiretroviral therapy choices include ritonavir-boosted atazanavir, darunavir, and fosamprenavir. Pill burden for the recommended PI's is least with ritonavir-boosted atazanavir (2 pills per day) and is most with both ritonavir-boosted fosamprenavir and LPV/r (4 pills per day). 4
Tolerability to ritonavir-boosted PI's in regards to adverse effects and metabolic abnormalities is a critical factor to consider when choosing an appropriate agent. Lopinavir/ritonavir given once-daily requires a 200 mg dose of ritonavir which increases the likelihood of gastrointestinal side effects as compared to the 100 mg twice daily administration. 26 Hyperlipidemia and specifically hypertriglyceridemia is more common with LPV/r as compared with other boosted PI's where as elevations in bilirubin are less likely with LPV/r.33–36 Metabolic complications such as dyslipidemia and insulin resistance may result in long-term consequences such as increased cardiovascular events. The metabolic abnormalities have been found to be dependent on the dose of the boosting agent ritonavir, so the decision on the agent and dosing regimen should be made with this factor in mind. 43
The cost effectiveness of the various boosted PI regimens is important in regards to patient preference as the cost for management of HIV is quite considerable. In one analysis conducted in 2007 using 2006 US wholesale acquisition costs, LPV/r was the most cost effective option when compared to other boosted PI's such as atazanavir, fosamprenavir and darunavir. 44 Other studies have looked at the clinical and related economic differences between LPV/r and ritonavir-boosted atazanavir and have found that the cost per month of each therapy to be $1250 and $1840, respectively and cost savings over 5- and 10-year periods to be $23,126 and $19,598, respectively.45,46
Place in Therapy
Reduction of HIV-related morbidity and mortality through the use of antiretroviral medications is the ultimate goal in the treatment of HIV. This is best accomplished through the use of potent combinations of agents that provide viral suppression and an immunologic response. The choice of the agents is based upon efficacy of antiviral response, tolerability of potential adverse effects, pharmacokinetic profile which promotes adherence and potential for development of cross-resistance. 4 Current US and International guidelines are consistent in their place in therapy of LPV/r.1,4
Both sets of guidelines recommend initiating one of the three following types of regimens for ART in the treatment-naïve patient: NNRTI + 2 NRTI, PI (ritonavir-boosted) + 2 NRTI, or integrase strand transfer inhibitor + 2 NRTI. The specific preferred PI-based regimens include ritonavir-boosted atazanavir and ritonavir-boosted darunavir. LPV/r is listed as an alternative regimen due to its need for 200 mg/day of ritonavir and subsequent higher rates of gastrointestinal side effects and hyperlipidemia when compared to boosted PI regimens only requiring 100 mg/day.1,4 In African countries, LPV/r is a second-line treatment option only. This review highlights some of the research associated with LPV/r and primarily the focus has been in simplification strategies in patients who are suppressed to below detectable limits on a combination triple-drug regimen to boosted protease inhibitor monotherapy. This strategy has demonstrated safety and efficacy in a high proportion of patients. This strategy is not to be used indiscriminately for all nucleoside reverse transcriptase inhibitors. However, to avoid some common toxicity of the nucleoside reverse transcriptase inhibitors, in virologically suppressed patients without previous failure to protease inhibitors, monotherapy with LPV/r may be an option if the viral load remains undetectable to reduce costs and preserve future treatment options.
Recommendations guiding the treatment of pediatric patients and pregnant women have been created based on study in both patient populations. The Working Group on Antiretroviral Therapy and Medical Management of HIV-Infected Children most recently published guidelines in February 2009. It is recommended that treatment-naïve children receive an antiretroviral combination containing 1 NNRTI + 2 NRTI or 1 PI (ritonavir-boosted) + 2 NRTI, with the preferred PI regimen being LPV/r. LPV/r is listed as the preferred regimen due to its durable virologic activity and low toxicity in both treatment-naïve and experienced children. In addition, dosing and efficacy data are available in infants under the age of 6 months. 2 Recommendations for the management of HIV infection vary in pregnant women depending on their history of receiving antiretroviral medications, the current level of viral suppression and other co-infections. LPV/r is listed as a recommended PI due to the lack of evidence of human teratogenicity, tolerability and short-term safety demonstrated in Phase I/II studies. 3
Conclusion
LPV/r is the only co-formulated ritonavir-boosted PI currently available. It is generally well tolerated and can be taken once- or twice-daily based on previous ART, patient characteristics and concomitant medications. LPV/r has been proven efficacious in children, adolescents and adults and is the preferred PI regimen in children and pregnant women and is an alternative PI regimen in adolescents and adults.
Disclosures
This manuscript has been read and approved by all authors. This paper is unique and not under consideration by any other publication and has not been published elsewhere. The authors and peer reviewers report no conflicts of interest. The authors confirm that they have permission to reproduce any copyrighted material.
Footnotes
Acknowledgement
The authors of this manuscript would like to thank Carol Peters for her excellent administrative assistance.
