Abstract
The serotonin-noradrenaline reuptake inhibitor duloxetine is a second-generation antidepressant for the treatment of major depressive disorder (MDD). Its inhibitory potency for serotonin (5-HT) and noradrenaline (NA) reuptake has been demonstrated in animal and in vitro studies. Human studies of peripheral markers of neurotransmission show inhibition of 5-HT reuptake by duloxetine and also support its potency in NA reuptake inhibition. Moreover, a positron emission tomography study in human brains shows that therapeutic doses of duloxetine result in >80% occupancy of 5-HT transporters. Duloxetine is metabolized by hepatic enzymes and cytochromes P450 CYP2D6 and CYP1A2, and plasma duloxetine concentrations increase linearly according to oral dose. In double-blind, randomized, placebo-control studies of MDD, duloxetine-treated patients show significantly increased response and remission rates and significantly longer time to relapse compared to placebo-treated patients. Moreover, duloxetine is efficacious for the treatment of generalized anxiety disorder (GAD) and pain-related diseases such as diabetic peripheral neuropathic pain (DPNP). A daily dose of 60 mg daily seems most effective for treating MDD, GAD and DPNP. Duloxetine is similar in efficacy to fluoxetine, paroxetine and escitalopram for MDD, although escitalopram is better for improving sleep. The most common treatment-emergent side effects of duloxetine are nausea, dry mouth, fatigue and decreased appetite. Studies suggest an association with the incidence of increased sweating or somnolence at higher doses and an association with irritability or anxiety at high plasma levels of duloxetine. Discontinuation rates due to adverse events are higher in patients who receive duloxetine versus placebo in short-term but not long-term studies. Interestingly, there is not a strong correlation between clinical efficacy and plasma levels of duloxetine. Therefore, although duloxetine is safe and tolerated at therapeutic doses of 60-120 mg/day, the dose should be adjusted while the patient is carefully monitored for efficacy and adverse events.
Keywords
Introduction
The serotonin-noradrenaline reuptake inhibitor (SNRI) duloxetine is a second-generation antidepressant. Second-generation antidepressants, including serotonin (5-HT) selective reuptake inhibitors (SSRIs) and SNRIs such as duloxetine, venlafaxine and milnacipran, are used widely for the treatment of major depressive disorder (MDD). Desvenlafaxine, sibtramine and tramadol are SNRIs that are used to treat depression, obesity and pain, respectively. Depression is also associated with anxiety and physiological or psychological pain. Duloxetine was developed and approved by the US Food and Drug Administration (FDA) in 2004 for the treatment of depression and diabetic peripheral neuropathic pain (DPNP). 1 In 2007, duloxetine was also approved by the FDA for the treatment of generalized anxiety disorder (GAD). 2
Pharmacology in Preclinical Studies
Duloxetine shows comparable affinities for human 5-HT transporters (Ki = 7.5 nM) and human NA transporters (Ki = 0.8 nM). Duloxetine has no or very low affinities to neurotransmitter receptors such as 5-HT1A, muscarinic, histamine H1, α1-adrenergic, α2-adrenergic and dopamine D2 receptors. 3 Duloxetine shows a well-balanced blocking property for 5-HT and NA reuptake as indicated by the selectivity ratio of 5-HT:NA reuptake inhibition. 4 The 5-HT:NA reuptake inhibition selectivity ratios of duloxetine, venlafaxine and milnacipran are 9.3, 30 and 1.6, respectively. 4 Studies using microdialysis probes implanted in rodent brains indicate that extracellular 5-HT and NA levels are elevated after duloxetine administration; this reflects the neurotransmitter concentrations in synaptic clefts and confirms that these agents act primarily to inhibit 5-HT and NA reuptake into presynaptic terminals.5,6 Duloxetine also blocks dopamine (DA) reuptake, as indicated by microdialysis studies,5,6 and also shows a marked affinity for the DA transporter.3,4 The Ki values for inhibition of human 5-HT transporter (in vitro binding) for duloxetine, venlafaxine and milnacipran are 0.8, 82 and 123 nM, respectively. 4
Muneoka et al 7 studied the effects of duloxetine (20 mg/kg/day) on monoamine levels in rats. Four-day treatment with duloxetine induced a marked increase in 5-HT while decreasing 5-HIAA levels in many brain regions; in contrast, there was no change in the DA level and only a small increase in NA in the nucleus accumbens (Table 1). Longer-term (21-day) treatment of rats with duloxetine increased the electrically stimulated release of 5-HT and NA in the hippocampus and midbrain, whereas only NA release was enhanced in the frontal cortex. 8 Finally, 28-day treatment of mice with duloxetine reduced 5-HT transporter density in the cortex but did not alter NA transporter density in the hippocampus. 9
Changes in 5-HT and NA contents, or 5-HT turnovers after short-term treatment with duloxetine or sertraline.
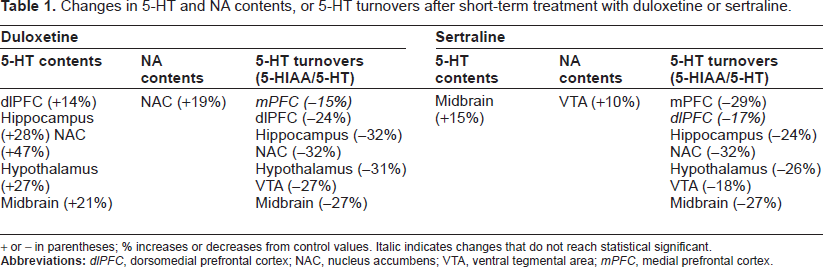
+ or - in parentheses; % increases or decreases from control values. Italic indicates changes that do not reach statistical significant.
Animal studies demonstrated the anxiolytic-like, antinociceptive and antidepressant-like effects of duloxetine; the latter was shown using the forced swimming test. 9 Decreased immobility time and increased climbing behavior accompanying duloxetine treatment implies serotonergic effects and noradrenergic or dopaminergic effects, respectively. 9 Chronic treatment with duloxetine (20 mg/kg/day) significantly increased the time animals spent in open areas in the zero maze, suggesting an anxiolytic action of the drug; acute treatment with duloxetine is associated with anxiogenic effects. 10 The antinociceptive effects of duloxetine have been evaluated in rodent experimental pain models.11–13 The results suggested that antidepressants that affect both 5-HT and NA have potent and efficacious antinociceptive effects, and that duloxetine is effective in the treatment of persistent or inflammatory pain in chronic pain models, but lacks analgesic effects in acute pain models.
Pharmacokinetics and Metabolism
The pharmacokinetics of duloxetine were evaluated in healthy men 14 as well as in a group of healthy men and women. 15 Steady state was reached after 3 days of treatment with doses of 20, 30 and 40 mg twice daily. Duloxetine was rapidly absorbed after oral administration, and plasma concentrations increased linearly. 14 The maximum plasma concentration (Tmax) was achieved in 6 h, 15 and the mean terminal elimination half-time was 12.5 h (range 9.2-19.1 h). 14 There was large intersubject variability in the pharmacokinetics. 14 Food and nighttime administration delayed Tmax, and nighttime administration tended to reduce the extent of absorption. 16 In a clinical study, plasma levels correlated with dosage 4 weeks after daily administration of duloxetine (30-120 mg/day), and plasma duloxetine levels ranged from 5 to 135 ng/ml (mean, 53.56 ng/ml). 17
Duloxetine undergoes extensive hepatic metabolism via cytochromes P450 CYP2D6 and CYP1A2 to form oxidative and conjugated metabolites.1,16 The major metabolites of duloxetine in plasma are 4-hydroxy duloxetine glucuronide and 5-hydroxy, 6-methoxy duloxetine sulfate. 18 Both metabolites are inactive and are excreted primarily in the urine: Of the 90.5% of radioactivity recovered from radiolabeled duloxetine, 72% is excreted in the urine and 18.5% in the feces. 19 The presence of mild renal dysfunction does not appear to have a marked effect on duloxetine clearance, 1 but patients with hepatic insufficiency have a decreased ability to metabolize and excrete duloxetine, resulting in elevated plasma levels. 20
Circulating duloxetine is 96% protein bound, primarily to albumin or α1-acidglycoprotein. Thus, elevated plasma levels of free duloxetine can occur in patients with low blood albumin levels. Moreover, free blood levels of warfarin and carbamazepine, which are also highly bound to albumin, may also be elevated in patients taking these drugs in combination with duloxetine.1,16,21
There are no clinically significant differences in the pharmacokinetics of duloxetine in healthy Japanese, Chinese and Caucasian subjects,22,23 and dosage adjustments are not required on the basis of gender.16,24
Duloxetine has differential effects on liver enzymes. It inhibits CYP2D6 isoenzyme to a moderate extent: Its inhibitory effect is intermediate between inhibition by sertraline and inhibition by fluoxetine or paroxetine. In contrast, duloxetine shows clinically insignificant inhibition of CYP1A2 and has no effect on CYP3A4, CYP2C9 and CYP2C19 isoenzymes.1,16,21 The drugs that interact with CYP2D6 drugs are mostly psychotropic agents, including the tricyclic antidepressants, phenothiazines and Type 1C antiarrhythmics, and pain medications like codeine and dextromethorphan. 1 Caution should be exercised when duloxetine is used in conjunction with paroxetine and fluoxetine, which are potent inhibitors of CYP2D6. Duloxetine should not be combined with other CYP1A2 inhibitors, like fluvoxamine and certain quinolone antibiotics. 1 There is a one third reduction in duloxetine exposure (i.e. in the area under the plasma concentration-time curve, AUC) in smokers versus nonsmokers due to the effect of smoking on CYP1A2. However, adjustment of duloxetine dose is not required on the basis of smoking status alone. 1
Physiological and Biochemical Effects in Healthy Subjects
The safety and tolerability of duloxetine (40-80 mg/day) were evaluated in a single-blind, placebo-controlled study in 12 healthy men. 14 In this study, somnolence, nausea, and dry mouth were experienced following initial administration, but these symptoms resolved with continuing drug administration. The same study showed that duloxetine was not associated with clinically significant changes in blood pressure or heart rate in the standing position, although small increases in systolic and diastolic blood pressure and small decreases in heart rate were observed. Supratherapeutic doses of duloxetine (120-400 mg/day) resulted in increased supine systolic and diastolic blood pressure and supine pulse rates 25 but did not affect ventricular repolarization as assessed by QTc (QT corrected for heart rate). 26 These data suggest that duloxetine is not associated with severe, clinically relevant adverse events, especially within the therapeutic dose range.
The effects of duloxetine on peripheral markers of the NA reuptake blockade (e.g. the pressor response to intravenous tyramine or urinary excretion of NA) and on peripheral markers of the 5-HT reuptake blockade (e.g. whole blood 5-HT concentration) were evaluated in healthy subjects.27,28 The results indicated that duloxetine is efficacious for blocking 5-HT reuptake at 20 mg/day, but is not efficacious for NA reuptake (as indicated by peripheral markers) even at 120 mg/day. However, further study showed that duloxetine blocked NA uptake; that study assessed the inhibitory efficacy of plasma on radioligand binding to the NA transporter, dihydroxyphenylglycol to NA (DHPG:NA) ratio in the plasma and urine, and the pressor response to tyramine. 29 Duloxetine occupancy of 5-HT transporters in the human brain was investigated using positron emission tomography (PET) with [11C]DASB. 30 The 5-HT transporter occupancy increased gradually with increased duloxetine dose and plasma duloxetine concentrations. Duloxetine at 60 mg/day seemed to be the optimal dose for maintaining high 5-HT transporter occupancy (>80%).
Efficacy in Major Depressive Disorder (MDD)
The main outcomes from randomized, double-blind studies for MDD are summarized in Table 2. Duloxetine (40-120 mg/day) was assessed in short-term studies with observation periods of 7 to 9 weeks. In a placebo-controlled study, duloxetine (60 mg/day) was superior to placebo in reducing the score on the 17-item Hamilton Rating Scale for Depression (HAM-D-17) 2 weeks after administration began. 31 The response rates after 8 to 9 weeks, defined as a 50% reduction in the HAM-D-17 score, were higher in patients receiving duloxetine versus placebo. The reported response rates to duloxetine (40-120 mg/day) were 65%, 32 64% 33 or 70%-77%. 34 Remission was defined as a score ≤7 points on the HAM-D-17. Reported remission rates were 43%-58% in the short-term studies (Table 2). Further analysis of two independent trials indicated significantly greater decreases in HAM-D-17 scores and Mainer scores in duloxetine-treated patients compared with placebo-treated patients, in the first and second weeks, respectively. 35 A post-hoc analysis of the data from four double-blind, placebo-controlled studies in which outpatients with MDD were randomized to duloxetine (60 mg/day) or placebo for 8 to 9 weeks indicated no significant improvements for sleep-related symptoms or genital symptoms. 36
Clinical outcomes from randomized, double-blind and placebo- or drug-control studies for the treatment of the major depressive disorder (MDD) with duloxetine.
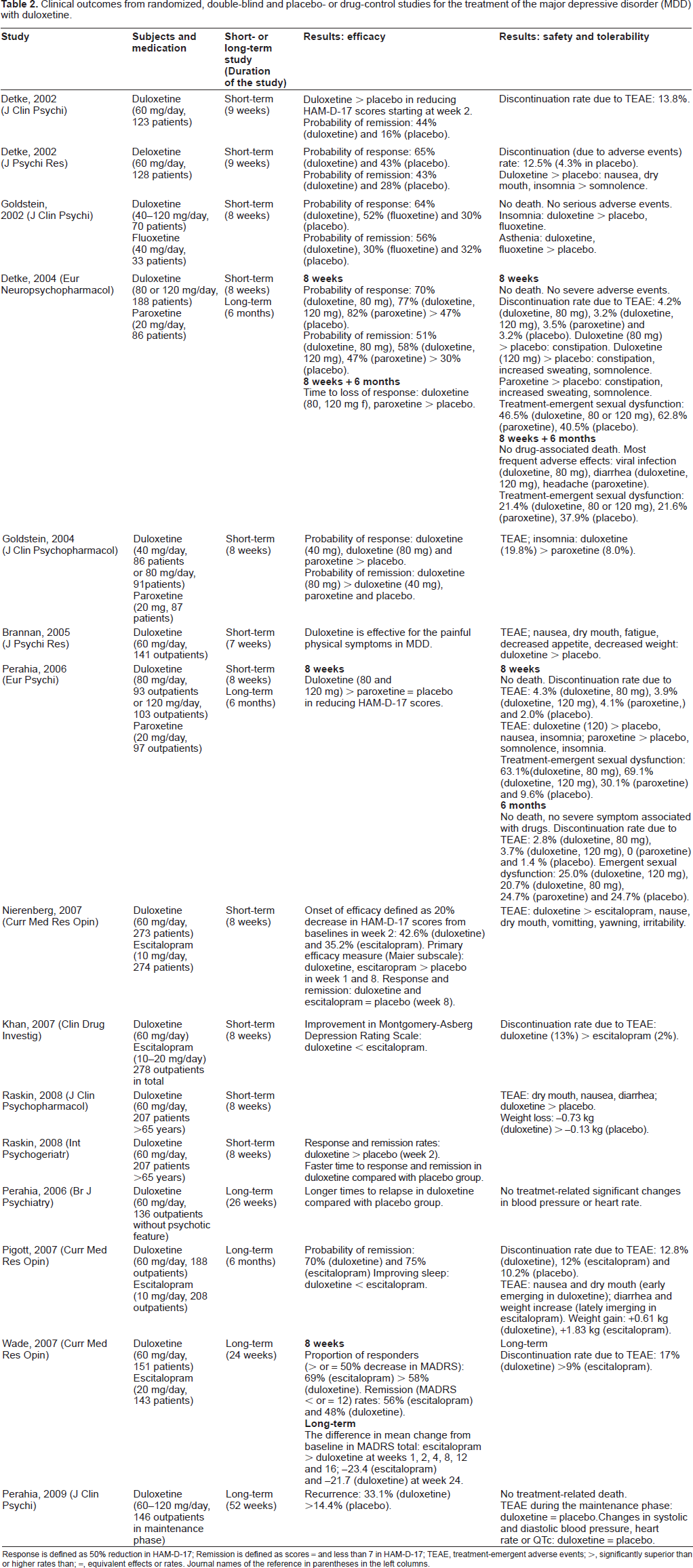
Response is defined as 50% reduction in HAM-D-17; Remission is defined as scores = and less than 7 in HAM-D-17; TEAE, treatment-emergent adverse events; >, significantly superior than or higher rates than; =, equivalent effects or rates. Journal names of the reference in parentheses in the left columns.
Six active drug-controlled studies have been conducted on duloxetine (Table 2). Overall, duloxetine was as effective as fluoxetine 33 and paroxetine34,37 in terms of response and remission rates for MDD. In one study, 80 or 120 mg/day of duloxetine showed significant efficacy at 8 weeks, whereas the efficacy of 20 mg/day paroxetine did not differ from placebo. 38 The onset of efficacy, defined as a 20% decrease in the HAM-D-17 score, was 42.6% two weeks after treatment with duloxetine (60 mg/day) versus 35.2% two weeks after treatment with escitalopram (10 mg/day). 39 In that study, the efficacy of duloxetine was similar to escitalopram in the first and eighth weeks compared with placebo. Another study comparing duloxetine (60 mg/day) and escitalopram (10-20 mg/day) over 8 weeks reported that escitalopram was superior in terms of Montgomery-Asberg Depression Rating Scale (MADS) scores. 40 A double-blind study showed that escitalopram improved the sleep of patients with MDD better than duloxetine. 41 Interestingly, the ability of duloxetine to improve episodic and working memory better than escitalopram was suggested in a study of patients with MDD who were antidepressantdrug naïve; although both drugs were effective in improving episodic memory, the working memory, mental processing speed and motor performance was more improved in patients given duloxetine. 42 Furthermore, a prospective 12-week open-label study showed that administration of reboxetine, a selective NA reuptake inhibitor, as an adjunct to duloxetine was effective in treating duloxetine-resistant depression. 43 Duloxetine appears to be efficacious for the treatment of depressive patients with atypical features, although generally monoamine oxidase inhibitors are reported to be more effective than SSRIs or SNRIs. 44 Taken together, evidence from these short-term studies suggests that duloxetine has efficacy equal to fluoxetine, paroxetine and escitalopram, whereas escitalopram more efficacious than duloxetine for sleep disorders.
The results of long-term studies of duloxetine for treating MDD are summarized in Table 2. Duloxetine-medicated patients had lower HAM-D-17 scores than did placebo group patients in a 26-week maintenance study. 45 The same study also showed that patients who received duloxetine (60 mg/day) had a significantly longer time to relapse than patients who received placebo. Patients who received duloxetine (60-120 mg/day) had a significantly lower recurrence rate versus a placebo group in a long-term study (52 weeks). 46 Another long-term study (6 months) indicated a significantly longer time to relapse in duloxetine- (60 or 120 mg/day) and paroxetine- (20 mg/day) treated patients compared with placebo-treated patients. 34 Remission rates were 70% and 75% in MDD patients who were treated with duloxetine (60 mg/day) and escitalopram (10 mg/day) for 6 months, respectively. 41 Another study comparing the long-term effects of duloxetine (60 mg/day) and escitalopram (20 mg/day) indicated that escitalopram was superior in terms of response but not remission rate in the acute phase. 47 Results of a placebo-controlled, randomized, double-blind study of patients with MDD treated for 8 weeks with duloxetine 60 mg/day (n = 273) or escitalopram 10 mg/day (n = 274), and for another 6 months with duloxetine up to 120 mg/day or escitalopram up to 20 mg/day, were subjected to post-hoc analysis. This analysis indicated that early changes in symptoms were treatment-specific, with early response in the core depression factor (Maier subscale), anxiety and motor activity for duloxetine, and in core depression factor and anxiety for escitalopram; further, a lack of early response as judged by depression symptom subscales was highly predictive of a lack of sustained remission. 48
A randomized double-blind, placebo-controlled study indicated that for patients with MDD that had not attained remission after 6 weeks, increasing duloxetine to 120 mg/day offered no advantage compared to those that continued taking 60 mg/day. 49 A dose-response relationship study using effect size analysis based on six double-blind, placebo-controlled clinical trials in patients with MDD indicated that duloxetine 60 mg daily is the most effective dose for treating MDD. 50 A post-hoc analysis of four double-blind, placebo-controlled studies indicated that duloxetine 60 mg daily produced significant improvement in symptoms compared with placebo regardless of baseline MDD severity. 51 Another study found a significant correlation between duloxetine dose and plasma levels in patients with MDD. 17 That study revealed the relationship between plasma concentrations of duloxetine and clinical improvement in anxiety symptoms, with the maximum effect observed at duloxetine plasma levels between 40 and 100 ng/ml and a modal dose of 60 mg/day.
An 8-week double-blind, placebo-controlled trial of duloxetine in elderly patients (65-90 years) with MDD indicated that duloxetine (60 mg/day) elicited significantly greater improvement in cognitive function, including learning and memory, than did placebo. 52 Duloxetine (60 mg/day) was also efficacious for the treatment of women of perimenopausal age with MDD. 53 Pooled data from doubleblind, placebo-controlled studies demonstrated that duloxetine induced significantly greater improvement than placebo in terms of depressive symptoms in MDD patients both with and without melancholic features. 54 Results from seven double-blind placebo-controlled clinical trials for MDD provided no evidence that duloxetine had differential efficacy or safety in Hispanic and Caucasian patients. 55
Efficacy in Generalized Anxiety Disorder (GAD)
The efficacy of duloxetine for the treatment of GAD has been demonstrated in placebo- and active drug-controlled studies (Table 3). Three short-term (10 week) placebo-controlled studies indicated the superior effects of duloxetine (60-120 mg/day) compared with placebo in reducing the Hamilton Anxiety Rating Scale (HAM-A) score.56–58 Treatment with duloxetine (60-120 mg/day) and venlafaxine extended-release (venlafaxine XR, 75-225 mg/day) demonstrated significantly greater improvement in the HAM-A psychic and somatic factor scores compared with placebo, whereas duloxetine treatment (20 mg/day) showed significantly greater improvement in the HAM-A psychic factor score but not the HAM-A somatic factor score compared with placebo. 57 A comparison of duloxetine and venlafaxine XR data from a 10-week, multicenter, randomized, placebo-controlled, double-blind study demonstrated that duloxetine (60-120 mg/day) was not inferior to venlafaxine XR (75-225 mg/day) for treating GAD. 59
Clinical outcomes from randomized, double-blind and placebo- or drug-control studies for the treatment of the generalized anxiety disorder (GAD) with duloxetine.
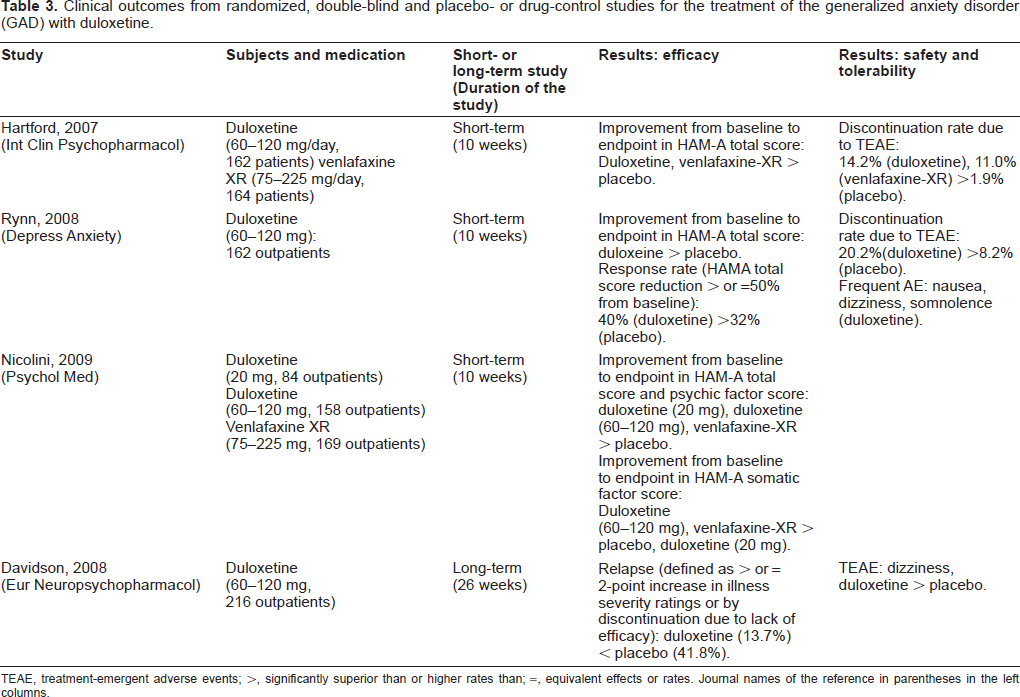
TEAE, treatment-emergent adverse events; >, significantly superior than or higher rates than; =, equivalent effects or rates. Journal names of the reference in parentheses in the left columns.
In a long-term study (26 weeks), a significantly lower frequency of relapse was observed in duloxetinetreated patients compared with placebo-treated patients: The relapse rates were 13.7% and 41.8% in duloxetine- and placebo-treated patients, respectively. 60 Multiple analyses of double-blind, placebo-controlled trials of duloxetine treatment (9-10 weeks) for GAD indicate that early improvement predicts endpoint response and remission; 61 further, patients treated with duloxetine, compared with placebo, experience a greater improvement in quality of life. 62 These data indicate that duloxetine (60-120 mg/day) is efficacious for treating GAD symptoms and for preventing relapse.
Efficacy in Pain-Related Diseases
Short-term double-blind placebo-controlled studies show that duloxetine has efficacy for the treatment of diabetic peripheral neuropathic pain (DPNP),63–66 chronic back pain 67 and pain associated with depression 68 (Table 4). Duloxetine (60-120 mg/day) is significantly more efficacious than placebo for treating DPNP,63–65 whereas duloxetine (20 mg/day) was not significantly better than placebo. 66 For chronic low back pain, duloxetine 60 mg daily showed significant efficacy in a placebo-controlled study. 67 Moreover, when duloxetine 60 mg daily was admini stered to patients with moderate pain associated with depression, patients had significantly reduced pain and improved depression symptoms after 8 weeks using the Brief Pain Inventory-Short Form (BPI-SF) and MADS; 68 the placebo did not have this effect. A post-hoc analysis of the data from a long-term study (52 weeks) showed that duloxetine (60 and 120 mg/day) was efficacious for treatment of DPNP in both younger (<65 years) and older (≥65 years) age groups. 65 Fibromyalgia is a chronic condition characterized by widespread pain, tender points and fatigue. A double-blind, placebo-controlled, 12-week trial indicated that duloxetine (120 mg/day) was an effective treatment for symptoms associated with fibromyalgia in patients with or without MDD. 69 In an indirect meta-analysis, duloxetine showed comparable efficacy to pregabalin and gabapentin for DPNP. 70 Post-hoc analysis of pooled data from two 9- and 10-week double-blind, placebo-controlled clinical trials indicated that duloxetine (60-120 mg/day) provided significantly greater pain reduction to patients with GAD. 71 These data indicate that duloxetine (60 mg/day) has analgesic effects for treating neuropathic pain and pain associated with psychological illness.
Clinical outcomes from randomized, double-blind and placebo- or drug-control studies for the treatment of pain related diseases with duloxetine.
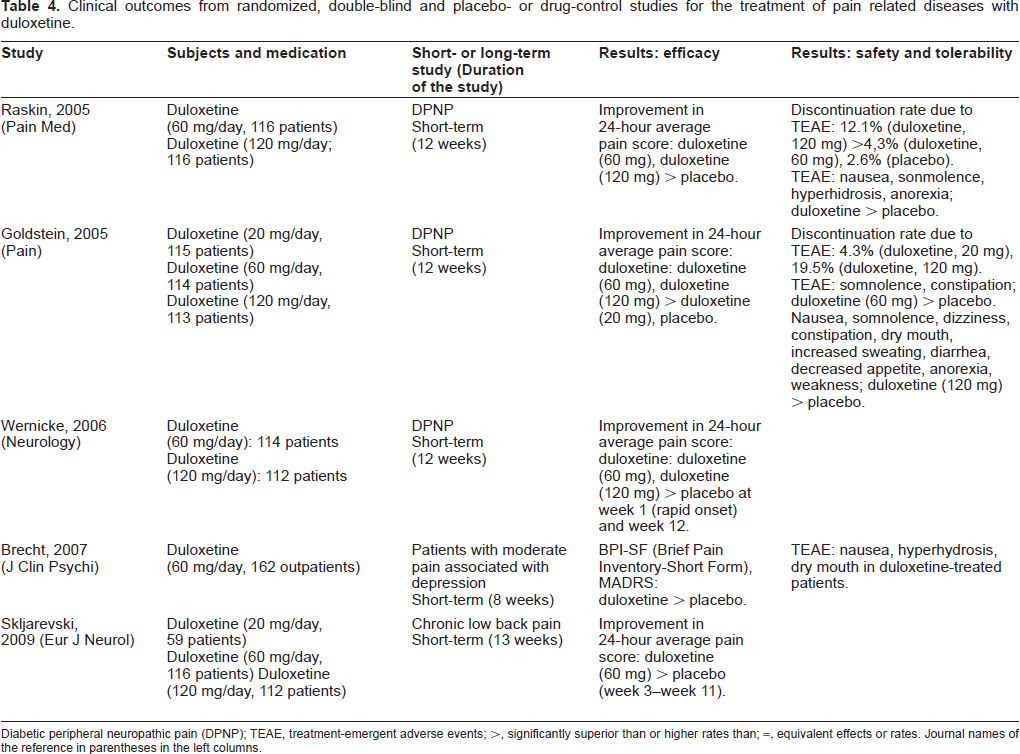
Diabetic peripheral neuropathic pain (DPNP); TEAE, treatment-emergent adverse events; >, significantly superior than or higher rates than; =, equivalent effects or rates. Journal names of the reference in parentheses in the left columns.
Efficacy for Treating Depression-related symptoms and other Mental Disorders
An 8-week open-label study indicated that duloxetine improved depression and vasomotor symptoms, as well as sleep, anxiety and pain in postmenopausal women. 72 Duloxetine (60-120 mg/day) may also be effective in treating dysthymia, although a double-blind, placebo-controlled study is needed. 73 In addition, case reports suggest that duloxetine is effective for treatment-resistant obsessive-compulsive disorder,74,75 for binging behavior in eating disorders 76 and for narcolepsy-catalepsy. 77
Safety and Tolerability in Patients (Tables 2–4)
In a short-term study of MDD, discontinuation rates due to adverse events in patients who received duloxetine were higher (12.5%) than in patients taking placebo (4.3%). 32 However, subsequent short-term studies showed no significant differences in discontinuation rates due to adverse events between duloxetine and placebo.34,38 The reported discontinuation rates due to adverse events in patients in a short-term study (8 weeks) were 4.3% (duloxetine-treated, 80 mg/day), 3.9%, (duloxetine-treated, 120 mg/day), 4.1% (paroxetinetreated, 20 mg/day) and 2.0% (placebo-treated). 38 There was a significantly higher discontinuation rate due to adverse events in duloxetine-treated patients compared with escitalopram-treated patients in another 8-week study. 40
A long-term study (52 weeks) found no significant difference between duloxetine (60-120 mg/day) and placebo 46 in terms of treatment-emergent adverse events during the maintenance phase of outpatients with MDD. In a long-term study (6 months), discontinuation rates due to adverse events were 2.8%, 3.7%, 0% and 1.4% in duloxetine-treated (80 mg/day), duloxetine-treated (120 mg/day), paroxetine (20 mg/day)-treated and placebo-treated patients, respectively. 38 A long-term study (6 months) by Pigott et al also indicated that there was no significant difference in discontinuation rates due to adverse events between duloxetine (12.8%), escitalopran (12%) and placebo (10.2%), 41 whereas a different long-term study (24 weeks) reported significantly lower discontinuation rates in escitalopram-treated (20 mg/day) patients compared with duloxetine-treated patients (60 mg/day). 47
Frequent treatment-emergent adverse events reported for short-term duloxetine medication were nausea, dry mouth, fatigue and decreased appetite. 78 In a shortterm trial, insomnia was detected more frequently in duloxetine-treated patients than in paroxetine-treated ones. 37 The most frequent treatment-emergent adverse events with duloxetine compared with escitalopram were nausea, dry mouth, vomiting, yawning and irritability. 39 The most frequent adverse effects reported in a 6-month study were viral infection, diarrhea and headache in treatment groups taking duloxetine (80 mg/day), duloxetine (120 mg/day) or paroxetine (20 mg/day). 34 In patients with MDD, the incidence of irritability and anxiety as treatment-emergent adverse effects was associated with higher duloxetine plasma levels.
A long-term study indicated that adverse events such as nausea or dry mouth tended to emerge early in treatment with duloxetine, whereas those associated with escitalopram, such as diarrhea and weight gain, tended to emerge later in treatment. 41 In a short-term study of elderly patients given duloxetine (60 mg/day), dry mouth, nausea, diarrhea and weight loss were reported as emergent adverse events. 79 In short-term studies33,34,38 and a long-term studies,34,45 there were no deaths related to duloxetine treatment. In short-term studies,33,34,38 there were no severe adverse events associated with duloxetine.
Weekly escalation of duloxetine dosages was examined in a non-blind, non-placebo-controlled study. 80 In weekly escalation from 60 mg to 90 mg to 120 mg duloxetine, the majority of adverse events occurred in the first week of duloxetine dosing (at 60 mg once daily); the data indicated that rapid dose escalation of duloxetine is safe and tolerable. Duloxetine was judged to be safe in overdose, based on four cases of acute overdose, with a maximum of 1400 mg, that resulted in no fatalities. 1 A meta-analysis of 9 short-term trials in which treatment with duloxetine was stopped abruptly investigated symptoms following abrupt duloxetine discontinuation. 81 Of duloxetine- and placebo-treated patients, 44.3% and 22.9%, respectively, experienced discontinuation-emergent adverse events (DEAEs). The most frequent DEAEs in duloxetine-treated patients compared with placebo-treated patients were dizziness, nausea, headache, paresthesia, vomiting, irritability and nightmares.
The frequency rates of treatment-emergent sexual dysfunction symptoms in a short-term trial (8 weeks) of duloxetin and paroxetine were 46.5% (duloxetine), 62.8% (paroxetine) and 40.5% (placebo); however, long-term (8 months), the frequency rates did not differ significantly between the three groups. 34 The incidence of treatment-emergent sexual dysfunction in MDD patients treated with escitalopram was higher compared with duloxetine or placebo with short-term treatment, but after 12 weeks, there were no statistically significant differences between the drugs. 82 A meta-analysis conducted on 4 double-blind, placebo-controlled studies showed that the incidence of acute treatment-emergent sexual dysfunction was significantly lower among duloxetine-treated male and female patients compared with those receiving paroxetine, although both rates were significantly higher than placebo. 83 In men, this outcome was manifested by the inability to achieve an orgasm and poor satisfaction from orgasm. 83
Sleep disturbance (insomnia and somnolence) has been reported as a treatment-emergent adverse effect in duloxetine-treated patients with MDD,32–34,37,38 GAD 58 and DPNP.63,64 The incidence of insomnia is higher than the incidence of somnolence in duloxetine-treated patients with MDD, 32 although higher doses of duloxetine might be associated with somnolence. 34 Furthermore, insomnia rates are higher in duloxetine-treated patients with MDD compared with fluoxetine-treated 33 or paroxetine-treated patients.34,37 Interestingly, somnolence seems more prevalent than insomnia in non-MDD patients58,63,64 or healthy volunteers. 14 In patients with MDD, treatment with duloxetine may be associated with a higher rate of insomnia compared to tricyclic antidepressants and SSRIs such as fluoxetine, paroxetine and escitalopram. 84
No significant changes in systolic and diastolic blood pressure, heart rate or QTc were reported during a long-term placebo-controlled trial of duloxetine (60-120 mg/day).45,46 In addition, meta-analyses demonstrated no clinically significant changes in electrocardiograms, although slight increases in heart rates and systolic (but not diastolic) blood pressure were observed in duloxetine-treated patients.85,86 However, it should be noted that there was a single case report of duloxetine-induced tachycardia; the tachycardia was alleviated with propranolol without discontinuing duloxetine. 87 Moreover, there were two case reports showing that duloxetine and another SNRI, venlafaxine, induced tachycardia in patients with chronic heart failure and left ventricular systolic dysfunction. 88 These clinical data suggest that caution should be exercised in using SNRIs with the potential to increase levels of NA, particularly for patients with heart failure or tachycardia.
A review of the hepatic effects of duloxetine noted transient transaminase elevation in about 1% of patients. 89 The use of duloxetine may exacerbate abnormal liver functions in patients with a history of affected liver functions, including those with hepatitis C and severe alcoholism. 1
A double-blind study revealed that food affected starting dose tolerance. 90 The study recommended that duloxetine (30 mg or 60 mg) should be taken with food for 1 week to improve initial tolerance. On the other hand, a post-hoc analysis of seven double-blind, placebo-controlled trails revealed that duloxetinetreated women had a significantly higher rate of nausea as a treatment-emergent adverse event than duloxetinetreated men; there was also more nausea reported by women given placebo compared with males. 91
Case studies have suggested a close temporal relationship between initiation of duloxetine treatment and the emergence of suicidal ideation in adult patients. 92 However, a meta-analysis of 12 placebo-controlled trials showed no evidence of an increased risk of suicidal behaviors or ideation during treatment with duloxetine compared with placebo. 93 Another analysis of the FDA summary basis of approval reports suggested caution in generalizing suicide risk. 94
In retrospective analysis of data from eight placebo-controlled, double-blind, randomized clinical trials of duloxetine in patients with non-bipolar major depression, a 0.2% incidence of hypomania was observed. This suggests that duloxetine is associated with a low incidence of treatment-emergent mania. 95
In GAD patients, the discontinuation rate due to adverse events was higher for duloxetine (60-120 mg/day) compared with placebo56,58 but did not differ from the discontinuation rate in patients treated with venlafaxine XR 56 (Table 3). Nausea, dizziness and somnolence were more frequent in duloxetine-treated patients than in placebo-treated patients. 58 In the use of duloxetine to treat DPNP (Table 4), the rate of discontinuation due to adverse events was higher in duloxetine (120 mg/day)-treated patients compared with duloxetine (60 mg/day) or placebo-treated patients. 64 The 12-week study showed that nausea, somnolence, hyperhidrosis and anorexia were treatment-emergent adverse effects observed more frequently in duloxetine-treated patients compared with placebo-treated patients. Duloxetine (120 mg/day) appeared to induce a larger number of adverse events compared with duloxetine (60 mg/day). 63 We conclude that duloxetine 60 mg daily is a well tolerated dosage for the treatment of MDD, GAD and DPNP.
Place in Therapy
Duloxetine can be used for the treatment of depression, including MDD with and without melancholic feature. In addition, this drug might be effective for dysthymia, depression with atypical features or postmenopausal symptoms. Duloxetine can also be used to treat anxiety disorders such as GAD or obsessive-compulsive disorder. Finally, duloxetine can be used for the treatment of pain-related diseases such as DPNP and fibromyalgia.
Relationship between Clinical Effects and Mechanism of Action
As described above, the primary action associated with the antidepressant effects of SNRIs is inhibition of reuptake of 5-HT and NA; this inhibition induces subsequent changes in monoaminergic metabolism in regions of the brain 7 and facilitates neurotransmission via autoreceptor desensitization. 96 Moreover, it should be noted that other neuronal systems, neurotransmitters and brain-derived molecules are probably involved in the pathology and therapeutic actions of SNRIs, including the mesolimbic dopaminergic system, glutamatergic system, the GABAergic system, neuropeptides, cytokines, neurosteroids and neurotrophic factors related to neurogenetic effects (reviewed by Shirayama and Chaki). 97
We hypothesize that blocking 5-HT reuptake primarily affects a pathologic state that is associated with anxiety, and that the improvement in depressive symptoms, which include many heterogeneous components, is the result of changes at the neural network level. The delayed response in MDD might be due to the time needed to unlearn or forget the learned “depressive” habits or adverse conditioning (extinction). 98 Indeed, it has been suggested that maladaptions to stress and impaired extinction of acquired fear is a basal pathology in anxiety and mood disorders; the prefrontal cortex and amygdala may be involved in this processes.99–101
Conclusions
Duloxetine is a potent well-balanced inhibitor of both 5-HT and NA reuptake. Its potency has been demonstrated in humans by brain imaging studies or on the basis of changes in peripheral markers. Duloxetine is efficacious and a safe and well-tolerated drug for the treatment of depressive disorders, anxiety disorders and pain-related diseases. Although 60 mg daily of duloxetine seems to be most effective for the treatment of these diseases, the dose should be fine-tuned to optimize its efficacy and prevent adverse events. The optimal therapeutic dose of duloxetine results in occupation of 5-HT reuptake sites in the human brain. Therefore, the primary effects of duloxetine appear to be the blockade of 5-HT reuptake, which may be associated with relief of anxiety. Other neurotransmitters and neural networks may be involved in the downstream neuronal processes related to overall recovery from the pathologic conditions.
Disclosure
The author reports no conflicts of interest.
