Abstract
As vascular endothelial growth factor (VEGF) plays a central role in tumor growth, invasion and metastasis, inhibiting tumor angiogenesis by blocking the actions of VEGF is a rational therapeutic strategy. Drugs targeting the VEGF system are currently in development and at the most advanced stage of development is bevacizumab. The effect of bevacizumab on breast cancer has been examined in many clinical trials, and promising results have been reported. The clinical effect of bevacizumab monotherapy for breast cancer is not clear; however, the ECOG-E2100 study showed that first-line anti-angiogenic therapy using bevacizumab combined with paclitaxel clearly improved the response for earlier stage metastatic breast cancer (MBC). As a stronger anti-tumor effect is expected when prescribing bevacizumab for patients at an early stage of MBC, many first-line clinical trials using bevacizumab with other combination regimens are currently ongoing. Although the common side effects of bevacizumab are hypertension, proteinuria, wound-healing complications, and thromboembolism, it is a comparatively safe agent. It is expected that the many ongoing clinical trials will establish bevacizumab as a standard first-line therapy for MBC.
Introduction
Angiogenesis is essential for tumor growth or metastasis, and one of the most important primary factor controlling vessel formation is hypoxia. Hypoxia triggers the secretion of pro-angiogenic factors, mainly vascular endothelial growth factor (VEGF), and stimulates new vessel formation to supply oxygen to the tumor. Thus, VEGF plays a key role in tumor angiogenesis.1–4 The members of the VEGF family are part of the VEGF-platelet-derived growth factor (PDGF) family. VEGF is a ligand for Flt-1 (VEGF receptor-1) and KDR or Flt-2 (VEGF receptor-2: VEGFR-2), and is a 34-46 KDa homodimeric glycoprotein. The VEGF family members include VEGF (VEGF-A), VEGF-B, VEGF-C, VEGF-D, VEGF-E, and placental growth factor (PIGF). VEGF-C and VEGF-D act on lymphatic endothelial cells, and are involved in lymphangiogenesis. 5 The expression of the VEGF gene is regulated by various factors including hypoxia, pH, growth factors, cellular formation, cytokines, inflammation, hormones, and oncogenes. For example, under hypoxic conditions, the transcription factors hypoxia-inducible factor-1α (HIF-1α) and HIF-2α is stabilized in cancer cells, and is transported to the nucleus where they interact with HIF-β. This complex binds to a specific sequence on the VEGF gene called the hypoxia response element (HRE), which stimulates VEGF gene transcription and VEGF protein is produced, then VEGF is secreted from the cancer cells.6,7 After the binding of VEGF to the VEGFR-2, which is expressed on the endothelial cell membrane, the homodimerization of the VEGFR-2 occurs. The effects of VEGF are then mediated by the activation of various intracellular signaling transduction pathways, such as the activation of receptor tyrosine kinase activity or the induction of autophosphorylation of the VEGFR-2. This receptor mediates the biological action via the phospholipase C (PLC)–-protein kinase C (PKC)–-mitogen-activated protein kinase (MAPK) pathway. Namely, PLC directly binds to the autophosphorylated to VEGFR-2, then tyrosine-phosphorylated and is activated. The activated PLC stimulates the PKC activation and activated Raf-1 to MAPK cascade.6,8 VEGF plays major biological roles in the growth and proliferation of vascular endothelial cells, the survival of immature endothelial cells through the prevention of apoptosis, 9 the increase of vascular permeability, 10 the anti-apoptotic effect on tumor cells, 11 the increased likelihood of metastasis,12,13 and the suppression of the anti-tumor immune response.14–16 Mechanism of VEGF signaling is illustrated in Figure 1. Many studies have shown the overexpression of VEGF in tumors or the circulation in various human malignancies, including breast cancer.17–19 VEGF expression was shown to increase as tumors progressed towards greater malignancy or to correlate with the extent of tumor vascularization,18,20–22 and a significant association was also reported between plasma VEGF levels and disease stage or metastasis, 23 which suggested that the expression of VEGF is associated with poor prognosis for various human tumors. Indeed, the expression of VEGF has been reported to correlate with poor prognosis for breast cancer.24–28
Preclinical Studies
VEGF plays a central role in tumor growth, invasion, and metastasis; therefore, inhibiting tumor angiogenesis by blocking the actions of VEGF is a rational therapeutic strategy. In several preclinical studies, a monoclonal antibody specific for VEGF completely suppressed neovascularization and inhibited the growth of tumors arising from human tumor cell lines injected into nude mice.29,30 These results strongly suggest that inhibiting VEGF blocks angiogenesis and tumor growth. The mechanism of the anti-tumor effect achieved through targeting VEGF might involve promoting apoptosis of the tumor vasculature, inhibiting metastasis, and promoting anti-tumor immune responses. Specifically, VEGF prevents the apoptosis of vascular endothelial cells in the immature blood vessels that are characteristic of tumors by inducing the expression of the anti-apoptotic proteins B-cell CLL/lymphoma 2 (Bcl-2) in human endothelial cells. 9 Thus, VEGF secreted by a tumor allows the endothelial cells in the immature tumor blood vessels to survive, and in this way maintains the tumor blood supply. The inhibition of VEGF might remove the protection from apoptosis that it confers to tumor blood vessel-associated endothelial cells, leading to disruption of the tumor blood supply. As angiogenesis and lymphangiogenesis are involved in the progression of tumors,31–33 VEGF is important for tumor metastasis. Animal tumor xenograft models treated with anti-VEGF antibody have demonstrated that anti-VEGF therapy can prevent tumor metastasis; hence, it has been suggested that targeting VEGF might inhibit metastasis.12,13 Furthermore, VEGF inhibits the functional maturation of dendritic cells, and these effects might encourage tumor growth by allowing the tumor to evade the host immune response.14–16 Thus, inhibiting VEGF might promote the anti-tumor immune response. Since inhibiting VEGF promotes the anti-tumor effect by such mechanisms, drugs such as bevacizumab (Avastin™), vatalanib, semaxinib, and ZD6474 which target the VEGF system are currently under development. The most promising approaches are offered by monoclonal antibodies directed against VEGF. The drug in this class that is currently at the most advanced stage of development is bevacizumab, which consists of 93% human and 7% murine components. Bevacizumab is highly specific to VEGF (VEGF-A), and by inhibiting the ligand, this antibody prevents angiogenesis by inhibiting VEGF signaling. In the xenograft model, it was reported that bevacizumab potently reduced the growth rate of several malignant tumors.34,35 These preclinical results suggest that bevacizumab might be effective for malignant tumors. The clinical efficacy of bevacizumab for breast cancer is described below.
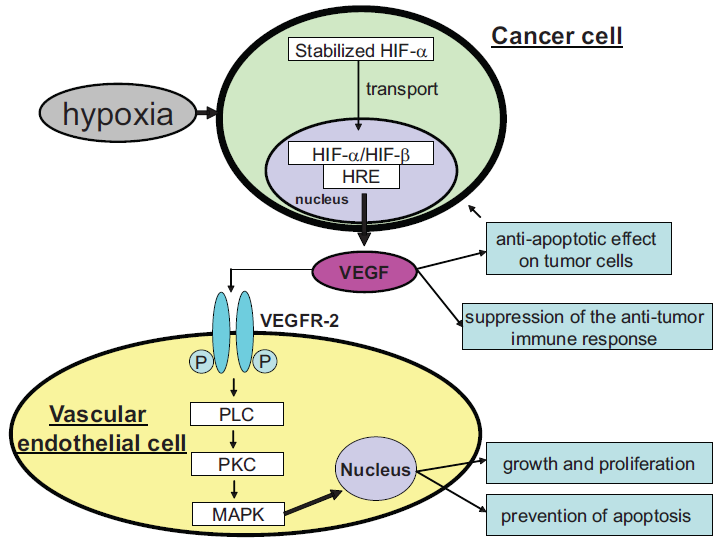
Mechanism of VEGF signaling. Under hypoxic conditions, the hypoxia-inducible factor-1α (HIF-1α), HIF-2α is stabilized, and is transported to the nucleus where they interact with HIF-β in the cancer cells. This complex then binds to a hypoxia response element (HRE), which stimulates VEGF gene transcription. After the binding of VEGF to the VEGFR-2, the homodimerization occurs. The effects of VEGF is then mediated by the activation of various intracellular signaling transduction pathways, such as the activation of receptor tyrosine kinase activity or the induction of autophosphorylation of the VEGFR-2, and this receptor mediated the biological action via the PLC-PKC-MAPK pathway.
Clinical Efficacy of Bevacizumab for Breast Cancer
Anti-angiogenic therapy using bevacizumab combined with standard chemotherapy was effective for colorectal cancer and non-small cell lung cancer (NSCLC).36,37 Bevacizumab has already been approved by the United States Food and Drug Administration (FDA) and it is currently in use for the treatment of colorectal cancer and NSCLC.38,39 The effect of bevacizumab on breast cancer has been examined in many clinical trials, and promising results have been reported.
Bevacizumab monotherapy
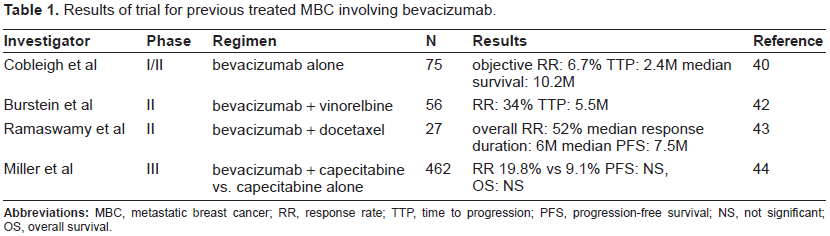
In a phase I/II trial, 75 patients with previously treated metastatic breast cancer (MBC) were treated with escalating doses of bevacizumab ranging from 3 to 20 mg/kg. The objective response rate (RR) was 6.7%. This trial established a recommended dosage of 10 mg/kg based on tolerability and active criteria. 40 However, the clinical effect of bevacizumab monotherapy for breast cancer is not clear, and the effect is expected to be enhanced by using a combination of conventional chemotherapy and bevacizumab. High levels of VEGF result in tumor vasculature that is excessively permeable and leaky, leading to high interstitial pressure within the tumor. The tumor vessel morphology also promotes unevenness in the blood flow and the delivery of oxygen and nutrients. This phenomenon can worsen the performance of conventional chemotherapy, because the delivery of chemotherapeutic and other agents to the tumor can be uneven, with some areas being inaccessible to the tumor cell killing activity. VEGF inhibition should be particularly effective in reducing vascular permeability, decreasing tumor interstitial pressure, and inducing tumor vasculature regression. An anti-VEGF agent could suppress angiogenesis and prevent tumor growth and metastasis, while improved delivery of the chemotherapy could enhance the tumor cell killing activity. These features indicate that anti-VEGF therapy would be an ideal partner for chemotherapy.36,41
Combination therapy of bevacizumab and conventional chemotherapy
In a phase II trial, 55 patients with previously treated MBC were administered bevacizumab plus vinorelbine. The RR was 34% and the time to progression (TTP) was 5.5 months. In addition, lower levels of baseline VEGF were associated with a longer TTP. 42 In another phase II trial, 27 patients with MBC were treated with bevacizumab plus weekly docetaxel as a first-line or second-line chemotherapy. The overall RR was 52% and the median progression-free survival (PFS) was 7.5 months. 43 Furthermore, in the first phase III trial, bevacizumab in combination with capecitabine was randomly assigned to 462 patients with MBC who had been previously treated with anthracycline or taxane-containing regimens. The combination therapy significantly increased the RR (19.8 vs. 9.1%; p = 0.001); however, the addition of bevacizumab did not improve the PFS or the overall survival (OS). 44 Results of trial for previous treated MBC involving bevacizumab are summarized in Table 1. The trial clearly demonstrated that bevacizumab had anti-tumor activity, although it did not translate into an improvement in either the PFS or the OS for this condition. This could be related to the diminishing influence of VEGF in the later stages of MBC. As the tumor develops further, additional factors are secreted, including basic fibroblast growth factor (FGF) and transforming growth factor (TGF-β). Later in development, PIGF, platelet-derived endothelial cell growth factor (PD-ECGF), and pleiotrophin can further stimulate angiogenesis.45,46 Thus; the inhibition of VEGF in the later stages of breast cancer might be less effective because of the increased expression of other proangiogenic factors that support tumor growth. These findings support the targeting of VEGF in the early stages of the disease and, hence, its use in first-line therapy for the treatment of MBC.25,47
Results of trial for previous treated MBC involving bevacizumab.
First-line treatment with bevacizumab in combination with chemotherapy for MBC
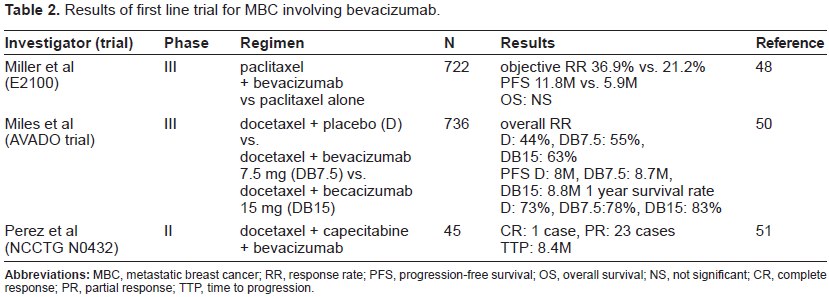
Based on this hypothesis, the randomized phase III Eastern Cooperative Oncology Group 2100 (ECOG-E2100) trial compared paclitaxel with or without bevacizumab in 722 patients with human epidermal growth factor receptor 2 (HER2)-negative MBC as first-line therapy. Combination therapy significantly increased the objective RR (36.9% vs. 21.2%; p < 0.001) and the PFS (11.8 vs 5.9 months; p < 0.001); however, the addition of bevacizumab did not improve the OS (26.7 vs 25.2 months; p = 0.16). 48 The ECOG-E2100 study showed that, although there was no difference in the OS, first-line anti-angiogenic therapy using bevacizumab clearly improved the response for earlier stage MBC. Based on this trial, on February 22, 2008, the FDA granted accelerated approval for bevacizumab to be used in combination with paclitaxel for the treatment of patients who have not received chemotherapy for metastatic HER2 negative breast cancer. 49 This approval was given on the basis of PFS data rather than OS data, which historically has been used as the standard clinical endpoint for evaluating first-line therapy. The results of the Avastin and Docetaxel (AVADO) trial, which compared docetaxel with or without bevacizumab (7.5 mg/kg or 15 mg/kg) in 736 patients with HER2-negative MBC as first-line therapy, were reported at the American Society of Clinical Oncology 44th Annual Meeting. 50 Combination therapy significantly increased the PFS (docetaxel plus placebo [D] = 8.0 months; docetaxel plus bevacizumab 7.5 mg/kg [DB7.5] = 8.7 months; and docetaxel plus bevacizumab 15 mg/kg [DB15] = 8.8 months) and the overall RR (D = 44%; DB7.5 = 55%; and DB15 = 63%). The 1-year survival rates were 73% for D, 78% for DB7.5, and 83% for DB15. The grade 3/4 adverse event rates were 67% for D, 75% for DB7.5, and 84% for DB15. The AVADO trial results demonstrated that first-line combination therapy of docetaxel with bevacizumab significantly improved the PFS and the overall RR compared with docetaxel alone for MBC, and the combination therapy was well tolerated. The North Central Cancer Treatment Group (NCCTG) N0432 phase II trial investigated combined docetaxel, capecitabine, and bevacizumab in 45 patients with MBC as first-line therapy. In total, 24 patients achieved a confirmed tumor response (a complete response [CR] in one patient, and a partial response [PR] in 23 patients), and the median TTP was 8.4 months. The reported adverse events were neutropenia (77%), hand-foot skin reaction (27%), general fatigue (20%), and grade 3 hypertension in one patient, although no grade 3/4 proteinuria or bleeding was documented. After two cycles of treatment, more than 50% of the patients required a dose reduction of capecitabine and docetaxel. Although the effect of the docetaxel, capecitabine, and bevacizumab combination therapy was equivalent to that of combination therapy with paclitaxel and bevacizumab in a phase III trial, concurrent treatment with docetaxel, capecitabine, and bevacizumab had comparatively strong side effects, and further studies of this regimen are warranted. 51 Results of first line trial for MBC involving bevacizumab are summarized in Table 2. As these trials clearly demonstrated that first-line treatment with bevacizumab had stronger anti-tumor activity, many first-line clinical trials using bevacizumab are currently ongoing. The MO19391 Avastin Therapy for Advanced Breast Cancer (ATHENA) trial is a first-line study investigating the safety profile of bevacizumab for MBC in combination with taxane-based chemotherapy. This study plans to enroll 2,300 patients, and the primary endpoint is safety. 52 RiBBON 1 (AVF3694g) is a phase III study for establishing standard first-line chemotherapy regimens containing bevacizumab for MBC. This study is comparing paclitaxel with or without bevacizumab in 722 patients with HER2-negative MBC as first-line therapy. Chemotherapy (an anthracycline-based regimen, taxane, or capecitabine) plus bevacizumab is being compared with chemotherapy plus placebo. The estimated enrollment is 1,200 patients, and the primary endpoint is PFS. 53 A first-line clinical trial using bevacizumab for triple-negative breast cancer is also ongoing. Triple-negative breast cancer does not react to hormone therapy or trastuzumab, and the prognosis is poor. 54 As chemotherapy is the only current treatment, combination therapy of chemotherapy and bevacizumab is expected to be effective for triple-negative breast cancer. A phase II trial of bevacizumab in combination with doxorubicin and carboplatin is being conducted for triple-negative MBC as first-line therapy. The estimated enrollment is 50 patients, and the primary endpoint is PFS. 55 A phase II trial of bevacizumab in combination with paclitaxel and carboplatin is also being conducted for triple-negative MBC. The estimated enrollment is 46, and the primary endpoint is overall RR. 56 If a stronger anti-tumor effect is shown by these clinical trials, it will suggest that bevacizumab can become an effective treatment strategy for a triple-negative breast cancer.
Results of first line trial for MBC involving bevacizumab.
First-line treatment of bevacizumab in combination with other molecular targeting therapy for MBC
A first-line clinical trial using bevacizumab combined with other molecular targeting therapy is also ongoing. Targeting HER2 signaling has led to the development of novel therapies such as the monoclonal antibody trastuzumab.57,58 HER2 plays a specific role in the regulation of VEGF expression. A preclinical study showed a strong association between HER2 and VEGF.59,60 Animal models showed superior efficacy when bevacizumab was combined with trastuzumab. 61 Based on these results, a phase I study of bevacizumab combined with trastuzumab was carried out. Among the nine patients treated with bevacizumab plus trastuzumab, five showed a confirmed tumor response (a CR in one patient, and a PR in four patients). The reported adverse events were grade 1/2 diarrhea, general fatigue, nausea, allergic reaction, hypertension, and proteinuria, although the combination therapy was well tolerated. Based on this trial, the recommended phase II dose was set as a 10 mg/kg bevacizumab plus trastuzumab 4 mg/kg loading dose, and then 2 mg/kg. 62 A phase II trial of combined therapy targeting HER2-amplified MBC or local relapse, and surgically unresectable breast cancer, was carried out using trastuzumab and bevacizumab as first-line treatment. A clinical response was documented in 13 of 28 (46%) patients, all of whom showed a PR. Grade 3/4 dyspnea, left ventricular dysfunction, and proteinuria were each reported in one patient, respectively, and hypertension was reported in 5 patients. 63 This data supported the clinical use of combination therapy with bevacizumab and trastuzumab, and suggested it as an effective firstline therapy for HER2-overexpressing MBC. Two ongoing phase III studies are verifying the efficacy of combination therapy with bevacizumab and trastuzumab. One is the ECOG-E1105 trial, which is studying weekly paclitaxel with or without carboplatin plus trastuzumab with or without bevacizumab. The estimated enrollment is 489 patients, and the primary endpoint is PFS. 64 The other is the AVEREL trial, which is studying docetaxel plus trastuzumab with or without bevacizumab. The estimated enrollment is 489 patients, and the primary endpoint is PFS. 65 Furthermore, a preclinical study showed that the epidermal growth factor receptor (EGFR) is associated with angiogenesis, 66 and also appears to regulate VEGF. 67 Xenograft models showed improved efficacy when anti-EGFR therapy was combined with anti-VEGF therapy, 68 and combination therapies with anti-EGFR agents and bevacizumab are expected to have a stronger anti-tumor effect. Based on these results, a phase II study that combined bevacizumab with erlotinib, which is a small-molecule EGFR tyrosine kinase inhibitor, was carried out in 38 patients with MBC who had been previously treated with one or two chemotherapy regimens. One patient achieved a PR for 52 months, and 15 patients showed stable disease (SD) for 9 weeks. The median TTP was 11 weeks. The reported adverse events were diarrhea (84%) and skin rash (76%), although they were mostly grade 1 or 2. Hypertension was seen in 18% of the patients and was grade 3 in 11%. 69 Sunitinib (SU011248), which is an oral small-molecular tyrosine kinase inhibitor that inhibits the VEGFR, exhibits potent antiangiogenic and antitumor activity. A phase II trial combining bevacizumab plus paclitaxel and sunitinib as first-line therapy for MBC is currently ongoing. 70
First-line treatment of bevacizumab in combination with hormone therapy for MBC
Cross-talk has also been demonstrated between the VEGF pathway and the hormone receptor pathway. The expression of VEGF was significantly correlated with the estrogen receptor status, 71 and the anti-estrogen tamoxifen was reported to inhibit the secretion of VEGF. 72 These preclinical results suggest that combination therapy with an aromatase inhibitor and bevacizumab might be more effective than either agent alone. The result of a phase II trial of combined bevacizumab and letrozole as first-line therapy for hormone-receptor-positive MBC is reported. Of the 25 patients evaluated, 2 have achieved a PR and 17 have showed stable disease (SD). Although some patients have grade 3 hypertension, combination therapy of letrozole and bevacizumab appears to be well tolerated. 73 A first-line clinical trial using bevacizumab for hormone-receptor-positive MBC is also ongoing. The AVF3544s trail is studying anastrozole or fulvestrant with bevacizumab as first-line therapy for postmenopausal MBC. Trastuzumab is being added for HER2-positive patients. The estimated enrollment is 80 patients, and the primary endpoint is PFS. 74 The Cancer and Leukemia Group B (CALGB)-40503 phase III trial is studying tamoxifen or letrozole together with bevacizumab compared with tamoxifen or letrozole alone in treating stage III or stage IV breast cancer. The estimated enrollment is 502 patients, and the primary endpoint is PFS. 75 The Spanish Breast Cancer Research Group is performing a phase III trial comparing letrozole with or without bevacizumab as first-line therapy for postmenopausal patients with advanced breast cancer or MBC. The estimated enrollment is 378 patients, and the primary endpoint is PFS. 76
Safety
Based on the data from numerous clinical trials, bevacizumab appears to be generally well tolerated, although it sometimes has specific adverse events. The common side effects are hypertension, proteinuria, wound-healing complications, and thromboembolism.7,77,78
Hypertension
The mechanism by which hypertension is caused by bevacizumab is not clear. It has been speculated that the inhibition of VEGF by bevacizumab induces less nitrous oxide for vasodilatation and so vasoconstriction might occur. Vasoconstriction can cause a significant increase in blood pressure resulting from changes in peripheral vascular resistance.78–81 Effect of VEGF inhibition on the control of blood pressure by the kidney might also be involved after bevacizumab treatment. 82 Clinically, hypertension caused by bevacizumab is usually readily controlled with standard therapies such as angiotensin-converting enzyme (ACE) inhibitors or β-blockers. 78 Patients should be monitored for the development or worsening of hypertension by frequent blood-pressure measurements during bevacizumab treatment. If the hypertension does not respond to antihypertensive therapy, the use of bevacizumab should be temporarily suspended. In the event of grade 4 hypertension or hypertensive crisis, bevacizumab should be permanently discontinued. 78
Proteinuria
The underlying mechanism is also poorly understood. Proteinuria might be related to the effects of VEGF on the renal glomerular capillaries. Treatment with bevacizumab resulted in a reduction in glomerular endothelial cell proliferation, as well as an increase in capillary damage. 78 The ECOG-E2100 trial reported that grade 3 or 4 proteinuria was more frequent in patients receiving bevacizumab and paclitaxel (3.6% vs. 0%, p < 0.001). 48 Patients should be monitored for proteinuria by frequent dipstick urinalysis during bevacizumab treatment. In cases where the proteinuria gets worse, bevacizumab should be carefully prescribed. George et al reported on a case of nephrotic syndrome after bevacizumab treatment, 83 and its use should be permanently discontinued in such patients.
Wound-healing complications
The mechanism is thought to involve an effect of VEGF on dermal-wound angiogenesis. 78 Recently, neoadjuvant therapy using bevacizumab for breast cancer has been tested, and bevacizumab should be administered for 30-60 days (based on its half-life: 13-21 days)84,85 prior to a surgical procedure.47,78
Thromboembolism
Bevacizumab induces the apoptosis of endothelial cells, and the resulting exposure of subendothelial cells initiates a coagulation cascade. Endothelial dysfunction and defects in the interior vascular lining can then result in the exposure of subendothelial collagen, increasing the frequency of thrombotic events.78,86 Patients should be monitored for venous and arterial thromboembolism. Bevacizumab should be carefully prescribed for those patients who have high risk factors for thromboembolism. Patients developing venous thromboembolism can continue with bevacizumab while being treated with full-dose anticoagulants, although its use should be immediately discontinued in those who develop arterial thromboembolism. 47
Conclusions and Future Directions
It is expected that the many ongoing clinical trials will establish bevacizumab as a standard first-line therapy for MBC. In order to control micro-metastasis by introducing antiangiogenic therapy at an early stage, clinical trials are currently evaluating the effect of bevacizumab in an adjuvant setting. The BETH study and the ECOG-E5103 study are phase III trials of adjuvant treatment for lymph-node-positive or high-risk, lymph-node-negative breast cancer. Their estimated enrollments are 3,500 and 4,950 patients, respectively, and their primary endpoints are disease-free survival (DFS).87,88 Both are mega-trials and their results are expected to determine the polarity of adjuvant therapy using bevacizumab for early breast cancer. The BEATRICE study is a phase III trial of adjuvant treatment for triple-negative breast cancer. The estimated enrollment is 2,530 patients, and the primary endpoint is DFS. 89 The results are expected to reveal whether bevacizumab is effective adjuvant therapy for triple-negative breast cancer. Furthermore, an ongoing clinical trial in a neoadjuvant setting is also studying the use of bevacizumab at an earlier stage. The National Surgical Adjuvant Breast and Bowel Project (NSABP)-B-40 is a randomized phase III trial of neoadjuvant therapy in patients with palpable and operable breast cancer. The estimated enrollment is 1,200 patients, and the primary endpoint is a pathologic CR (pCR) of the primary tumor in the breast. 90 The German Breast Group is also running a phase III trials program exploring the integration of bevacizumab, everolimus (RAD001), and lapatinib into current neoadjuvant chemotherapy regimes for primary breast cancer. The estimated enrollment is 2,547 patients, and the primary endpoint is pathological CR. 91 The results are expected to reveal the role of bevacizumab in neoadjuvant therapy for early breast cancer. Although the common side effects of bevacizumab are hypertension, proteinuria, wound-healing complications, and thromboembolism, it is a comparatively safe agent. However, its long-term safety will need to be verified for use as an anti-angiogenic treatment. Moreover, although there are currently no biomarkers to predict the effect of bevacizumab, it is likely that biomarkers of response will be identified to determine which subtypes of breast cancer patients will specifically benefit from bevacizumab therapy. Recently, Schneider et al reported the association of VEGF genotype with outcome in E2100 trial of paclitaxel compared with paclitaxel plus bevacizumab. 92 This research concluded that the data supported an association between VEGF genotype and median OS as well as grade 3/4 hypertension when using bevacizumab in MBC. On the other hand, Ramaswamy et al also reported that E-selectin, an endothelial cell-specific membrane glycoprotein implicated in angiogenesis, was statistically, significantly associated with the response to the combination therapy using bevacizumab. 43 These reports should contribute to the establishment of predictive markers for bebacizumab treatment.
In conclusion, anti-angiogenic therapy combined with the novel treatments hold great promise as the next generation of anticancer therapeutics in breast cancer.
Disclosure
The authors report no conflicts of interest.
