Abstract
Depression is the predominant mood state in patients with bipolar I or II disorder over the course of illness. In spite of this, relatively few pharmacological treatments have been shown to be effective for treating depressive episodes associated with bipolar disorder in adults. Combination therapy with olanzapine and fluoxetine (OFC) is approved in the US for the treatment of acute depressive episodes in adults with bipolar I disorder. The short-term efficacy and safety of OFC for the treatment of bipolar depression are supported by results of four randomized, acute-phase studies. OFC has been associated with significantly greater depressive symptom improvement than placebo, and with higher rates of treatment response and remission than placebo and olanzapine monotherapy. OFC has also been shown to improve depressive symptoms to a greater degree than modestly dosed lamotrigine, with similar rates of positive treatment response and remission. Although OFC was generally well tolerated in each of the reviewed studies, clinically significant weight gain, adverse changes in glycemic and lipid profile, and prolactin elevation may complicate both short- and long-term treatment. OFC was not associated with significantly increased risk of treatment-emergent mania in any of the reviewed studies. The broader effectiveness of OFC for the treatment of bipolar depression across clinically relevant subtypes (eg, patients with bipolar II disorder and comorbid substance abuse) and over long-term follow-up are needed. Comparative effectiveness studies of OFC and other available agents are also needed in order to determine its place among other available options for treating acute bipolar depressive episodes.
Introduction
Longitudinal studies of patients with bipolar disorder have challenged the notion of a purely episodic illness characterized primarily by mania or hypomania.1,2 Mood symptoms are more chronic and persisting than previously thought, and acute depressive episodes and persisting depressive symptoms during non-acute phases of the illness are predominant.3,4 Over the lifetime course of bipolar I or II disorder, depressive episodes and persisting depressive symptoms account for greater disability and adverse functional impact than manic or hypomanic mood episodes.3–5
In spite of this, relatively few pharmacological options have demonstrated effectiveness for the treatment of acute bipolar depressive episodes. Traditional mood stabilizers, eg, lithium and valproic acid, have substantially greater effectiveness for treating acute mania compared with acute bipolar depression.6,7 The effectiveness of lamotrigine has been better established for bipolar maintenance8,9 than acute bipolar depression, although more severely depressed patients may derive greater benefit. 10 Antidepressants (with mood stabilizer or atypical antipsychotic co-treatment) have been shown to be effective for some patients;11–13 however, their use in bipolar depression is controversial due to risk of cycle acceleration and mood switching. 14 The risk of manic induction or cycle acceleration may be higher with tricyclic antidepressants and venlafaxine compared with SSRIs,15,16 although rates of hypomanic/manic induction associated with antidepressants may also be attenuated if they are combined with mood stabilizers. 17 There is also controversy regarding the efficacy of adjunctive antidepressants for treating bipolar depression. The large-scale Systematic Treatment Enhancement Program for Bipolar Disorder (STEP-BD) study showed no additional benefit from sertraline or bupropion over placebo when added to mood stabilizers. 18
Clinical trials for the treatment of bipolar depression are far outnumbered by those for acute manic or mixed episodes. 19 Available pharmacological options for acute mania also far outnumber those available for acute bipolar depression–-nine drugs have regulatory approval in the US for treatment of acute mania, while only two are approved for the treatment of bipolar depression. The first pharmacotherapeutic agent approved for the treatment of acute depressive episodes in adult patients with bipolar I disorder was the olanzapine-fluoxetine combination (OFC), marketed by Eli Lilly and Co. under the brand name, Symbyax®. In this paper, we provide an overview of the clinically relevant pharmacological properties of OFC, and review the published acute-phase clinical studies of OFC for the treatment of bipolar depression.
Literature search
We searched MEDLINE (1990–2010) to identify published randomized, double-blind, trials, written in English, that compared OFC with an active comparator and/or placebo for the treatment of bipolar depression in adults. The following key words were used as an initial step: (olanzapine OR fluoxetine OR olanzapine fluoxetine OR olanzapine fluoxetine combination) AND (bipolar OR bipolar depression OR bipolar disorder). We then applied limits on article type (randomized controlled trial), language (English), and age (all adult: 19+ years). Abstracts of article citations were then reviewed to select articles that met selection criteria.
Mechanism of Action
Olanzapine
OFC is a combination of the atypical antipsychotic drug, olanzapine, and the selective serotonin reuptake inhibitor (SSRI) antidepressant, fluoxetine. 20 Like most atypical antipsychotic drugs, olanzapine combines relatively potent dopamine D2 receptor antagonism with even more potent serotonin 5-HT2A blockade.21,22 This receptor binding profile is believed to confer at least some of olanzapine's atypical antipsychotic properties, including a lower propensity than typical neuroleptics (eg, haloperidol, etc.) for causing side effects related to basal ganglia dysfunction (dystonia, parkinsonism, akathisia, etc.), prolactin elevation, and tardive dyskinesia at clinically relevant doses. Olanzapine displays high affinity binding at several other neuroreceptors, including serotonin 5-HT2C, histamine H1, α1 and α2 adrenergic, and muscarinic M1–5 receptors.23,24 Olanzapine acts as a pharmacological antagonist at these sites, which may confer many of the drug's adverse effects. 23
Fluoxetine
Fluoxetine, like other SSRIs, blocks serotonin re-uptake pumps (5-HTT), thus increasing synaptic availability of the neurotransmitter and heightening serotonergic neurotransmission. 25 This activity is believed to confer the antidepressive effects of fluoxetine, other SSRIs, and other serotonin-potentiating antidepressants. 26 Fluoxetine also has low binding activity at norepinephrine and dopamine re-uptake pumps, although this is not believed to be potent enough to contribute to the drug's therapeutic effects. 27 Fluoxetine is not believed to interact with any specific monoamine neuroreceptors, or interact with other neurotransmitter systems that regulate central monoamine neurotransmitter activity.
The olanzapine/fluoxetine combination (OFC)
The neurobiological basis of bipolar depression is unknown. However, like unipolar depression, dysfunction in monoamine neurotransmission has been implicated as one important neuropathophysiological factor in bipolar depression. 28 In preclinical models, the addition of olanzapine to fluoxetine has been shown to result in greater serotonergic activity than with fluoxetine alone, and to reverse fluoxetine-induced inhibition of noradrenergic activity. 29 In other pre-clinical studies, addition of olanzapine to fluoxetine has been shown to reverse fluoxetine-associated suppression of locus coeruleus firing, and enhance noreadrenergic activity in prefrontal cortex.30,31 In addition, olanzapine has been shown in animal models to enhance cortical release of dopamine. 31 The enhanced catecholaminergic activity in these studies has been shown to occur in brain regions critical for higher-order cognitive functioning, emotional regulation, motivation/drive and attention. 32 Dysfunction in each of these domains is characteristic of patients with bipolar and unipolar depression. Thus, the combination of olanzapine with fluoxetine may result in greater catecholaminergic neurotransmission than would be observed with the use of fluoxetine alone. While the neurobiology of mood switching is poorly understood, 33 coadministration of olanzapine, other atypical antipsychotics and mood stabilizers with antidepressants may limit the potential for antidepressant-induced switch in bipolar mood polarity or increases in mood cycle frequency.17,34
Pharmacokinetics
Both of OFC's constituent agents are rapidly absorbed and extensively distributed following oral ingestion. 20 Both undergo extensive hepatic metabolism. Olanzapine is converted into several metabolites via interaction with cytochrome P450 (CYP450) 1A2 and 2D6 isoenzymes. 35 Olanzapine's elimination half-life ranges from 21 to 54 hours, with a mean of 30 hours. While olanzapine's metabolic by-products are not thought to contribute significantly to its pharmacological activities, the primary metabolite of fluoxetine, norfluoxetine, has the same pharmacodynamic profile as its parent compound (eg, potent inhibition of 5-HTT), and a much longer mean elimination half-life (fluoxetine, 1–4 days vs. norfluoxetine, 7–15 days). 36 Although studies are lacking, the comparatively long elimination half-lives of fluoxetine and norfluoxetine may be advantageous for patients with bipolar disorder who have difficulty adhering to pharmacotherapeutic regimens. Whether or not clearance times relate to risk of drug-drug interactions or mood switching/cycle acceleration in patients with bipolar disorder is unknown. There are no known clinically significant pharmacokinetic effects of combining olanzapine with fluoxetine. 20
Clinical Efficacy of OFC in Acute Bipolar Depression
There are four published reports from acute-phase or short-term randomized studies of OFC in patients with bipolar depression.37–40 The design features of these studies are presented in Table 1. All four studies examined the short-term effectiveness of OFC over 7–8 weeks of treatment, and involved randomized comparisons of OFC and one or more comparator groups. Three studies were randomized, double-blind studies that employed incomplete factorial (OFC vs. olanzapine monotherapy vs. placebo), 37 factorial (OFC vs. olanzapine monotherapy vs. fluoxetine monotherapy vs. placebo), 38 or traditional parallel-group (OFC vs. lamotrigine) designs. 39 The fourth study was conducted in two periods. 40 The first period (Period 1) consisted of open-label treatment with OFC over 7 weeks. Patients who achieved positive treatment response during Period 1 were then randomized to either 12 weeks of continued OFC therapy or to olanzapine monotherapy on an open-label basis. Thus, the short-term, acute-phase effectiveness of OFC for bipolar depression in this study was examined prior to randomization. For this study, we will summarize results from both Periods 1 and 2. Information about this study was identified in the published report 40 and a separate study summary available online. 41
Design features of four acute-phase or short-term randomized studies of the olanzapine-fluoxetine combination (OFC) in patients with bipolar depression.

The study by Tohen et al (2003) did not include a fluoxetine (FLX) monotherapy arm, and is therefore referred to in this review as having an incomplete factorial design;
The Tamayo et al (2009) study was conducted in two phases, beginning with open-label OFC treatment for 7 weeks (Period 1). Subjects who achieved satisfactory response during Phase I (CGI-BP score ≤ 3 and ≥ 50% improvement [reduction] in MADRS total score from baseline at week 7) were eligible for randomization to continued OFC or OLZ monotherapy for 12 weeks (Period 2);
Additional study data was accessed on 7/16/2010 at: http//:www.clinicalstudyresults.org/documents/company-study_3066)0.pdf;
In the study by Amsterdam and Shults (2005), a total of 5 (15%) patients continued their pre-study mood stabilizers after randomization (lithium, n = 4; valproic acid, n = 1). The distribution of these 5 patients in the four study arms was not specified;
A total of 41 subjects were enrolled in the Amsterdam and Shults (2005) study. Of these, 5 were screen failures. Two additional patients withdrew consent after their baseline study visits, to yield a total of 34 randomized subjects with follow-up data;
Represents dropouts from randomization to study end point for all studies–-with the exception of Tamayo et al (2009), for which dropouts for both Periods 1 and 2 are presented.
The 28-item and 17-item “atypical” symptom profile versions of the HAM-D scale were utilized in the study by Amsterdam and Shults (2005);
Defined as a ≥50% improvement (reduction) in MADRS total score from baseline. In the Tohen et al (2003) study, completion of at least 4 weeks of follow-up was also required. In the Brown et al (2006) study, response was also defined as achieving a CGI-S score ≤ 3;
Defined as a MADRS total score of ≤ 12 at study end point. In the Tohen et al (2003) study, completion of at least 4 weeks of follow-up was also required. In the Brown et al (2006) study, a MADRS total score of ≤ was also considered as a more conservative alternative definition of remission;
Defined as satisfying DSM-IV criteria for manic or hypomanic episodes. YMRS scores of ≥8 and ≥12 were also considered;
Defined as a ≥ 50% improvement (reduction) in MADRS total score from baseline and a CGI-BP score < 3 at study endpoint.
Randomized, double-blind studies
The key efficacy results, including rates of categorical positive treatment response and remission, from each of the reviewed studies are provided in Table 2. The first rigorous study of OFC in bipolar depression was an 8-week, phase III, multi-site study of 833 patients with bipolar I disorder (BP-I), depressed phase. 37 Eligible subjects were required to have a Montgomery Asberg Depression Rating Scale (MADRS) 42 score of ≥20 at study entry, and were randomized to 8 weeks of double blind treatment with OFC (mean modal dose = 7.4/39.3 mg/day), olanzapine monotherapy (OLZ mono; mean modal dose = 9.7 mg/day) or placebo (PLC). In the OFC group, olanzapine and fluoxetine were taken together, but were administered as separate capsules. All study drugs were flexibly dosed. Depressive symptoms were assessed using the MADRS. Positive treatment response was defined as a reduction in MADRS total score of ≥50% from baseline, while remission was defined as achieving a MADRS total score of ≤12 at study endpoint. Additional criteria for positive treatment response and remission required completion of at least 4 weeks of the study. Additional efficacy endpoints included the Clinical Global Impressions Bipolar Version-Severity of Depression scale (CGI-BP-S), 43 the Young Mania Rating Scale (YMRS) 44 and the Hamilton Anxiety Rating scale (HAM-A). 45 OFC was associated with significantly greater improvement over time in MADRS total score than OLZ mono or PLC (Table 2). Greater improvement in MADRS scores with OFC compared with placebo and OLZ mono occurred as early as weeks 1 and 4, respectively, and continued throughout the remainder of the study. Therapeutic effect sizes were greater for OFC than OLZ mono. Similar results were obtained for change in CGI-BP-S scores, with minor variation. OLZ monotherapy did not significantly improve core depressive features, while OFC significantly improved both depressive core features and neurovegetative disturbance. Improvement in HAM-A scores was significantly greater for OFC- and OLZ mono-treated subjects compared with placebo; however, differences between OFC and OLZ mono were not statistically significant. Positive treatment response and remission rates were significantly greater for OFC than either OLZ mono or placebo. Median time to positive treatment response was significantly shorter in the OFC group than both the OLZ mono (21 vs. 55 days; log-rank test χ 2 = 7.9, df = 1, p = 0.005) and PLC groups (21 vs. 59 days; log-rank test χ 2 = 23.8, df = 1, P < 0.001). Median time to remission was also significantly shorter with OFC than either OLZ mono (42 vs. 57 days; log-rank test χ 2 = 6.0, df = 1, P = 0.01) or PLC (42 vs. 59 days; log-rank test χ 2 = 18.0, df = 1, P < 0.001).
Selected efficacy results from four acute-phase or short-term randomized studies of the olanzapine-fluoxetine combination (OFC) in patients with bipolar depression.
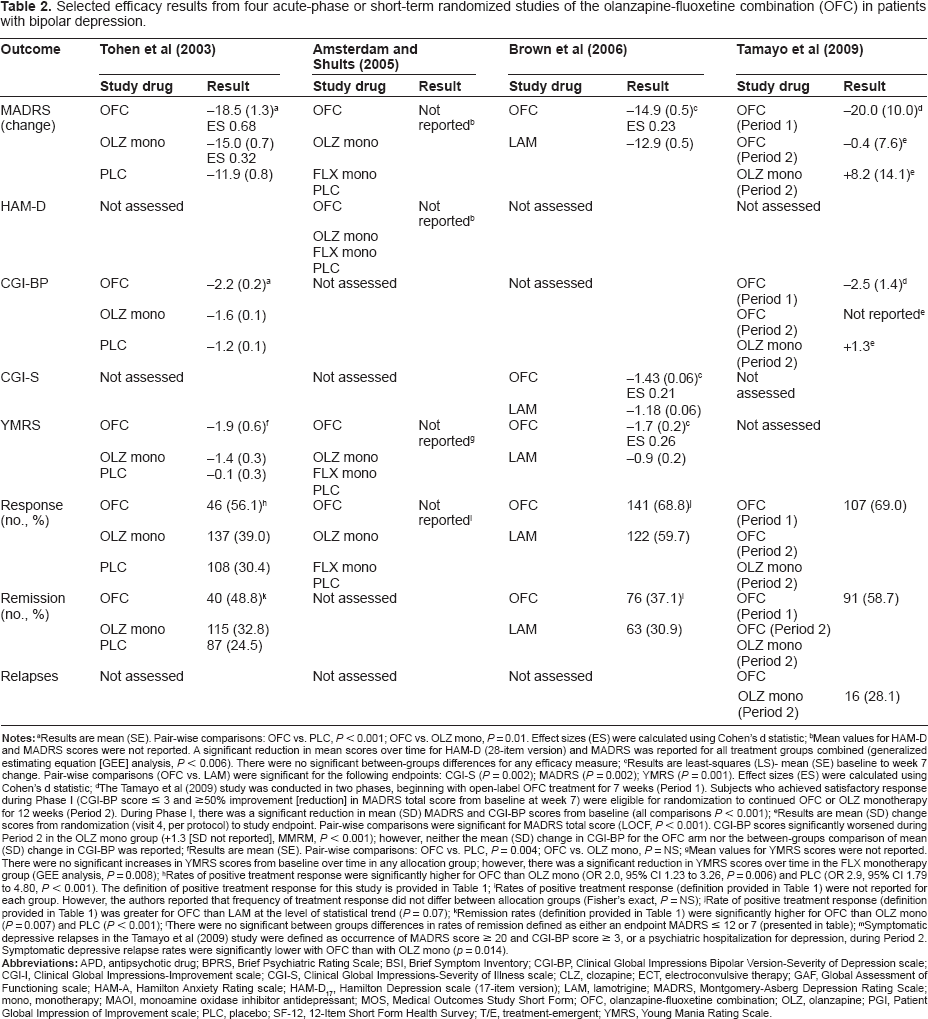
Results are mean (SE). Pair-wise comparisons: OFC vs. PLC, P < 0.001; OFC vs. OLZ mono, P = 0.01. Effect sizes (ES) were calculated using Cohen's d statistic;
Mean values for HAM-D and MADRS scores were not reported. A significant reduction in mean scores over time for HAM-D (28-item version) and MADRS was reported for all treatment groups combined (generalized estimating equation [GEE] analysis, P < 0.006). There were no significant between-groups differences for any efficacy measure;
Results are least-squares (LS)- mean (SE) baseline to week 7 change. Pair-wise comparisons (OFC vs. LAM) were significant for the following endpoints: CGI-S (P = 0.002); MADRS (P = 0.002); YMRS (P = 0.001). Effect sizes (ES) were calculated using Cohen's d statistic;
The Tamayo et al (2009) study was conducted in two phases, beginning with open-label OFC treatment for 7 weeks (Period 1). Subjects who achieved satisfactory response during Phase I (CGI-BP score ≤ 3 and ≥50% improvement [reduction] in MADRS total score from baseline at week 7) were eligible for randomization to continued OFC or OLZ monotherapy for 12 weeks (Period 2). During Phase I, there was a significant reduction in mean (SD) MADRS and CGI-BP scores from baseline (all comparisons P < 0.001);
Results are mean (SD) change scores from randomization (visit 4, per protocol) to study endpoint. Pair-wise comparisons were significant for MADRS total score (LOCF, P < 0.001). CGI-BP scores significantly worsened during Period 2 in the OLZ mono group (+1.3 [SD not reported], MMRM, P < 0.001); however, neither the mean (SD) change in CGI-BP for the OFC arm nor the between-groups comparison of mean (SD) change in CGI-BP was reported;
Results are mean (SE). Pair-wise comparisons: OFC vs. PLC, P = 0.004; OFC vs. OLZ mono, P = NS;
Mean values for YMRS scores were not reported. There were no significant increases in YMRS scores from baseline overtime in any allocation group; however, there was a significant reduction in YMRS scores over time in the FLX monotherapy group (GEE analysis, P = 0.008);
Rates of positive treatment response were significantly higher for OFC than OLZ mono (OR 2.0, 95% CI 1.23 to 3.26, P = 0.006) and PLC (OR 2.9, 95% CI 1.79 to 4.80, P < 0.001). The definition of positive treatment response for this study is provided in Table 1;
Rates of positive treatment response (definition provided in Table 1) were not reported for each group. However, the authors reported that frequency of treatment response did not differ between allocation groups (Fisher's exact, P = NS);
Rate of positive treatment response (definition provided in Table 1) was greater for OFC than LAM at the level of statistical trend (P = 0.07);
Remission rates (definition provided in Table 1) were significantly higher for OFC than OLZ mono (P = 0.007) and PLC (P < 0.001);
There were no significant between groups differences in rates of remission defined as either an endpoint MADRS ≤ 12 or 7 (presented in table);
Symptomatic depressive relapses in the Tamayo et al (2009) study were defined as occurrence of MADRS score ≥ 20 and CGI-BP score ≥ 3, or a psychiatric hospitalization for depression, during Period 2. Symptomatic depressive relapse rates were significantly lower with OFC than with OLZ mono (p = 0.014).
Results of a 24-week, open-label extension of this acute-phase study were published, 46 and will be reviewed briefly here. A total of 376 patients who completed the acute-phase study by Tohen et al (2003) 37 were enrolled. All subjects received flexibly-dosed olanzapine monotherapy (5-20 mg/day) for one week. Patients were then allowed to choose between OLZ mono or OFC (6/25, 6/50, or 12/50 mg/day) during the remaining follow-up visits. Thus, at study endpoint, three treatment groups were identified: those who remained on OLZ mono (mean modal dose, 8.3 mg/day), those who took OFC (mean modal dose, 8.1/41.9 mg/day), and those who switched treatment between OLZ mono and OFC during follow-up. Patients were also classified into two groups based on treatment response at the end of the acute-phase study: those who had achieved remission, and those who had not. For those who had achieved remission, there were no significant changes MADRS total scores during the open label extension phase in the OFC or OLZ mono groups. For subjects who did not achieve remission, there was significant further reduction (improvement) in MADRS total scores in the OFC (P = 0.001) and OLZ mono (P = 0.004) groups, and in those who switched treatment groups during extension-phase treatment (P = 0.015). Of the initially non-remitted patients, 66.7% of OFC-treated patients and 64.7% of the OLZ mono patients achieved remission, defined as a MADRS total score ≤ 12, while 62.5% of patients who switched treatment groups also achieved remission during this time period. There were no significant between-groups differences in rates of relapse into acute depressive (overall, 27.4%) or manic episodes (overall, 5.9%).
In a much smaller 8-week, double-blind, placebo-controlled study conducted by Amsterdam and Shults (2005), 38 34 patients with BP-I or -II (94% BP-I), depressed phase (HAM-D [17-item] 47 ≥18 at study entry), were randomized to receive OFC (n = 8; mean [SD] dose = 7.1 ± 3.4/18.0 ± 9.9 mg/day), OLZ mono (mean [SD] dose = 6.6 ± 4.5 mg/day), fluoxetine monotherapy (FLX mono, mean [SD] dose = 17.8 ± 10.g mg/day) or PLC. All study drugs were flexibly dosed. Of note, 5 patients (15% of the total sample) continued on their pre-study mood stabilizer after randomization (lithium, n = 4; valproic acid, n = 1). Efficacy measures included the Hamilton Depression Rating scale (HAM-D) and MADRS. There was a significant reduction in both HAM-D and MADRS scores over time for the entire 34- patient sample (P < 0.006); however, there were no statistically significant differences in improvement according to study drug. Rates of positive treatment response, defined as a ≥50% reduction in HAM-D (17-item) scores from baseline, also did not differ significantly between treatment groups.
Brown and colleagues (2006) 39 conducted a larger randomized, double-blind, 7-week comparison of OFC and the mood stabilizer, lamotrigine, in acutely depressed subjects with BP-I. Eligible subjects were required to have a MADRS total score ≥20 and a Clinical Global Impressions-Severity (CGI-S) scale score of ≥4 at study entry. The majority (53.6%) of OFC-treated patients took the 12/50 mg dosage at study endpoint. The mean modal dose of OFC during follow-up was 10.7 ± 2.4/38.3 ± 12.5 mg/day, while the mean modal dose of lamotrigine was 106.4 ± 81.9 mg/day. Most of the lamotriginetreated patients (73.3%) were successfully titrated to 200 mg/day target dose by week 5 of the study, while one patient required a reduction in dosage to 150 mg daily. The remainder dropped out before of the study prior to achieving the lamotrigine target dose. OFC treatment resulted in significantly greater improvement over time in Clinical Global Impression-Severity 48 (CGI-S) and MADRS scores. OFC also resulted in significantly greater baseline to end-point change in CGI-S, MADRS, and YMRS total scores on last observation carried forward (LOCF) analysis (Table 2). Median time to positive treatment response, defined as ≥50% reduction in MADRS total score, was significantly reduced in the OFC group (17 days, 95% CI 14 to 22 days) compared with the lamotrigine group (23 days, 95% CI 21 to 34 days; P = 0.01). There were trend level differences in rates of positive treatment response (OFC, 68.8% vs. lamotrigine, 59.7%, P = 0.07). Neither remission rate, nor time to remission, differed significantly between treatment groups when remission was defined as either a MADRS score ≤12 or ≤7. Greater improvements in Global Assessment of Functioning (GAF), 49 Brief Symptom Inventory-Severity of Illness scale (BSI), 50 Clinical Global Impressions-Improvement (CGI-I), 48 and Patient Global Impression of Improvement (PGI) 51 scale scores were significantly greater with OFC than lamotrigine, with therapeutic effect sizes ranging from 0.21 to 0.25 for these secondary effectiveness measures. Interestingly, there was significantly greater improvement on the suicide item (item 10) of the MADRS in the OFC group compared with the lamotrigine group (OFC, -0.9 vs. lamotrigine, -0.74, P = 0.004), with significant between groups differences noted at weeks 1, 2, 4 and 5.
Six-month data from this study were also published and will be mentioned briefly here. 52 Six month completion rates in the OFC (33.2%) and lamotrigine groups (33.7%) were low, but not significantly different. Mean modal doses of OFC and lamotrigine were 10.7/40.8 mg/day and 149.7 mg/day, respectively. Over the 6 month observation period, there was significantly greater improvement in CGI-S (P = 0.008), MADRS (P = 0.005), and YMRS (P < 0.001) scores over time in the OFC group compared with the lamotrigine group. Mean change (improvement) in CGI-S (P = 0.04), MADRS (P = 0.02), and YMRS (P = 0.001) scores from the end of lamotrigine titration (week 5) to the study endpoint (week 25) was also significantly greater for the OFC group. Among the patients who achieve remission at the end of the 7-week acute phase study, 39 there were no significant between groups differences in rates of relapse, defined as a MADRS total score >15 during follow-up.
Randomized, open-label studies
As mentioned earlier, Tamayo et al (2009) 40 conducted a randomized, open-label study of OFC in patients with bipolar depression that proceeded in two periods (Periods 1 and 2). During Period 1, 161 subjects with BP-I or –II and a MADRS score of ≥20 at study entry received 7 weeks of open-label treatment with OFC (initiated at 12/25 mg/day; mean modal dose, 10.8/27.8 mg/day during Period 1). Those who achieved positive treatment response (Clinical Global Impressions of Severity of Bipolar Depression (CGI-BP-D) 43 score of ≤3 and a reduction in MADRS score of ≥50% from baseline) were randomized in Phase 2 to continued OFC treatment (mean modal dose, 10.3/33.6 mg/day) or OLZ monotherapy (mean modal dose, 10.8 mg/day). The primary study objective was to assess the efficacy of OFC during Period 1; OFC resulted in significant improvement in MADRS total score from baseline over 7 weeks (Table 2). Significant improvement in MADRS and CGI-BP-D scores were observed at the OFC starting dose during week 1. A total of 107 (69.0%) subjects achieved positive treatment response, while 91 (58.7%) achieved remission, defined as a MADRS total score of ≤12 at 7 weeks. A total of 114 subjects achieved remission and/or positive treatment response and were randomized to OFC or OLZ monotherapy (Period 2).
During Period 2, there was minimum further improvement in MADRS total scores over 12 weeks in the OFC group; however, MADRS total CGI-BP-D scores increased (worsened) in the OLZ monotherapy arm (Table 2). Rates of relapse into either a depressive or manic episode were significantly lower with OFC than OLZ monotherapy (28.1 vs. 10.5%, P < 0.05). Rates of depressive relapse, defined as having a MADRS total score ≥ 20 or hospitalization for depression during Phase 2 follow-up, were significantly lower in the OFC group than the OLZ monotherapy group. Manic relapses were infrequent in both groups, and between-groups differences were not statistically significant (OFC, 1.8% vs. OLZ monotherapy, 0.0%, P = NS).
Short-Term Safety and Tolerability
Discontinuation related to adverse effects
A summary of safety and tolerability results from each of the short-term randomized studies are presented in Table 3. Rates of study discontinuation due to treatment-emergent adverse events (TEAEs) were generally low for all treatment groups. In the Tohen et al (2003) study, 37 rates of discontinuation due to TEAEs was significantly lower in the OFC group compared OLZ monotherapy, but not placebo. Differences in rates of study discontinuation due to TEAEs did not differ significantly between groups in the OFC-lamotrigine comparison by Brown et al (2006). 39 OFC discontinuation due to TEAEs in Period 1 of the Tamayo et al (2009) 40 study was numerically similar to corresponding OFC discontinuation rates in the Tohen et al (2003) 37 study.
Selected tolerability and safety results from four acute-phase or short-term randomized studies of the olanzapine-fluoxetine combination (OFC) in patients with bipolar depression.
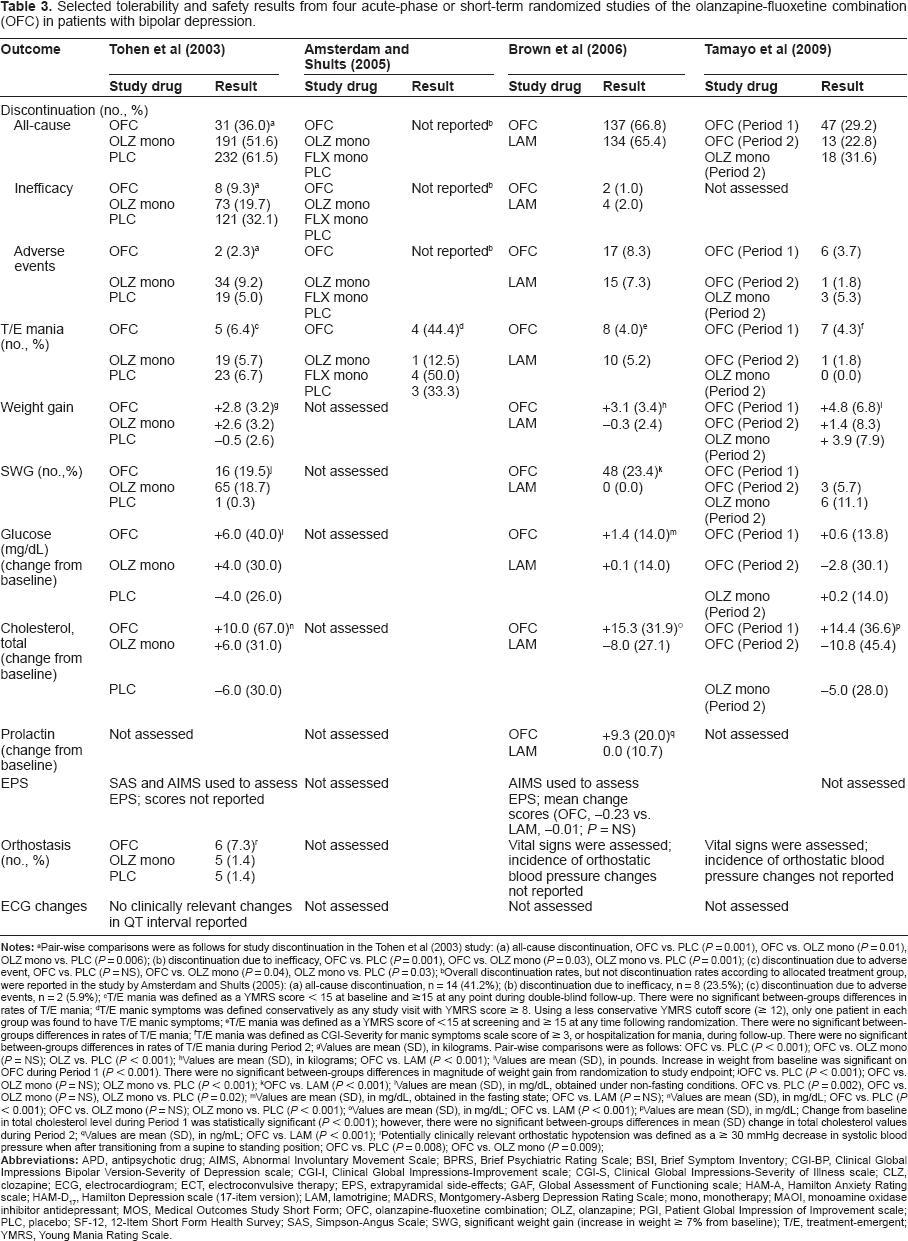
Pair-wise comparisons were as follows for study discontinuation in the Tohen et al (2003) study: (a) all-cause discontinuation, OFC vs. PLC (P = 0.001), OFC vs. OLZ mono (P = 0.01), OLZ mono vs. PLC (P = 0.006); (b) discontinuation due to inefficacy, OFC vs. PLC (P = 0.001), OFC vs. OLZ mono (P = 0.03), OLZ mono vs. PLC (P = 0.001); (c) discontinuation due to adverse event, OFC vs. PLC (P = NS), OFC vs. OLZ mono (P = 0.04), OLZ mono vs. PLC (P = 0.03);
Overall discontinuation rates, but not discontinuation rates according to allocated treatment group, were reported in the study by Amsterdam and Shults (2005): (a) all-cause discontinuation, n = 14 (41.2%); (b) discontinuation due to inefficacy, n = 8 (23.5%); (c) discontinuation due to adverse events, n = 2 (5.9%);
T/E mania was defined as a YMRS score < 15 at baseline and ≥15 at any point during double-blind follow-up. There were no significant between-groups differences in rates of T/E mania;
T/E manic symptoms was defined conservatively as any study visit with YMRS score a 8. Using a less conservative YMRS cutoff score (≥ 12), only one patient in each group was found to have T/E manic symptoms;
T/E mania was defined as a YMRS score of <15 at screening and ≥ 15 at any time following randomization. There were no significant between-groups differences in rates of T/E mania;
T/E mania was defined as CGI-Severity for manic symptoms scale score of ≥ 3, or hospitalization for mania, during follow-up. There were no significant between-groups differences in rates of T/E mania during Period 2;
Valuesare mean (SD), in kilograms. Pair-wise comparisons were as follows: OFC vs. PLC (P < 0.001); OFC vs. OLZ mono (P = NS); OLZ vs. PLC (P < 0.001);
Values are mean (SD), in kilograms; OFC vs. LAM (P < 0.001);
Values are mean (SD), in pounds. Increase in weight from baseline was significant on OFC during Period 1 (P < 0.001). There were no significant between-groups differences in magnitude of weight gain from randomization to study endpoint;
OFC vs. PLC (P < 0.001); OFC vs. OLZ mono (P = NS); OLZ mono vs. PLC (P < 0.001);
OFC vs. LAM (P < 0.001);
Values are mean (SD), in mg/dL, obtained under non-fasting conditions. OFC vs. PLC (P = 0.002), OFC vs. OLZ mono (P = NS), OLZ mono vs. PLC (P = 0.02);
Values are mean (SD), in mg/dL, obtained in the fasting state; OFC vs. LAM (P = NS);
Values are mean (SD), in mg/dL; OFC vs. PLC (P < 0.001); OFC vs. OLZ mono (P = NS); OLZ mono vs. PLC (P < 0.001);
Values are mean (SD), in mg/dL; OFC vs. LAM (P < 0.001);
Values are mean (SD), in mg/dL; Change from baseline in total cholesterol level during Period 1 was statistically significant (P < 0.001); however, there were no significant between-groups differences in mean (SD) change in total cholesterol values during Period 2;
Values are mean (SD), in ng/mL; OFC vs. LAM (P < 0.001);
Potentially clinically relevant orthostatic hypotension was defined as a ≥ 30 mmHg decrease in systolic blood pressure when after transitioning from a supine to standing position; OFC vs. PLC (P = 0.008); OFC vs. OLZ mono (P = 0.009);
Treatment-emergent mania/hypomania
Treatment emergent mania (TEM) was variously defined across studies as having a YMRS score of <15 at baseline and ≥15 at any point during follow-up in two studies (Tohen et al 2003; Brown et al 2006);37,39 satisfying DSM-IV criteria for a manic or hypomanic episode or achieving a YMRS total score of ≥8 or ≥12 (two symptom-based thresholds were tested) during follow-up in the study by Amsterdam and Shults, 2005; 38 and having a CGI-BP-Mania score ≥3 or a hospitalization for mania at any time during follow-up in the Tamayo et al (2009) study. 40 In spite of these differences, TEM rates were generally low across studies (Table 3). The highest TEM rates were reported in the study by Amsterdam and Shults (2005). 38 However, because each treatment group consisted of only 8–9 randomized subjects and a low threshold for defining TEM was used (YMRS ≥8), interpretation of reported TEM rates is limited. Moreover, at a more conservative YMRS cut-off (≥12), only one patient in each group was defined as having TEM.
Weight gain
Mean (SD) changes in weight in each of the reviewed studies are presented in Table 3. Patients randomized to OFC experienced significantly greater weight gain than placebo in the study by Tohen et al (2003), 37 and lamotrigine in the study by Brown et al (2006). 39 Significantly higher rates of clinically significant weight gain (defined as a ≥7% baseline to endpoint increase in body weight) were also observed with OFC compared with placebo and with lamotrigine in these studies. In the study by Tohen et al 37 there were no significant differences in mean weight gain or rates of clinically significant weight gain between patients who received OFC and OLZ monotherapy. In the study by Tamayo et al (2009), 40 significant weight gain was observed in OFC-treated patients (~5 lbs.) during the 7-week pre-randomization period (Period 1) (P < 0.001). Following randomization (Period 2), further weight gain occurred in both OFC and OLZ monotherapy groups, with OFC-treated patients experiencing a numerically greater increase; however, between groups differences in magnitude of weight gain from randomization to study endpoint were not significant.
Clinical laboratory indices
Changes in clinical laboratory parameters in the reviewed studies are also presented in Table 3. Greater mean changes in fasting glucose and total cholesterol occurred with OFC compared with placebo, but not OLZ monotherapy, in the study by Tohen et al. 37 In the Brown et al (2006) study, 39 there were no significant between groups differences for mean change in fasting glucose; however, there were significant differences between OFC and lamotrigine for change in total cholesterol. Mean cholesterol values were lower at study endpoint than those at baseline in the lamotrigine group, while mean total cholesterol values increased in the OFC group. In the Tamayo et al (2009) study, 40 total cholesterol values increased significantly from baseline in the pre-randomization period (P < 0.001). Following randomization, there was no significant difference in mean change in total cholesterol between OFC- and OLZ monotherapy-treated groups.
Brown et al (2007) 39 also examined changes in glycosylated hemoglobin (HgbA1c), high-density lipoprotein cholesterol (LDL-C), low-density lipoprotein cholesterol (LDL-C), and triglycerides during treatment with OFC and lamotrigine. Mean baseline to endpoint (7 weeks) changes in HgbA1c (OFC, 0.0 ± 0.2% vs. lamotrigine, -0.1 ± 0.2%, P < 0.001), HDL-C (OFC, +1.8 ± 10.4 mg/dL vs. lamotrigine, -0.6 ± 8.0 mg/dL, P = 0.03), LDL-C (OFC, +9.1 ± 26.6 mg/dL vs. lamotrigine, -6.8 ± 25.3 mg/dL, P < 0.001), and triglycerides (OFC, +27.0 ± 91.3 mg/dL vs. lamotrigine, -13.5 ± 110.3 mg/dL, P = 0.001) were significantly greater with OFC than lamotrigine.
In the Brown et al (2007) study, 39 there were significantly greater increases in prolactin values in the OFC group compared with the lamotrigine group. Change in prolactin concentration during treatment was not assessed in the remaining studies.
Side-effects related to basal ganglia dysfunction
The Simpson-Angus Scale (SAS) 53 and Abnormal Involuntary Movement Scale (AIMS) 54 was used to assess parkinsonism and tardive dyskinesia, respectively, in the report by Tohen et al (2003). 37 Although actual scores were not reported, the incidence of extrapyramidal symptoms were described by the authors as being low, with no statistically significant between groups differences noted. There were no significant differences in tardive dyskinesia symptoms, as measured by the AIMS, between OFC and lamotrigine groups in the study by Brown et al (2007) 39 (Table 3). Movement-related adverse effects were not formally assessed in the remaining studies.
Cardiac side-effects
No clinically relevant electrocardiographic changes in QT interval were reported in the study by Tohen et al (2003). 37 In the Tohen et al (2003) study, the incidence of orthostatic blood pressure changes was low in all three groups (Table 3). No significant differences in incidence of orthostastic blood pressure changes were noted. Although vital signs were assessed in the studies by Brown et al (2007) 39 and Tamayo et al (2009), 40 incidence of orthostatic changes were not reported.
Modifying Factors
There are no randomized, controlled studies of OFC for the treatment of bipolar depression in elderly patients. However, one single-dose pharmacokinetic study of healthy volunteers showed that the mean terminal half-life of olanzapine is ~1.5-fold longer in elderly (aged 65–79 years) compared with younger subjects (aged 20–41 years). 35 In a sample of hospitalized patients, weight-corrected olanzapine concentration/dose ratios increased by 9.4% with each increasing decade of life. 55 Comparisons were adjusted for smoking status. Age is believed to have less impact on fluoxetine pharmacokinetics, although a substantial numerical increase in the half-life for both fluoxetine and norfluoxetine (by 25%–33%) in elderly versus non-elderly volunteers in one study. 56 These age-dependent differences were not statistically significant due to high sample variability. Nevertheless, lower doses may be required when treating elderly subjects with OFC, regardless of indication.
The presence of hepatic or renal impairment is not thought to significantly alter the pharmacokinetic profile of olanzapine. 20 While renal impairment is also not believed to significantly affect fluoxetine pharmacokinetics, impaired hepatic function may significantly reduce the metabolic clearance of both fluoxetine and norfluoxetine. 57 The half-life of fluoxetine has been shown to be increased in subjects with impaired liver function.57,58 Thus, reduction in dosage may be required in patients with hepatic compromise.
Potential Drug Interactions
Several potential drug-drug interactions should be considered prior to initiating OFC therapy. First, OFC should not be given with monoamine oxidase inhibitors, or within 2 weeks of MAOI discontinuation, due to risk of potentially fatal toxicity characterized by acute mental status change, autonomic instability, rigidity and myoclonus, and hyperthermia.20,59 Caution is advised when combining OFC with serotonin-potentiating medications (including antidepressants, lithium, and others) and drugs that affect coagulation (such as warfarin). 60 Numerous other potential drug-drug interactions are described in the US prescribing information for OFC and elsewhere.20,61,62 These include interactions with drugs metabolized by CYP 2D6 and/or 3A4 oxidative isoenzymes, which are inhibited by fluoxetine. 60
Limitations
There are a number of unanswered questions and limitations of available data to consider. First, a number of important clinical subtypes of bipolar patients were excluded from the reviewed studies, including those with BP-II and comorbid substance use disorders. The smallest of the reviewed studies (n = 36 patients, inclusive) did include patients with BP-II; however, only 6% of the total sample (~2 patients) were diagnosed BP-II. Although it is more difficult to recognize, BP-II may be more prevalent than BP-I in clinical practice, and may be associated with greater burden from depressive episodes and residual symptoms. 63 The high prevalence and profound negative impact of comorbid substance use disorders on the natural history and treatment course of bipolar disorders is well recognized. 64 Additional studies of OFC for treating these important and common bipolar clinical subtypes are needed.
Second, generalizability of study findings is limited by the relatively brief follow-up periods in the reviewed studies. The focus of this paper was on short-term, acute-phase studies of OFC in bipolar depression. That said, most patients with bipolar depression will require long-term maintenance treatment, preferably with the same medication that resulted therapeutic benefit during acute-phase treatment. Currently, OFC is not indicated for bipolar maintenance treatment. The study by Brown et al (2006) was designed a priori to last 6 months, with a planned 7 week data analysis. 39 The 6-month results of this study, results of a 25-week open-label extension of the Tohen et al (2003) 37 study, and Phase 2 of the Tamayo et al (2009) study 40 are, to our knowledge, the only published reports of longer-term effects of OFC in the context of BP-I depression.46,52 Additional studies of the long-term effects of OFC treatment are needed.
Data from these studies and long-term studies of OFC for unipolar major depression suggest that the adverse effects of OFC may be similar to those of olanzapine. 65 This profile may be expected to include weight gain, and heightened risk of adverse changes in glycemic and lipid profile. 66 These metabolic risks must be carefully considered when deciding among the options bipolar depression treatment. Patients with bipolar disorder are in physically poorer health than the general population, and are thus already at risk for profound cardiovascular morbidity and early mortality. 67
Additional limiting factors include high dropout rates across studies;37–40 low power, 38 and lack of a placebo 39 or any comparison group 40 during acute-phase treatment in individual studies. In the 7-week OFC-lamotrigine comparison study by Brown et al (2006), 40 the prolonged lamotrigine titration, relatively low lamotrigine doses, and wider availability of OFC dosing options relative to that of lamotrigine may have introduced some bias towards more favorable short-term symptom improvement with OFC. Only in phase 2 of the Tamayo et al (2009) study did a randomized comparison occur between OFC and OLZ monotherapy. Phase 2 was enriched with OFC responders from phase 1, which may have introduced bias towards a more favorable OFC response after randomization. In addition, while olanzapine monotherapy may be beneficial for some patients,37,68 it is not an established treatment option for depression associated with either bipolar I or II disorder. Although results of a recent meta-analysis of randomized, controlled bipolar depression trials suggests that OFC and quetiapine, also approved for this indication, may have comparable efficacy with regard to reduction in MADRS score and response/remission rates, 69 there is a paucity of direct head-to-head comparisons between OFC and established agents for treating acute depressive episodes in patients with bipolar disorder. It is thus difficult to determine the exact placement of OFC among the other available treatment options.
Conclusions
Depression is considered by many to be the predominant mood pole in patients with bipolar I and II disorder;5,70,71 however, there are comparatively fewer agents approved by the US Food and Drug Administration (FDA) to treat bipolar depression compared with mania. OFC is approved for the treatment of bipolar I depression, and has been shown to be effective for the short-term treatment of acute bipolar I depression largely on the basis of two of the reviewed randomized, double-blind clinical trials.37,39 In these studies, OFC significantly improved depressive symptoms to a greater degree than placebo, 37 and resulted in higher rates of categorical positive treatment response and remission than placebo and olanzapine monotherapy. 37 OFC improved depressive symptoms to a greater degree than lamotrigine, and resulted in similar rates of positive treatment response and remission. 39 Although OFC was generally well tolerated, it was associated with significantly greater weight gain than placebo 37 and lamotrigine. 39 Compared with lamotrigine, OFC was also associated with more adverse changes in lipid profile, glycosylated hemoglobin, prolactin levels during short-term follow-up. 39 The most common adverse effects in the reviewed studies were dry mouth, weight gain, increased appetite and somnolence,37,39,40 an adverse effect profile that is similar to olanzapine alone.68,72 OFC was not associated with increased risk of treatment-emergent mania in any of the reviewed studies. A signal for reduced suicidality for OFC relative to lamotrigine was detected in the Brown et al (2006), 39 but is in need of replication.
Until then, available data from short-term trials suggest that OFC is an effective and generally well tolerated option for the treatment of acute depressive episodes in adults with BP-I. The broader effectiveness of OFC for the treatment of bipolar depression across clinically relevant subtypes and over long-term follow-up are needed. Comparative effectiveness studies of OFC and other available agents would also be of interest.
Disclosures
This manuscript has been approved by all authors. It represents original work by the authors, and has not been published elsewhere. In the past, Dr Bobo has received grant/research support from Cephalon. Dr Shelton has received grant/research support from Eli Lilly and Company, GlaxoSmithKline, Janssen Pharmaceutica, Pfizer, Sanofi-Aventia, Wyeth-Ayerst Laboratories, AstraZeneca Pharmaceuticals, and Abbott Laboratories; has served as a paid consultant for Pfizer, Janssen Pharmaceutica, and Sierra Neuropharmaceuticals. Dr Epstein reports no potential conflicts of interest. The authors confirm that they have permission to reproduce any copyrighted material.
Footnotes
Acknowledgements
Dr. Bobo is supported by Grant number 1K23MH087747-01A1 from the National Institutes of Mental Health.
