Abstract
Although statins are well tolerated, their widespread use has raised awareness of rare adverse events. For example, the United States Food and Drug Administration (FDA) has issued product label warnings about statin-associated myopathy, incident diabetes, and cognitive impairment. Additional statin-associated adverse effects include proteinuria, transaminitis, and hemorrhagic stroke. The underlying pathophysiology of these adverse events remains unclear, and thus little guidance exists on addressing these issues at the bedside. Although statin-associated myopathy is common, many patients eventually tolerate a statin – albeit at less than ideal doses. Incident diabetes remains difficult to predict, but high statin potency, increasing age, and preexisting diabetes risk factors may predispose patients. Cognitive impairment – described mostly in case reports – may resolve after discontinuation of the offending statin. Rosuvastatin is linked to dose-dependent proteinuria. Elevated transaminases are reversible and unlikely to cause severe consequences. History of cerebrovascular disease increases the risk of hemorrhagic stroke on statin therapy. Overall, a thorough understanding of statin-associated adverse events will help health care providers manage patients who develop these complications. Here, we review the evidence for several statin-associated adverse effects and the implications for clinical practice.
Introduction
Statins, inhibitors of 3-hydroxy-3-methyl-glutaryl-CoA (HMG-CoA) reductase, are among the most widely prescribed medications in the United States. 1 Statins are well tolerated, reduce low-density lipoprotein cholesterol (LDL-C), and prevent atherosclerotic cardiovascular disease (ASCVD). 2
Although a large body of evidence demonstrates the benefits of statin therapy, a number of rare statin-associated adverse effects have raised concern in patients and prescribers alike. In 2012, the U.S. Food and Drug Administration (FDA) issued new product labeling requirements for statins regarding the risk of statin-associated adverse effects including muscle injury due to drug–drug interactions, incident or worsened diabetes, and cognitive impairment. 3
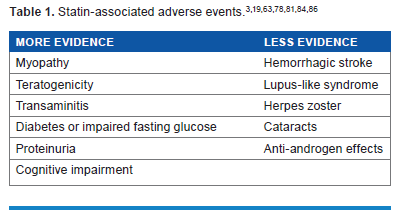
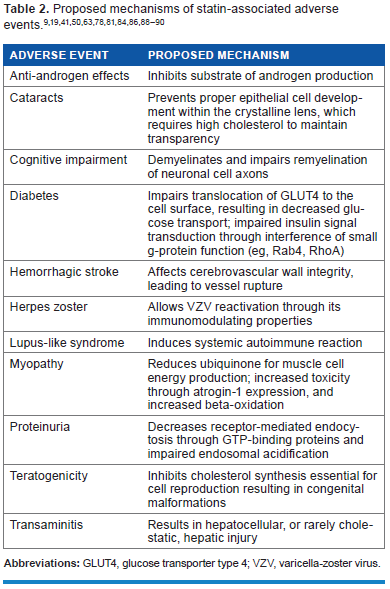
Additional statin-associated adverse effects, identified in either clinical trials or post-marketing reports, include transaminitis, teratogenicity, proteinuria, and hemorrhagic stroke (Table 1). The mechanism, clinical significance, and consequences of these risks also remain unclear (Table 2). This review discusses the evidence for several rare statin-associated adverse effects and the implications for clinical practice. We searched the current literature for “statin” and “adverse events” using PubMed and Ovid MEDLINE databases. We excluded literature with poorly documented adverse events.
Benefits of Statins
Over 30 years of published studies have shown that statins lower the risk of ASCVD events. Recently, a meta-analysis by the Cholesterol Treatment Trialists (CTT) Collaborators found that lowering LDL-C with a statin reduced the risk of a major ASCVD event by 21% for every ~39 mg/dL (1 mmol/L) reduction – irrespective of age, sex, baseline LDL-C, or previous ASVCD. The proportional reduction of events in patients with low risk for vascular disease (ie, baseline 5-year risk <10%) was comparable to the reduction seen in high-risk patients. 4 Another meta-analysis by the CTT Collaborators found that high-intensity statin regimens resulted in a 15% further reduction in major vascular events (95% CI: 11–18; P < 0.0001). 5 Both meta-analyses also describe a 10% reduction in all-cause mortality per ~39 mg/dL LDL-C reduction (RR: 0.91; 95% CI: 0.85–0.97 and RR: 0.90, 95% CI: 0.83–0.97, respectively).4,5
Statins may have cholesterol-independent or “pleiotropic” effects: improving endothelial function, stabilizing atherosclerotic plaques, decreasing oxidative stress and inflammation, and inhibiting thrombogenesis. 6 Of note, the evidence for these benefits comes from animal studies; more evidence is necessary to validate these effects in humans.
Adverse Events
Diabetes risk
In 2012, the FDA added information concerning an effect of statin on incident diabetes and increases in hemoglobin A1C (HbA1C) and/or fasting plasma glucose to statin labels. Their concern was based on studies of rosuvastatin and atorvastatin, the findings of a meta-analysis, and data from the Women's Health Initiative. 3
Statins have several proposed effects on glucose homeostasis, including diminished insulin release, altered glucose metabolism, and decreased insulin sensitivity. 7 Diminished insulin release may be the result of impaired ubiquinone synthesis, and consequently, delayed adenosine triphosphate production. 8 Altered glucose metabolism may be the result of either decreased plasma levels of adiponectin, a hormone that suppresses hepatic gluconeogenesis and reverses insulin resistance, 8 or increased expression of glucose transporter type 1 (GLUT1) in adipocytes, resulting in decreased insulin-stimulated glucose disposal. 7 Atorvastatin is linked to a concentration-dependent underexpression of glucose transporter type 4 (GLUT4), which incorporates extracellular glucose into cells.8,9 Impaired translocation of GLUT4 reduces glucose transport into adipocytes and causes insulin resistance. Atorvastatin depresses insulin signal transduction by interfering with small g-proteins (eg, Rab4, RhoA), which anchor the receptor to the cell membrane. 9 The primary mechanism is depletion of isoprenyl groups required for Rab4 and RhoA function. 9 RhoA may regulate another factor in insulin signal transduction, namely, insulin receptor substrate 1 (IRS-1), which atorvastatin affects through decreased tyrosine phosphorylation. 9
The Justification for the Use of Statins in Primary Prevention: An Intervention Trial Evaluating Rosuvastatin (JUPITER) study first reported an increased risk of incident diabetes in 2008. 10 JUPITER evaluated rosuvastatin 20 mg/day (n = 8,901) versus placebo (n = 8,901) in patients with normal LDL-C (<130 mg/dL) and elevated high-sensitivity C-reactive protein (hsCRP) (≥2 mg/dL) for a median of 1.9 years. 10 Incident diabetes occurred more frequently in the rosuvastatin arm: 270 (3.0%) of patients on rosuvastatin developed diabetes compared to 216 (2.4%) of patients on placebo [hazard ratio (HR): 1.25; 95% CI: 1.05–1.49; P = 0.01]. 10 Regardless of baseline diabetes risk factors, the median HbA1C increased by 0.1% for patients on rosuvastatin compared to patients on placebo at 24 months (HbA1C = 5.9% and 5.8%, respectively, P = 0.001). 10 Subsequent post hoc analyses of JUPITER reported that the average time to diabetes diagnosis was 5.4 weeks earlier in patients in the rosuvastatin group. For patients in the rosuvastatin arm with ≥1 diabetes risk factors (including metabolic syndrome, impaired fasting glucose, body mass index >30 kg/m2, and HbA1C >6%), the HR for developing diabetes was 1.28 (95% CI: 1.07–1.54) compared to placebo.10,11 Oddly, the risk of diabetes remained similar as the number of diabetes risk factors increased. Of note, the JUPITER study population may be inherently at greater risk for developing diabetes: the inflammatory biomarker hsCRP has been described as an independent predictor of cardiovascular events and incident hypertension and type 2 diabetes, and nearly all of the study patients who developed diabetes on rosuvastatin therapy had impaired fasting glucose at baseline.12,13
A meta-analysis by Sattar et al evaluated 13 randomized controlled trials (n = 75,507) from 1994 to 2009 to determine the relationship between statin use and development of diabetes.14–26 The included trials had more than 1,000 patients, identical follow-up in each group, and duration >1 year. 27 The meta-analysis reported a 9% increased likelihood of incident diabetes for patients on statin therapy (OR: 1.09; 95% CI: 1.02–1.17) and one additional case of diabetes per 255 patients taking a statin for 4 years (95% CI: 150–852). 27 The authors deemed this a low risk given the demonstrated reduction of coronary events with statin therapy.27,28 Sattar et al also noted an association between age and incident diabetes risk (P = 0.019 for meta-regression of baseline age).10,27
Statin potency may affect the risk of developing diabetes. Several studies of low-to-moderate intensity statins failed to find associations with diabetes incidence. The West of Scotland Coronary Prevention Study (WOSCOPS) evaluating the low-to-moderate intensity statin pravastatin did not find an increased risk.21,27 Japanese post-marketing surveillance data of the low to moderate intensity statin pitavastatin (n = 19,925) failed to identify diabetes as a common adverse event. 29 In fact, patients with type 2 diabetes (n = 6,852) enjoyed a 0.28% decrease in A1C after 104 weeks of pitavastatin therapy (P < 0.001). 29 Several Japanese studies have reported neutral effects of pitavastatin on fasting blood glucose in patients with metabolic syndrome, although these studies were limited in sample size or retrospective in design.30–32 The Japan Prevention Trial of Diabetes by Pitavastatin in Patients with Impaired Glucose Tolerance (J-PREDICT) study, scheduled for completion in 2015, will be the first randomized, controlled trial to evaluate the effect of statins on incident diabetes as a primary endpoint.33,34 Over 1,200 patients were randomized to either lifestyle modifications alone or pitavastatin 1–2 mg/day in addition to lifestyle modifications for a 5-year follow-up. 34
Overall, published data suggest a modest 6% to 9% increased relative risk of incident diabetes due to statin therapy. The risk of diabetes appears to be magnified depending upon the intensity of statin therapy and perhaps also on age and whether patients have pre-existing diabetes risk factors. The 2013 ACC/AHA Blood Cholesterol guidelines note that the long-term adverse effects of statin-associated cases of diabetes over a 10-year period are unclear and are unlikely to be equivalent to a myocardial infarction (MI), stroke, or ASCVD death. 26 As such, the benefits of statins outweigh the diabetes risk in patients with moderate-to-high cardiovascular risk.27,28 In patients with the lowest cardiovascular risk, the risk of incident diabetes should be weighed. 27 All individuals receiving statins should be counseled on healthy lifestyle habits. Individuals receiving statin therapy should be evaluated for new-onset diabetes mellitus according to the current diabetes screening guidelines; if diabetes develops, the ACC/AHA guidelines recommend continuing statin therapy to reduce their risk of ASCVD events. 26
Myopathy
Statin-induced myopathy remains one of the most frequently discussed side effects of statin treatment. We recently published an in-depth review on statin-induced myopathy; therefore, only an abbreviated discussion is included here. 35
Symptoms typically involve proximal, symmetric muscle weakness or soreness, muscle tenderness, and/or cramping with or without elevations in serum creatine kinase. Recently, the National Lipid Association Statin Muscle Safety Task Force defined statin-related muscle problems in terms of the spectrum of severity: myalgia, myopathy, myositis, myonecrosis, and rhabdomyolysis. 36 Several risk factors for statin-induced myopathy have been reported, including higher doses, older age, female sex, lower BMI, diabetes mellitus, hypertension, hypothyroidism, renal disease, and hepatic disease.
Discrepancy exists between clinical practice and clinical trials in reporting the prevalence of statin-associated muscle problems. Statin therapy is associated with muscle problems in approximately 11% to 29% of patients treated in clinical practice, whereas it is reported to be about 1% to 5% in controlled clinical trials. 36 A meta-analysis of 42 randomized trials of statins found little or no excess risk of myalgias, creatine kinase elevations, rhabdomyolysis, or discontinuation of therapy versus placebo. 37 The study did find a slightly increased incidence of muscle problems with statin treatment (12.7%) than with the placebo group (12.4%, P = 0.06). 37
Clinical trials tend to exclude participants with a history of muscle-related problems and do not retain those who develop such problems, and therefore they may lead to underreporting. On the other hand, many observational studies have reported a higher prevalence of statin-associated muscle pain. An observational study in an outpatient setting in France of 7,924 hyperlipidemic patients receiving high-dosage statin therapy (PRIMO – Prédiction du Risque Musculaire en Observationnel or Prediction of Muscular Risk in Observational Conditions) reported muscular symptoms in 832 patients (10.5%), with a median time of onset of 1 month following initiation of statin therapy. 38 Another observational study (USAGE – Understanding Statin Use in America and Gaps in Patient Education), which surveyed 10,138 statin current and former users, reported muscle-related side effects in 60% and 25% of former and current users, respectively (P < 0.05). 39 In order to study the discrepancy between clinical practice and clinical trials more carefully, a double-blinded clinical trial of 420 healthy statin-naïve participants (STOMP – Effect of Statins on Skeletal Muscle Function and Performance) compared symptoms and measured creatine kinase, exercise capacity, and muscle strength before and after atorvastatin 80 mg/day versus placebo for 6 months. In this study, more subjects on atorvastatin (n = 19) than placebo (n = 10) developed myalgia (P = 0.05). 40
The exact mechanism for pathogenesis of statin-induced muscle injury remains unclear. Several mechanisms have been proposed. The most popular involves reduction in ubiquinone, as it plays a role in muscle cell energy production.41–43 Other factors continue to be implicated: elevated levels of β-sitosterol due to reduced fat synthesis and increased β-oxidation 44 and altered expression of atrogin-1, a muscle-specific ubiquitin protein ligase that plays a role in toxicity. 45
In approaching patients with statin-induced muscle problems, a commonly used strategy is statin withdrawal and rechallenge, in which the statin is stopped for at least 2 weeks and then restarted to see if symptoms return. The ACC/AHA Blood Cholesterol Guideline recommends this method, 28 although the effectiveness of this strategy has not been studied systematically.
A recent retrospective cohort study at the Brigham and Women's Hospital and Massachusetts General Hospital including 2,721 outpatients showed that, 12 months after the initial statin-related discontinuation, 92.2% of patients were tolerating statin therapy. 46 Additionally, a retrospective chart review of 1,605 patients at the Cleveland Clinic Preventive Cardiology Service with documented statin intolerance who were rechallenged with statin therapy observed that 72.5% of patients were able to tolerate long-term statin therapy. 47
For those patients who fail the rechallenge, non-statin lipid-lowering agents can be used, such as ezetimibe, fibrates, cholestyramine, colesevelam, colestipol, and niacin. LDL-apheresis may be an option in severe cases of LDL-C elevations such as in individuals affected with familial hypercholesterolemia. 48
Cognitive impairment
The 2012 FDA-issued drug safety communication regarding statin-associated cognitive impairment was founded upon a review of existing literature and the Adverse Event Reporting System (AERS) database. 3 The clinical presentation of statin-associated cognitive impairment was outlined by the FDA as follows: “notable but ill-defined memory loss,” onset ranging from 1 day to several months after statin exposure, and reversible after discontinuation with a median time to resolution of 3 weeks. Anecdotal reports have also noted a variety of psychiatric adverse events, including aggressive behavior, amnesia, confusion, dementia, depression, hallucination, insomnia, and/or somnolence. 49
Cholesterol composes approximately 50% of the central nervous system dry matter, particularly the myelin sheaths and cell synapses in neuronal cells.50,51 The brain also serves as a site of de novo synthesis of cholesterol. When statins penetrate the blood–brain barrier, they may inhibit cholesterol synthesis and cause chemical demyelination and/or impaired re-myelination of the neuronal cell axons. Damage to the myelin sheath may alter impulse conduction, resulting in decreased membrane excitability and severe impairment of nerve cell function. Statins may also affect cognition by impairing mitochondrial functioning and increasing oxidative stress. 52 Statins inhibit mevalonate, which is the precursor to coenzyme Q10, also known as ubiquinone, in addition to cholesterol. Coenzyme Q10 is a lipid-soluble molecule that serves as an energy transporter for adenosine triphosphate production and a free-radical scavenger.
Parker et al used functional magnetic resonance imaging (fMRI) to evaluate neuronal activation in an elderly patient with statin-associated cognitive decline (documented with neuropsychological tests). 50 MRI findings included reduced activation of the dorsolateral prefrontal cortex (DLPFC) and hyperactivation of the prefrontal cortex (PFC) during neuropsychological tests. DLPFC is involved in motor planning, organization, memory, and sensory integration, and reduced activation of this area has been noted in patients with certain psychological disorders such as schizophrenia and major depressive disorder. PFC plays a role in executive function, and hyperactivity of the PFC is also manifested in schizophrenic patients as disturbed thought processes. After discontinuation of the statin, cognitive symptoms resolved and neuronal activation became similar to off-statin findings in healthy individuals. Subsequent MRI findings demonstrated that the patient unlikely had overt cognitive impairment or baseline dementia that would cause fluctuations in cognition during statin therapy.
Prospective studies of statins and cognitive function have found either neutral or protective effects. The PROSPER study evaluated the effect of pravastatin on cognitive function as a tertiary objective in 5,804 individuals, aged 70–82 years 23 ; the results of four repeated psychometric examinations failed to identify any effect of pravastatin on cognitive function compared to placebo. 53 Randomized controlled trials of simvastatin and atorvastatin failed to find protective effects for the progression of Alzheimer's disease, as defined by the change in Alzheimer's Disease Assessment Scale-Cognitive Subscale.54,55 The Cardiovascular Health Study, a prospective epidemiological study of 3,334 elderly patients, found a lower rate of decline in the Mini-Mental State Examination among patients on statin therapy compared to an untreated group in which lipid-lowering therapy was not recommended (95% CI: 0.12–0.85; P = 0.009). 56 Swiger et al performed a meta-analysis of studies evaluating statins and cognitive decline as the primary outcome 57 ; pooled results showed a 29% relative risk reduction and 2% absolute risk reduction in incident dementia in statin users (HR: 0.71; 95% CI: 0.61–0.82; NNT for 6.2 years = 50).
The effects of statins on cognitive decline remain unclear, although anecdotal reports describe convincing cases of statin-induced confusional state and memory impairment. Data are lacking regarding patient-specific (eg, baseline cognition, concomitant medications) and statin-specific (eg, potency) factors that may predispose patients. When a patient presents with cognitive impairment, the ACC/AHA Blood Cholesterol guidelines committee recommends an evaluation for non-statin causes, such as exposure to other drugs, as well as for systemic and neuropsychiatric causes, in addition to the possibility of adverse effects due to statins. Little guidance is available for managing hyperlipidemia in patients who develop statin-induced cognitive decline. Case reports seem to suggest that the offending statin can be stopped, and once the symptoms resolve, an alternative agent may be used.50,51
Transaminitis
When statins first became widely used in the 1980s, statin-induced transaminitis was regarded as an important side effect, and, historically, routine monitoring of liver enzyme levels was recommended. For example, early data with lovastatin found it to cause mild alanine aminotransferase (ALT) elevations in 10% of recipients and more serious elevations – more than 3 times the upper limit of normal (ULN) – in 1% to 3% of patients. 58
More recent data from both retrospective and placebo-controlled studies indicate statin-associated transaminitis to be either insignificant or similar to placebo groups. A review of a health maintenance organization's computerized records from 1997 to 2001 on 23,000 patients who were receiving statins found that only 17 (0.1%) had an ALT level more than 10 times the ULN attributable to statin therapy. 59 In the Scandinavian Simvastatin Survival Study, patients developing an ALT level greater than 3 times the ULN did not differ between the simvastatin (0.7%) and placebo (0.6%) groups. 60 Another study, which pooled 112,000 person-years of pravastatin exposure in double-blind randomized trials, showed no excess liver function abnormalities between pravastatin and placebo. 61
In a prospective registry by the U.S. Drug Induced Liver Injury Network between 2004 and 2012, 22 out of 1,188 cases of drug-induced liver injury were attributed to a statin. 62 Four of those 22 cases were considered severe, and one patient died; otherwise, the liver injury was largely mild-to-moderate in severity and self-limited in due course. The only fatal case occurred in a patient with severe underlying alcoholic liver disease. 62
In summary, elevations of transaminases rarely occur with statin therapy, and the elevation is usually reversible without any significant clinical consequences. Frequent monitoring may explain why clinical trials report a higher incidence of transaminitis than observational studies; elevations of liver enzymes, ALT in particular, also occur with gallbladder disease, infectious liver disease, passive hepatic congestion secondary to congestive heart failure, and non-alcoholic fatty liver disease. When initiating statin therapy, the current FDA guidelines recommend liver enzymes be checked; routine monitoring, however, is unnecessary if the liver enzymes are normal at baseline. 3
Proteinuria
Statin-induced proteinuria, as well as hematuria and two cases of acute renal failure, surfaced in clinical trials of rosuvastatin 80 mg/day, a dose never approved by the FDA. Roughly 12% of patients exposed to rosuvastatin 80 mg/day developed proteinuria.63,64 In addition, 40 mg of rosuvastatin was also associated with proteinuria, as measured by urine dipstick, albeit at a lower rate of 3% to 5% according to the FDA's pooled safety analysis.
Several clinical trials have shown that rosuvastatin causes more proteinuria than other statins.63,64 Stein et al reported a shift in urine dipstick (from “non” or “trace” at baseline to “+” or greater in the first 4 weeks) in 6.4% of patients receiving rosuvastatin 40 mg/day (n = 308; 95% CI: 3.9%–9.8%) and 1% of those receiving simvastatin 80 mg/day (n = 318; 95% CI: 0.2%–2.9%). 65 In the SATURN study, rosuvastatin 40 mg/day (n = 694) caused more new proteinuria (defined as 2+ or greater protein on urinalysis during the follow-up period in patients with negative finding or trace protein at baseline) than atorvastatin 80 mg/day (n = 691) (3.8% vs 1.7%, P = 0.02). 66
Several mechanisms have been proposed for statin-induced proteinuria. High-dose statins cause renal tubular degeneration related to an inhibition of proximal tubular protein reabsorption. 67 These findings may be due to impairment of receptor-mediated endocytosis affecting the activity of GTP-binding proteins or drug accumulation impairing endosomal acidification. 68 A study in an opossum kidney cell line supported this hypothesis by relating protein uptake inhibition to the degree of HMG-CoA reductase inhibition. 69
In approaching patients who develop statin-associated proteinuria, the FDA guidelines recommend considering dose reduction in addition to investigating for alternative causes. 70
Hemorrhagic stroke
Overall, statins reduce the risk of “any” stroke. 5 The benefit, however, is limited to the risk of ischemic strokes 5 ; the benefit for hemorrhagic strokes remains unclear, and, in fact, statins may slightly increase the risk of hemorrhagic stroke and intracerebral hemorrhage.5,71,72
For example, the Heart Prevention Study (HPS), a study of simvastatin 40 mg/day (n = 10,269) vs placebo (n = 10,267), found an overall reduction of 25% (95% CI: 15–34) for the incidence rate of first stroke. 16 This benefit was attributed almost entirely to the reduced risk of ischemic stroke, while no differences existed in the rates of hemorrhagic stroke (simvastatin 0.5% vs placebo 0.5%, nonsignificant). The Stroke Prevention by Aggressive Reduction in Cholesterol Levels (SPARCL) trial, a study of atorvastatin 80 mg/day vs placebo in patients with a history of stroke or transient ischemic attack, found an overall 16% reduction in the risk of nonfatal or fatal stroke but an increased HR of 1.66 (95% CI: 1.08–2.55) for hemorrhagic stroke. 73
The underlying mechanism remains unclear, although it appears to be linked with low circulating cholesterol levels. A meta-analysis of 23 prospective cohort studies (n = 1,430,141) – all investigating the association between lipid levels and risk of hemorrhagic stroke – found an overall inverse association between total cholesterol levels and the risk of haemorrhagic stroke (OR: 0.69; 95% CI: 0.59–0.81, P < 0.01). Potential mechanisms include low cholesterol levels leading to compromised cerebrovascular wall integrity and promotion of arterial medial layer smooth muscle cell necrosis, 74 or cholesterol levels reflecting nutritional status, suggesting nutritional deficiencies that increase the risk of hemorrhagic stroke.5,74
Other Reported Adverse Events
The FDA classifies statins as pregnancy category X: contraindicated during pregnancy. 75 The FDA derived this recommendation from animal studies of maternally toxic doses of lovastatin, simvastatin, and atorvastatin. 76 Teratogenicity includes fetal skeletal malformations, decreased body weight, and/or developmental delay. 76 Women of childbearing potential should be counseled prior to initiation of statins. They should consider using contraception, and statin therapy should be withdrawn a few months prior to attempting conception.75,77
Leuschen et al reported an increased risk of cataract among statin users (n = 13,626) compared to nonusers (n = 32,623) in a propensity score-matched cohort analysis using a military database (OR: 1.27; 95% CI: 1.15–1.40). 78 A large prospective cohort by Foody et al (n = 2,004,692) also showed an increased risk of cataract during statin therapy (NNH = 33; 95% CI: 24–57). 79 In contrast, most observational studies note a neutral effect or decreased risk. 80 More robust studies are necessary to support a stronger association or justify any changes in therapy.
Statins have been suggested as a treatment for polycystic ovarian syndrome (PCOS), perhaps due to an antisteroidogenic effect. 81 Meta-analyses by Gao et al and Schooling et al report statistically significant decreases in total testosterone.82,83 Schooling et al observed a greater testosterone reduction in middle-aged men (-0.66 nmol/L; 95% CI: -0.14 to -1.18) compared to young women with PCOS (-0.44 nmol/L, 95% CI:-0.75 to -0.13). 83 The long-term implications of these anti-androgenic properties remain unknown.
De Jong et al found that 3% of all lupus-like syndrome cases (n = 3,362) in the World Health Organization global individual case safety reports database (VigiBase) reported statins as the suspected drug. 84 Several anecdotal reports have reported patients on simvastatin or atorvastatin developing skin eruptions months after statin initiation. 85
A retrospective cohort study in Asia found a greater risk of herpes zoster in patients on statin therapy (HR = 1.21; 95% CI: 1.13–1.29). 86 This study identified a higher risk among younger statin users (<49 years; HR = 1.35; P < 0.01 vs <64 years; HR = 1.33; P < 0.001 vs >65 years; HR = 1.24; P < 0.001) despite the fact that the incidence of herpes zoster is usually higher among older patients in the general population.86,87
Conclusions
Clinical trials and post-marketing reports note a variety of statin-associated adverse events. Complications such as myopathy, transaminitis, and proteinuria are well documented and usually resolve upon discontinuation of the statin. Incident diabetes and cognitive impairment were identified more recently, but data are lacking regarding underlying mechanisms and management strategies. Overall, health care providers should be aware of these potential adverse effects and, when appropriate, weigh the benefits and risks of statins for individual patients.
Author Contributions
Conceived and designed the experiments: WCY, HK, ZA. Analyzed the data: WCY, HK, ZA. Wrote the first draft of the manuscript: WCY, HK. Contributed to the writing of the manuscript: WCY, HK, ZA. Agree with manuscript results and conclusions: WCY, HK, ZA. Jointly developed the structure and arguments for the paper: WCY, HK, ZA. Made critical revisions and approved final version: WCY, HK, ZA. All authors reviewed and approved of the final manuscript.