Abstract
Drug resistance has emerged as a major obstacle to global control of tuberculosis (TB). Treatment with currently available medications requires prolonged courses of numerous drugs, many of which are associated with significant adverse effects. New drugs are urgently needed to meet the challenge posed by drug-resistant TB, in order to relieve the burden that drug resistance has placed on individual patients and health systems. Fortunately, many new drugs have been developed for the treatment of drug-resistant TB in recent years. The new antimycobacterial agents are derived from various medication classes, work by means of an assortment of mechanisms of action, and are at differing phases of development. This review provides a survey of the most promising new agents, in order to promote familiarity with these emerging drugs and compounds.
Introduction
The twentieth edition of the World Health Organization's Global Tuberculosis (TB) Report describes a disease that continues to bring about a startling amount of death and dysfunction.
1
In 2014, 9.6 million people developed active TB, and 1.5 million people died of the disease. Though there have been significant declines in TB prevalence and mortality rates in recent decades, these gains have been threatened by the rampant emergence of drug-resistant strains of
Multidrug resistance presents a major challenge to TB treatment and control. Even in the setting of pan-sensitive disease, TB management places a significant burden on patients and health systems, requiring the use of multiple medications, each with significant toxicities, for durations of at least six months.5–7 In the setting of multidrug resistance, both the number of drugs and the duration of treatment need to be substantially increased. 4 The most recent World Health Organization guidelines recommend that at least five drugs, including an injectable agent, be utilized for the management of MDR-TB, with a total length of treatment expected to be at least 20 months. 8 Despite the intensity of therapy, treatment failure is woefully common. When treating MDR-TB in a programmatic setting, cure is achieved in only 62% of cases overall, and in only 40% of the subset of patients with XDR-TB. 9
Effective management of drug-resistant TB depends on the availability of effective therapeutics. Experience with MDR-TB has suggested that the use of additional drugs leads to improved outcomes, suggesting that developing a successful therapy may have more to do with increasing the number of effective drugs used than on extending the duration of treatment.10–12 For the effective management of drug-resistant TB, it is therefore imperative that the universe of TB drugs be expanded and that shorter, effective treatment regimens become available. 13
One of the great achievements in TB management over the past 15 years has been the development of new antimycobacterial drugs. These new agents (summarized in Table 1) provide hope that future treatment regimens for drug-resistant TB will be more effective and more efficient than the current programs. Since there is a great need to fold new TB drugs into practice, this review intends to provide an overview of these new agents, including descriptions of their mechanisms of action, evidence for their efficacy and safety, and highlights of their pending investigations.
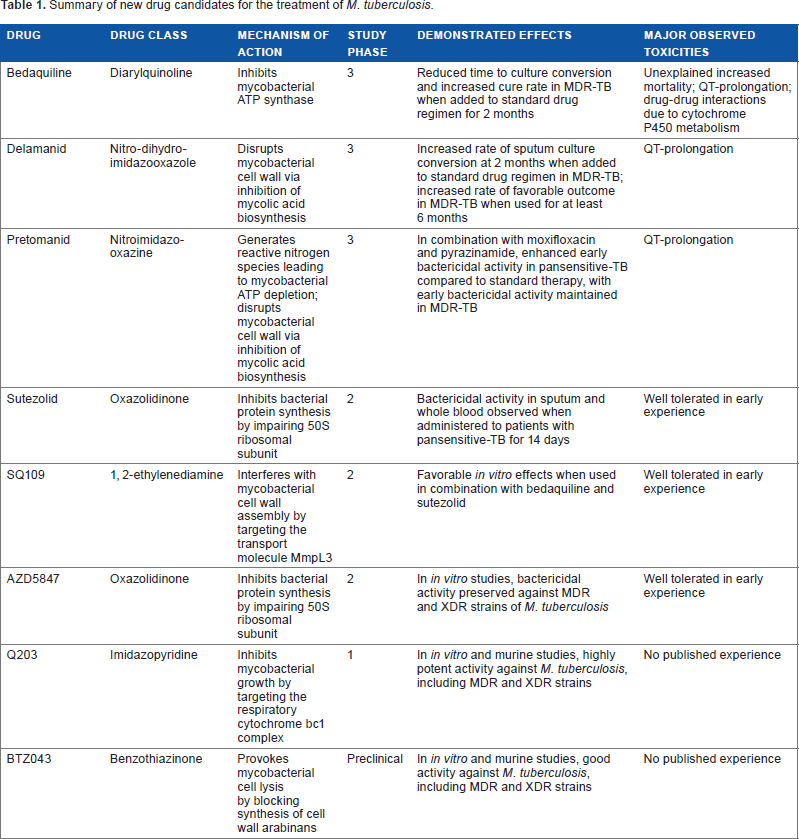
Summary of new drug candidates for the treatment of
Bedaquiline
On December 31, 2012, the United States Food and Drug Administration (FDA) granted accelerated approval of bedaquiline for use in MDR-TB, making bedaquiline the first new TB drug to be approved by a major regulatory agency in over 40 years. 14 In March 2014, the European Medicines Agency (EMA) also provided conditional approval for bedaquiline. 15 Bedaquiline is a diarylquinoline, a new class of antimicrobials that target mycobacterial ATP synthase and exhibit bactericidal activity. 16 The target of bedaquiline's antimycobacterial activity, and that of the other new agents, is represented schematically in Figure 1. The discovery of the anti-TB properties of diarylquinolines was hailed with “considerable excitement and optimism” and hope of a “quantum therapeutic leap” that would radically change TB treatment – potentially reducing the number of drugs and the duration of treatment required for effective therapy. 17 However, clinical experience with bedaquiline, though generally favorable, has tempered this initial enthusiasm.
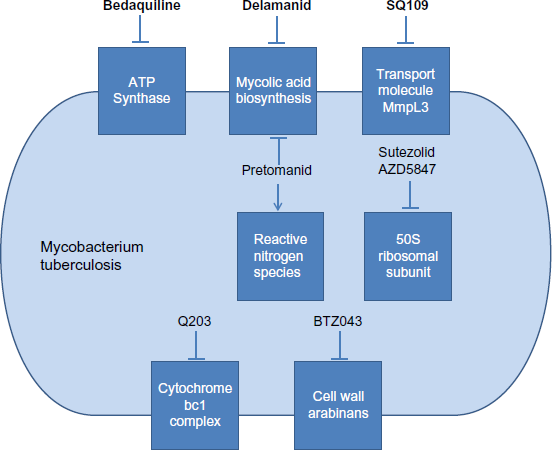
Targets of antimycobacterial activity of novel drugs for the treatment of tuberculosis.
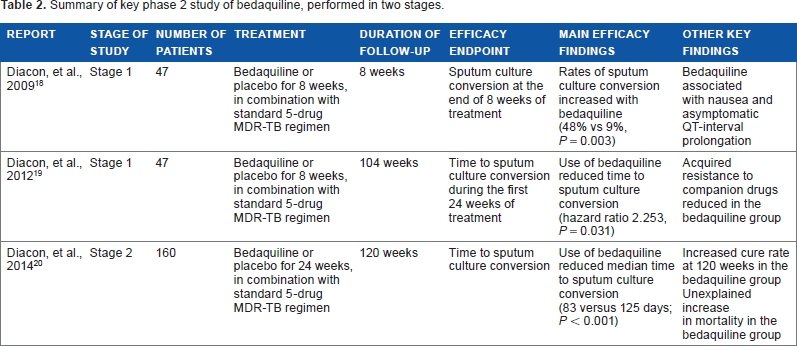
The US and European regulatory agencies provided conditional approval for bedaquiline on the basis of the results from a phase 2 randomized, controlled trial, performed in two stages.18–20 A summary of the study is presented in Table 2. In the first stage of the study, 47 patients with newly diagnosed smear-positive MDR-TB were randomized to receive bedaquiline (which at that time was referred to as TMC207) or placebo, in combination with a standard five-drug MDR-TB treatment regimen, for a period of eight weeks.
18
The primary antimycobacterial efficacy endpoint was sputum culture conversion, which at the end of eight weeks was achieved by 48% of those in the bedaquiline group versus 9% in the placebo group (
Summary of key phase 2 study of bedaquiline, performed in two stages.
Subsequently, the second stage of the phase 2 trial was conducted, investigating a longer duration of bedaquiline therapy in a greater number of patients.
20
In this second stage, 160 patients with newly diagnosed smear-positive MDR-TB were randomized to receive bedaquiline or placebo, in combination with a preferred five-drug MDR-TB treatment regimen, for a period of 24 weeks. The patients were followed-up for 120 weeks. The primary efficacy endpoint was the median time to culture conversion, which was reduced in the bedaquiline group as compared with placebo (83 versus 125 days;
Though the antimycobacterial effects of bedaquiline have been found to be generally favorable to the extent that they have been studied, treatment with bedaquiline has been associated with an increase in sudden unexpected death. 20 Though published experience with bedaquiline remains quite limited, in the phase 2 trial studying 160 patients who received either bedaquiline or placebo for 24 weeks, 13% of patients in the bedaquiline group died versus 2% in the placebo group, with all but one of the deaths occurring after completion of the 24-week exposure to the study drug. 20 In light of this finding, the US FDA's approval of bedaquiline includes a black box warning of an increase in all-cause mortality with the use of the agent. 21 In addition, bedaquiline has been associated with QT interval prolongation (which is also highlighted in an FDA black box warning), hepatic toxicity, nausea, headache, and arthralgia. 21 Potential drug interactions are also a concern, as bedaquiline is metabolized by the cytochrome P450 system. 22
In light of concerns regarding increased mortality associated with its use, the current role of bedaquiline in the treatment of MDR-TB remains rather limited, and primarily driven by necessity due to resistance to other drugs. 21 The duration of bedaquiline therapy should be limited to 24 weeks, dosed at 400 mg daily for the first 2 weeks, followed by 200 mg three times weekly for the remaining 22 weeks – in line with published experience, regulatory agency guidelines, and expert opinion. Additional phase 2 and 3 trials investigating bedaquiline-containing treatment regimens are planned or underway, and with the accumulation of knowledge, it is expected that the role of bedaquiline in the treatment of MDR-TB will change. For now, bedaquiline appears to be a useful new agent for use in MDR-TB, but has not yet lived up to the initial hopes of becoming a game-changing medication in the global fight against TB.
Delamanid
On April 28, 2014, the EMA granted delamanid conditional approval for use in MDR-TB, making delamanid the second (and most recent) novel TB drug to be approved by a major regulatory agency in the past 40 years. 23 To date, delamanid has received regulatory approval in Japan and South Korea, along with the European nations governed by the EMA, but it has not been approved by the US FDA.
Delamanid is derived from the nitro-dihydroimidazooxazole class of compounds. It exerts its antimycobacterial action by inhibiting mycolic acid biosynthesis, which leads to disruption of the mycobacterial cell wall and produces bactericidal activity.24,25 As with bedaquiline, the best current evidence to support the use of delamanid in MDR-TB is limited to phase 2 clinical trials. The first reported evidence on the effectiveness of delamanid derives from a randomized, placebo-controlled, multinational trial of 481 patients with MDR-TB, which remains the largest published study investigating the drug.
26
In this trial, all the patients received a standard MDR-TB treatment regimen and were randomized to receive one of the three study medications: delamanid dosed at 100 mg twice daily, delamanid dosed at 200 mg twice daily, or placebo. The study drug was continued for two months, and the primary efficacy endpoint was the rate of sputum culture conversion at the end of those two months. Those who had been administered delamanid achieved sputum culture conversion at two months at increased rates as compared with those who had been administered placebo, with 45.4% of those having received delamanid 100 mg twice daily and 41.9% of those having received delamanid 200 mg twice daily achieving sputum conversion, compared with 29.6% of those having received placebo (
In the randomized, controlled trial, delamanid was found to be well tolerated, though asymptomatic prolongation of the QT interval was more frequent among patients receiving delamanid as compared with those receiving placebo. 26 In light of the finding of QT interval prolongation, care will need to be used when considering combining delamanid with other QT-prolonging medications, including bedaquiline. A phase 2 trial has been proposed to investigate the safety and tolerability of delamanid and bedaquiline used in combination, with particular attention to the effects on QT interval prolongation. 29 In addition, a phase 3 trial further investigating the safety and efficacy of delamanid for six months in patients with MDR-TB is under way. 30 At this point, delamanid appears to be a promising new agent for the treatment of MDR-TB, but ongoing clinical testing will reveal more about the benefits and risks of the drug, including the preferred dosing and duration of treatment.
Pretomanid
Pretomanid, formerly known as PA-824, is a nitroimidazooxazine that exerts bactericidal activity against mycobacteria by means of two mechanisms as follows: by serving as a nitric oxide donor with consequent intracellular ATP depletion (akin to the effect of cyanide) and by inhibiting cell wall mycolic acid biosynthesis (similar to the effect of isoniazid).31,32 Pretomanid has been found to exert antimycobacterial activity in both aerobic and anaerobic conditions, and in the setting of both replicating and nonreplicating mycobacteria, likely due to these disparate mechanisms of action. 32
Though not yet approved for clinical use by any major regulatory agencies, pretomanid has yielded promising results in early studies. Perhaps the most intriguing results have come from studies investigating pretomanid as part of a novel TB regimen, comprising pretomanid, moxifloxacin, and pyrazinamide (abbreviated as PaMZ). The potential of PaMZ was first noted in a murine model of TB, in which mice treated with PaMZ were cured more rapidly than were mice treated with a standard regimen of isoniazid, rifampin, and pyrazinamide. 33 Subsequently, a prospective, randomized, early bactericidal activity study was performed in patients with treatment-naïve pulmonary TB to investigate a number of novel TB drug combinations and included the PaMZ regimen in one of the treatment arms. 34 The PaMZ regimen was found to have the strongest early bactericidal effect among the novel drug combinations, with its effect being comparable with the standard combination of isoniazid, rifampicin, pyrazinamide, and ethambutol.
A phase 2 trial of bactericidal activity currently provides the most robust experience with the PaMZ regimen, and with pretomanid overall. 35 In this multicenter, open-label, partly randomized clinical trial, 207 patients with drug-susceptible, smear-positive, pulmonary TB were randomized to receive eight weeks of one of the following three regimens: (1) PaMZ with pretomanid dosed at 100 mg daily (Pa100MZ); (2) PaMZ with pretomanid dosed at 200 mg daily (Pa200MZ); or (3) standard combination therapy with isoniazid, rifampicin, pyrazinamide, and ethambutol. Apart from this, 26 patients with MDR-TB were nonrandomly assigned to receive Pa200MZ. Among the patients with drug-susceptible TB, Pa200MZ showed superior bactericidal activity as compared with standard therapy. In addition, significantly more patients who received either of the PaMZ regimens achieved sputum culture negativity in liquid media at eight weeks, as compared with those who received standard therapy. Among the patients with MDR-TB, the Pa200MZ regimen also exhibited effective bactericidal activity, comparable with that achieved in the patients with drug-susceptible TB who received standard therapy. This final finding, suggesting that a novel regimen may prove effective for rapid treatment of MDR-TB, is tantalizing, but requires substantial further investigation.
The experience to date suggests that pretomanid is well tolerated at doses of 100–200 mg daily.35,36 As with bedaquiline and delamanid, pretomanid has been associated with QT interval prolongation. To date, pretomanid remains a promising new agent with the potential to change the approach to drug-resistant TB, particularly when used as a component of novel treatment regimens.35,37
Sutezolid
Sutezolid is a novel oxazolidinone undergoing premarketing evaluation, which – similar to linezolid, itself a first-generation oxazolidinone – exerts bactericidal activity by inhibiting bacterial protein synthesis.
38
Sutezolid has antimycobacterial activity, and in a murine model, it was found to be more potent than linezolid against
In the first study of sutezolid in patients with pulmonary TB, sutezolid was administered to 50 patients with drug-sensitive pulmonary TB at doses of 600 mg twice daily or 1200 mg once daily for 14 days. 43 Both dosing regimens were generally safe and well tolerated, and both resulted in readily detectable bactericidal activity in sputum and blood. The early experience with sutezolid suggests that it does not cause the myelosuppressive effect that can be seen with linezolid, but concern still remains for the potential development of peripheral neuropathy with extended use.42,43
SQ109
SQ109 is a 1,2-ethylenediamine that was derived from the pharmacophore of ethambutol.
44
However, SQ109 has a distinct mechanism of action from ethambutol, and it maintains activity against ethambutol-resistant strains of
AZD5847
AZD5847 is, similar to sutezolid, a novel oxazolidinone currently under investigation, though the experience with AZD5847 is more limited. As with sutezolid and linezolid, the mechanism of AZD5847's antimycobacterial activity appears to be mediated by the impairment of mycobacterial 50S ribosomal subunit, with consequent inhibition of mycobacterial protein synthesis.
50
In
Q203
Q203 is a new imidazopyridine that has shown promise as a candidate drug for TB in preclinical studies. Q203 exerts antimycobacterial activity by targeting the respiratory cytochrome bc1 complex, thereby blocking mycobacterial growth.
53
In initial studies, Q203 demonstrated highly potent anti-TB activity, including
BTZ043
BTZ043 is the most advanced compound in a novel antimycobacterial class of benzothiazinones. The benzothiazinones exert antimycobacterial activity by blocking synthesis of cell wall arabinans, thereby provoking mycobacterial cell lysis.
55
Novel Treatment Regimens
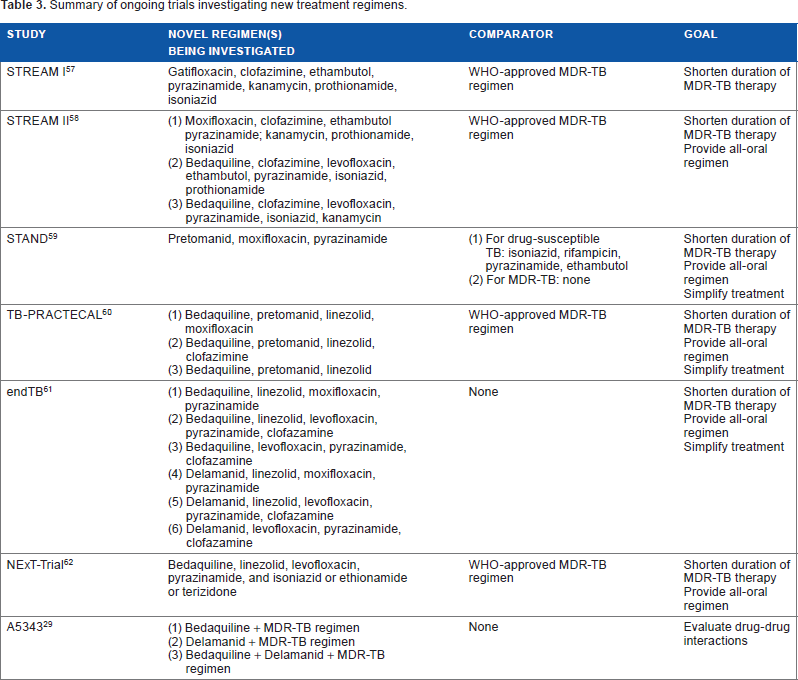
The early experience with the novel agents suggests that it is unlikely that a single new drug will fundamentally change the course of drug-resistant TB. More likely, it will be new combinations of medications, which may include one or more novel agents, which will provide new paradigms in the management of drug-resistant disease. We have already discussed the role of novel treatment regimens in the context of pretomanid, which has been studied most rigorously in the setting of the novel combination treatment PaMZ.33–35 The importance of new combinations of medications has been highlighted by early experience with the
Summary of ongoing trials investigating new treatment regimens.
Summary
In recent decades, drug-resistant TB has emerged as a major challenge, greatly complicating TB management for the individual patient and for health systems. In response to this challenge, several novel TB drugs have been developed. Though we are still in the early stages of this new era of TB drug development, the varied classes and mechanisms of action of the novel agents have generated hope that the great scourge of drug-resistant TB can be overcome. Two of the new agents, bedaquiline and delamanid, have achieved conditional regulatory approval and are starting to be used in a limited way in the treatment of drug-resistant TB. Other agents, including candidate drugs from multiple different classes of compounds, have shown considerable promise at various stages of preclinical and clinical studies. As more drugs become available, it will become possible to generate entirely new treatment regimens for TB, which could radically change the approach to drug-resistant disease. In the future, as knowledge about these new agents accumulates, we may well find tools to meet the challenges of drug-resistant TB.
Author Contributions
Wrote the first draft of the manuscript: JMC. Contributed to the writing of the manuscript: JMC, PE. Agree with manuscript results and conclusions: JMC, PE. Jointly developed the structure and arguments for the paper: JMC, PE. Made critical revisions and approved final version: JMC, PE. Both authors reviewed and approved of the final manuscript.
