Abstract
Among newer cytotoxic agents, pemetrexed gained much interest because of its particular mechanism of action. In fact, pemetrexed is a multi-target agent able to inhibit at least three crucial enzymes involved in the folate pathway: thymidylate synthase, dihydrofolate reductase and glycinamide ribonucleotide formyl transferase. The ability to inhibit multiple enzymes, confers to this drug a clinical advantage by increasing the spectrum of tumors with biochemical profiles potentially sensitive to the drug. Due to the good toxicity profile of pemetrexed, the combination with cisplatin resulted feasible and effective. Pemetrexed is currently approved in combination with cisplatin for first line treatment in patients with unresectable malignant pleural mesothelioma. Pemetrexed is the first agent approved for the treatment of malignant pleural mesothelioma. Generally, pemetrexed is considered a well tolerated drug and the addition of folic acid, vitamin B12, and dexamethasone markedly reduced the incidence of grade 3 and 4 hematologic and non-hematologic toxicities. Pharmacology, pharmacokinetics, efficacy, safety, and current and potential roles of pemetrexed in therapy for malignant pleural mesothelioma were reviewed.
Introduction
In the last few decades, several efforts have been made to improve the outcome of patients affected by human malignancies and the efficacy achievable with chemotherapy seems to have reached a therapeutic plateau. However, among newer cytotoxic agents, pemetrexed (ALIMTA® Eli Lilly, Indianapolis, Indiana) gained much interest because of its particular mechanism of action. Pemetrexed is a multi-target agent that inhibits at least three crucial enzymes involved in the folate pathway: thymidylate synthase (TS), dihydrofolate reductase (DHFR) and glycinamide ribonucleotide formyl transferase (GARFT). This quality increases the spectrum of tumors with biochemical profiles potentially sensitive to the drug. Malignant pleural mesothelioma (MPM) is an aggressive tumor with a dismal prognosis. Most patients are candidates for chemotherapy during the course of their disease. At the moment, the only Food And Drug (FDA)-approved agent for MPM is pemetrexed. Pemetrexed may be more active in mesothelioma than in other cancers because of a high capacity cell membrane transporter in mesothelioma which is highly specific for pemetrexed. 1 In particular, pemetrexed was shown to have activity as a single agent in a phase II trial in patients with MPM 2 and in phase I trials in combination with platinum analogs.3,4 Moreover, the combination of pemetrexed with cisplatin resulted feasible. Pemetrexed is currently approved in combination with cisplatin for first line treatment of MPM. In fact, a large phase III trial testing pemetrexed and cisplatin versus cisplatin alone in 448 chemo-naïve patients with MPM has shown a significant advantage with the combined regimen in overall survival (OS), time to progression, response rate, quality of life and symptom control. 5 For patients unfit to receive a cisplatin-based chemotherapy, pemetrexed alone 2 or combined with carboplatin6–8 has been proposed as an alternative choice of treatment. The goals of this article were to summarize the pharmacology, pharmacokinetics, efficacy, and safety of pemetrexed, and to review its current and potential roles in therapy for MPM.
Mechanism of Action, Metabolism and Pharmacokinetic Profile
Pemetrexed (L-glutamic acid, N-[4-[2-(2- amino-4,7-dihydro-4-oxo-1H-pyrrolo[2,3-d]pyrimidin-5-yl) ethyl]benzoyl]) is a new-generation antifolate (Fig. 1). Unlike the “classic” antimetabolite drugs, such as methotrexate, which selectively target a single enzyme, pemetrexed exerts its action by disrupting several folate-dependent metabolic processes essential for cell replication. In particular, pemetrexed inhibits at least three crucial enzymes involved in the folate pathway: thymidylate synthase (TS), dihydrofolate reductase (DHFR) and glycinamide ribonucleotide formyl transferase (GARFT). Thymidylate synthase and DHFR are enzymes involved in pyrimidine synthesis process, whereas GARFT is a folate-dependent enzyme involved in de novo purine biosyntesis. 9 Purines and pyrimidines are both involved in the DNA synthesis. The ability of pemetrexed to inhibit multiple enzymes, confers to the drug a clinical advantage by increasing the spectrum of tumors with biochemical profiles potentially sensitive to the drug. Among these enzymes, thymidylate synthase is considered the primary target of pemetrexed. In fact, pemetrexed is a weak inhibitor of GARFT and when the TS is inhibited, the tetrahydrofolate oxidation is stopped, making the DHFR activity unnecessary. 10 Pemetrexed enters the cell through the reduced folate carrier (RFC), a bidirectional transporter and the major cellular transport system for folates. In addition to the RFC, pemetrexed is a substrate, with an affinity exceeding that of folic acid, for folate receptor-α, a cellular membrane receptor that mediates cell entry of folates by formation of endocytic vesicles. A low pH transporter also appears involved in pemetrexed internalization. Pharmacologic activity is achieved after conversion of pemetrexed to a polyglutamated form; the pentaglutamated form is the most predominant. The formation of polyglutamated metabolites is essential for intracellular accumulation and retention of anti-metabolites. Pentaglutamated pemetrexed is a potent inhibitor of TS, which catalyzes the transformation of deoxyuridine monophosphate to deoxythymidine monophosphate (dTMP). Inhibition of TS decreases formation of dTMP, a progenitor of the nucleotide deoxythymidine triphosphate (dTTP) needed for DNA synthesis. The enzyme deoxycytidine deaminase is negatively regulated by dTTP. The depletion of dTTP levels by pemetrexed inhibition of TS results in a corresponding decrease in phosphorylation of deoxycytidine monophosphate to the nucleotide deoxycytidine triphosphate. Pentaglutamated pemetrexed is also an inhibitor of GARFT, which results in inhibition of de novo purine synthesis. The DHFR-binding affinity of pemetrexed is 1000 times less than that of methotrexate, and the inhibition of DHFR by pemetrexed is reportedly minimal11,12 (Fig. 2).
Chemical structure of pemetrexed.
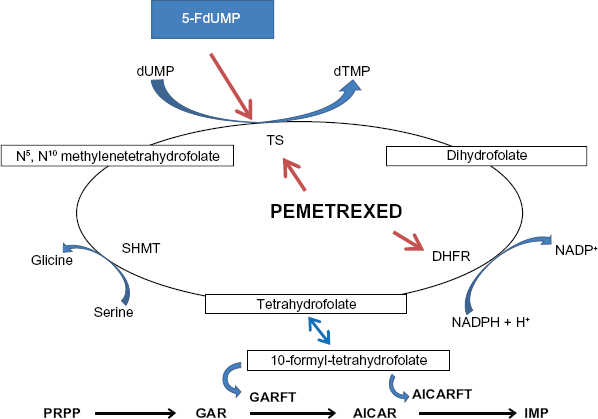
The folate cycle.
The pharmacokinetic properties of pemetrexed after single-agent administration have been assessed in patients with a variety of solid tumors at doses ranging from 0.2 to 838 mg/m2 infused over a 10-minute period. 13 Pemetrexed displayed linear pharmacokinetics, with total systemic exposure (AUC) and Cmax increasing proportionally with dose.13–16 Elimination occurred predominantly through the urine with minimal metabolism; 70% to 90% of the dose was recovered unchanged within the first 24 hours of administration. Pemetrexed has limited tissue distribution, as indicated by a steady-state distribution volume of 16.1 L determined in population pharmacokinetic analysis. 17 This relatively small volume of distribution is attributed to the polarity of the compound. Elimination t1/2 was estimated at 3.5 hours, and total systemic plasma clearance is 91.8 mL/min. 34–37 Cmax occurred at or near termination of the 10-minute infusion and ranged from 67.4 to 251.3 pg/mL at a 600 mg/m2 dose. 15 No dose accumulation or alteration in pharmacokinetic parameters occurred over multiple treatment cycles. Population pharmacokinetic analysis revealed small interpatient variability with coefficients of variation ≤22%. 17 Pemetrexed is -81% bound to plasma proteins. 13 No notable difference in plasma binding was observed for varying degrees of pemetrexed plasma concentration or renal impairment. 13 The effect of pleural or peritoneal effusions, or other third-space fluids, on pemetrexed pharmacokinetics has not been reported in the literature. In patients with clinically significant third-space fluid, consideration should be given to draining the effusion before administration of pemetrexed. 13 Plasma clearance of pemetrexed was noted to decrease with decreasing renal function, as expected for a drug that is eliminated primarily by urinary excretion.14–16 The use of pemetrexed in patients with impaired renal function was compared in 34 patients with various solid tumors. 18 Patients were stratified according to glomerular filtration rate (GFR) as measured by technetium Tc 99 m-DPTA clearance and grouped into the following cohorts: >80 mL/min, 60 to 79 mL/min, 40 to 59 mL/min, 30 to 39 mL/min, 20 to 29 mL/min, and <20 mL/min. Pemetrexed doses ranging from 150 to 600 mg/m2 based on renal function were administered as a 10-minute infusion every 3 weeks. Pemetrexed plasma clearance was proportional to renal GFR clearance (r 2 = 0.63). Pemetrexed 500 mg/m2 was well tolerated in patients in the cohort with GFR 60 to 79 mL/min and GFR 40 to 59 mL/min, with no dose-limiting myelosuppression. Only 1 patient with GFR <20 mL/min was treated at a dose of 150 mg/m2 and died of subsequent febrile neutropenia. Pemetrexed undergoes limited hepatic metabolism. No relationships between aspartate aminotransferase (AST), alanine aminotransferase (ALT), or total bilirubin and alteration of pemetrexed pharmacokinetics were identified in a population pharmacokinetic.
The combination of pemetrexed with cisplatin was found to be feasible and effective. The pharmacokinetics of total platinum and pemetrexed were evaluated in patients with MPM using population pharmacokinetic methods and there was no significant influence of concomitant cisplatin administration on pemetrexed clearance or of concomitant pemetrexed administration on cisplatin clearance. Moreover, the pharmacokinetics of free platinum derived from cisplatin were not altered by co-administration with pemetrexed, and in agreement with this, no unexpected cisplatin-induced toxicities were observed when these drugs were combined.
Pemetrexed is currently approved in combination with cisplatin for first line treatment of MPM, 5 as a single agent for second line treatment of advanced NSCLC 19 and has recently gained approval in Europe and in United States for first line therapy in combination with cisplatin for non squamous NSCLC patients.
Clinical Studies
First line treatment
The only FDA-approved agent for MPM is pemetrexed. In fact, pemetrexed was shown to have activity as a single agent in a phase II trial in patients with MPM 2 and in phase I trials in combination with platinum analogs3,4 (Table 1).
First-line pemetrexed-based chemotherapy in MPM patients.
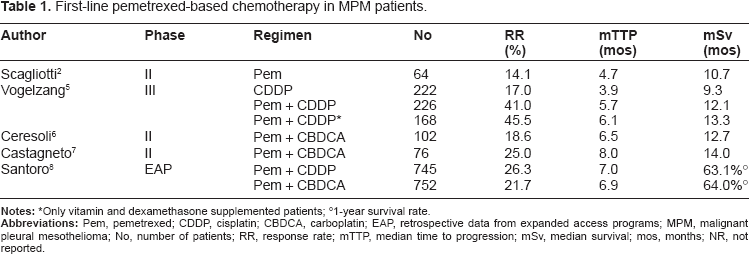
Only vitamin and dexamethasone supplemented patients;
1-year survival rate.
The phase II clinical trial evaluated the efficacy of pemetrexed (500 mg/mq) for the treatment of 64 MPM patients with a histological proven diagnosis, chemotherapy-naive measurable lesions, and adequate organ function. Most patients (43/64) also received folic acid and vitamin B12 supplementation to improve safety. As single agent, pemetrexed resulted in a moderate RR (14.1%), with a median TTP of 4.7 months and a median OS of 10.7 months. 2 Seven of the nine responders were vitamin supplemented. The median OS was 13.0 months for supplemented patients and 8.0 months for non-supplemented patients. Vitamin-supplemented patients completed more cycles of therapy than non-supplemented patients (median, six versus two cycles, respectively). Grade 3/4 neutropenia (23.4%) and grade 3/4 leukopenia (18.8%) were the most common laboratory toxicities. Fatigue and febrile neutropenia were the most commonly reported non-laboratory events (grade 3, 6.3%; grade 4, 0.0% each) and the incidence of these toxicities was generally lower in the vitamin-supplemented patients.
In a phase I trial designed to determine the maximum-tolerated dose (MTD), the dose-limiting toxicities (DLT), and the pharmacokinetics of pemetrexed combined with cisplatin, two patients had objective remissions of disease (one mesothelioma patient, one colon cancer patient). 3 The MTD was pemetrexed 600 mg/m2 and cisplatin 75 mg/m2. DLTs were neutropenic sepsis, diarrhea, and skin toxicity. In an other phase I trial the combination of pemetrexed plus carboplatin was found active and well tolerated in MPM patients, although no vitamin supplementation was administered. 4 The recommended dose of the combination for phase II studies was pemetrexed 500 mg/m2 and carboplatin area under the plasma concentration-time curve (AUC) 5 mg/mL/min.
The use of pemetrexed in MPM patients was approved by the Food and Drug Administration (FDA) based on a single-blind, placebo-controlled, phase III study. In this trial, chemotherapy-naive patients who were not eligible for curative surgery were randomized to pemetrexed 500 mg/m2 and cisplatin 75 mg/m2, or placebo and cisplatin 75 mg/m2. 5 Both regimens were given intravenously every 21 days. A total of 456 patients were assigned: 226 received pemetrexed and cisplatin, 222 received cisplatin alone, and eight never received therapy. The RR for the combination was significantly greater than for single-agent cisplatin (41% vs. 17%; P < 0.001). Pemetrexed/cisplatin treated patients had a median OS of 12.1 months, compared with 9.3 months for patients treated with cisplatin alone (P = 0.020). The hazard ratio for death of patients in the pemetrexed/cisplatin arm versus those in the control arm was 0.77. Time to progression was also superior for patients treated with the combined chemotherapy (5.7 vs. 3.9 months, P = 0.001). In addition, treatment with this combination resulted in a significant improvement in pulmonary function, quality of life, and symptoms such as pain and dyspnea. After the first 117 patients enrolled in this study, all patients were supplemented with dietary doses of folate and vitamin B12. Vitamin supplementation improved RR (45.5% vs. 19.6%; P < 0.001) and survival (TTP 6.1 vs. 3.9 months, P = 0.008; OS 13.3 vs. 10 months, P = 0.051) in both treatment arms, and reduced the incidence of serious toxicity. In preclinical models, there is a very significant decrease in pemetrexed activity as the extracellular folate level increases above the physiologic range. This suggests that it may be appropriate to limit folate supplementation to no more than 400 μg, the amount found in a multivitamin, rather than the 1000 μg that is more frequently prescribed. 20
Malignant pleural mesothelioma is a disease of the older patient, with a median age of onset of 74 years. 21 The typical non-haematological toxicity profile of cisplatin (gastro-intestinal, neurologic, and renal) is questionable in the context of a palliative treatment, especially for poor performance and elderly patients. Carboplatin has the potential advantages of having a better adverse effect profile and better ease of administration. In a phase I study in 25 patients with MPM, the combination of pemetrexed and carboplatin was active and well tolerated, with a reported RR of 32%. 4 Starting from these data, some combined schedules containing carboplatin, instead of cisplatin, were tested in MPM patients in an attempt to reduce toxicity maintaining the same survival outcomes.6,7
In a phase II trial of 102 MPM patients treated with pemetrexed plus carboplatin a similar time to progression (6.5 months) and overall survival (12.7 months) were observed as in the phase III trial of pemetrexed-cisplatin. 6 The toxicity profile seemed to be better in the pemetrexed-carboplatin trial than in the pemetrexed-cisplatin trial, especially considering the non-hematological toxicity. A 76-patient phase II study reported a time to progression of 8.0 months, a median survival of 14 months, and a response rate of 25% using the same regimen. 7 Moreover, no significant difference was observed in terms of overall disease control (60.4 vs. 66.9%, P = 0.47), TTP (7.2 vs. 7.5 months, P = 0.42) and survival (10.7 vs. 13.9 months, P = 0.12) between elderly patients compared with younger individuals in a retrospective analysis of pooled data from the two phase II trials of pemetrexed and carboplatin as first-line therapy. 22
Data from the International Expanded Access Program (EAP) suggested an activity of both pemetrexed plus cisplatin and pemetrexed plus carboplatin in 1704 chemonaïve MPM patients not amenable to curative surgery, showing similar time to progressive disease and 1-year survival rates. In particular, the pemetrexed plus cisplatin group obtained a RR of 26.3% compared with 21.7% for the pemetrexed plus carboplatin group, with 1-year survival rates of 63.1% versus 64.0% and median TTP disease of 7 months versus 6.9 months. 8
There are several unresolved questions regarding timing and duration of pemetrexed treatment. Some epithelial mesothelioma patients may have prolonged stable disease for months or even years without chemotherapy. It is not known whether these patients should be treated at diagnosis, at symptom progression, or at radiographic progression. In a very small pilot study from the Royal Marsden Hospital, there was a trend toward a longer time to symptomatic progression and overall survival in those patients who received chemotherapy at diagnosis rather than at symptom progression; 23 however, these results need to be validated in a larger study with a more active chemotherapy regimen than was employed in that study. We also do not know the optimum length of treatment. Most patients receive between 4 and 8 cycles of pemetrexed with cis- or carboplatin, few can tolerate more. Should they stop treatment at that point, or continue with single-agent pemetrexed? A small, non-randomized Dutch feasibility study of pemetrexed maintenance demonstrated that maintenance is well tolerated, and that responses can occur after six cycles of treatment. 24 The CALGB is currently designing a larger, randomized study to more definitively address this question.
Second line treatment
In the last years, pemetrexed has been extensively explored as second-line therapy or beyond in MPM patients not previously exposed to this agent (Table 2). Sørensen et al reported the results of a study in which data sets of treatment of two different cohorts were combined. 25 Thirty-nine patients previously treated with platinum-based regimens without pemetrexed were included. Twenty-eight patients received pemetrexed alone (in 3 cases as third-line treatment), whereas 11 patients received pemetrexed plus carboplatin. Treatment was generally well tolerated. PR rates were 21% and 18%, median TTP was 21 weeks and 32 weeks, and median survival was 42 weeks and 39 weeks with pemetrexed and pemetrexed/carboplatin, respectively. Jänne et al 26 reported the results of the use of pemetrexed alone or in combination with cisplatin within an Expanded Access Program in 187 patients who had received previous systemic chemotherapy. Patients were treated with pemetrexed alone (n = 91) or in combination with cisplatin (n = 96). Gemcitabine was the most common prior therapy used, followed by cisplatin, carboplatin, paclitaxel and vinorelbine. Response data were available for 153 patients. The overall response rate was 32.5% for pemetrexed/cisplatin and 5.5% for pemetrexed alone; stable disease was achieved in 36.3% and 41.1% of patients, respectively. Median OS was 7.6 months in patients receiving combination therapy, and 4.1 months in those receiving single-agent pemetrexed. However, due to the limitations of the study design, no comparison can be done between treatment groups. In fact, patients receiving combination chemotherapy were younger and fitter at baseline, and had an higher response rate to first-line treatment. This is reflected by the increased number of treatment cycles administered to the combination therapy group. In another analysis from the Expanded Access Program database, the safety and efficacy data of MPM patients who were treated with single-agent pemetrexed were reported. 27 Of a total of 812 patients, 643 were evaluable for efficacy. The overall response rate for the pretreated patients (n = 396) was 12.1%; median TTP was 4.9 months, and the median OS was not estimable due to high censoring. Hematological toxicity was mild in both groups, with neutropenia (<18%) as the main side effect.
Second-line chemotherapy in pemetrexed-naïve MPM patients.
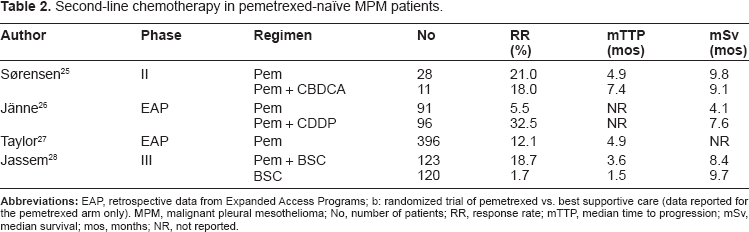
The use of pemetrexed in this setting was further explored prospectively in a randomized, multi-center phase III study examining single-agent pemetrexed as second-line chemotherapy versus best supportive care (BSC). 28 Primary endpoint of the study was OS. Secondary end points included response rate, TTP, progression-free survival, time to treatment failure, and toxicity. Patients with relapsed MPM after first-line chemotherapy were randomly assigned to receive pemetrexed 500 mg/mq plus BSC every 21 days or BSC alone. The study enrolled 243 patients. PR was achieved in 18.7% of patients receiving pemetrexed, and a disease control (PR + SD) was achieved in 59.3% and 19.2% of patients in pemetrexed and BSC arms, respectively (P < 0.0001). Median TTP was significantly improved in pemetrexed arm (3.8 vs. 1.5 months), as well as the other time-to-event measures. Chemotherapy was well tolerated, with expected mild (4% to 7%) grade 3 and 4 hematological toxicities. Use of post-discontinuation chemotherapy was significantly greater and earlier among BSC patients. Median OS time was not significantly different between the arms (8.4 months for patients treated with pemetrexed vs. 9.7 months for those receiving BSC only), possibly because of the significant imbalance in post-study chemotherapy (PSC). A trend towards a survival benefit was observed for patients who had responded to first-line therapy. 28
In conclusion, in pemetrexed-naïve patients, data from a randomized trial versus best supportive care suggest the use of single agent pemetrexed as a standard second-line treatment. This evidence is supported also by the results of the Expanded Access Programs. In the growing population of pemetrexed-pretreated patients, there is no standard approach. In selected cases with a prolonged response to first-line pemetrexed-based chemotherapy, re-treatment with a pemetrexed-based regimen should be considered. When a trial is not available or patients are not eligible for an experimental approach, single agents vinorelbine or gemcitabine seem to be a reasonable option for palliation. However, second-line therapy in MPM remains an ideal field in which to test new chemotherapy agents as well as new therapeutic strategies.
Retreatment with pemetrexed–based chemotherapy
Nearly all MPM patients progress during or after first-line treatment. Second-line chemotherapy is being increasingly used in clinical practice, because patients frequently still have a good performance status at the time of disease progression. 29 In pemetrexed-naïve MPM patients, data from a large randomized trial versus best supportive care and from the Expanded Access Programs support the use of single agent pemetrexed as a standard second-line treatment.27,28
In pemetrexed-pretreated MPM patients, there is no approved drug or drug combination for second-line therapy, and this remains an ideal field in which to test new agents. A number of phase II trials have exploited different chemotherapeutic and targeted agents, but results have been generally disappointing. Several phase II and phase III studies are ongoing. 30
In selected cases with a prolonged response to first-line pemetrexed-containing regimens, small case series have shown a clinical benefit of re-treatment with a pemetrexed-based chemotherapy (PBC).31–34
In our institution, 32 patients (22 males and 10 females) received re-treatment with PBC as second line (19 patients) or beyond second line therapy (13 patients). 35 Sixteen patients were re-treated with pemetrexed alone, and 16 with a pemetrexed/platinum combination. An objective response was achieved in 6 patients (one CR and 5 PRs), for a response rate of 19%. Ten patients (31%) had SD after treatment. Overall, the disease control rate was 50%. Median PFS and OS after re-treatment with PBC were 3.9 months and 10.3 months, respectively. PFS and OS after re-treatment with PBC were correlated with PFS achieved after first-line PBC. Patients with a first-line PFS > 12 months had a median PFS after re-treatment of 5.6 months, while patients with a FL-PFS ≤ 12 months had a median PFS after re-treatment of 2.5 months; no patients in this group was progression-free at 1 year. Toxicity was mild, with grade 3 or 4 hematological toxicity occurring in 9.3% of patients. In conclusion, re-treatment with PBC should be considered as second-line therapy in MPM patients achieving a durable (>12 months) disease control with first-line PBC. However, further evaluation of this therapeutic option is warranted.
Safety
Generally, pemetrexed is considered a well tolerated drug, also in MPM pateints (Table 3). In particular, during the early Phase I and II trials of pemetrexed, myelosuppression presented as a considerable toxicity, experienced as grade 3/4 in >50% of patients. In studies by Paz-Ares et a1 36 and Cripps et al 37 the selected Phase II pemetrexed dose was reduced from 600 to 500 mg/m2 due to considerable grade 3/4 neutropenia (absolute neutrophil count [ANC] <1.0 x 10 9 /L), grade 3/4 thrombocytopenia (platelets <50.000 x 10 9 /L), febrile neutropenia, mucositis, and septicemia. Drug related mortalities due to myelosuppression, sepsis, and diarrhea were seen in the gastric, bladder, head and neck, and colorectal Phase II studies; mortality rate approached 4%.36,38,39 Homocysteine and methylmalonic acid (MMA) are biological markers for folate status. Earlier research has established the reciprocal relationship between homocysteine levels and serum folate and red blood cell folate levels.40,41 Methionine synthase, the enzyme which metabolizes homocysteine to methionine, requires the folate 5-methyl tetrahydrofolate (THF) to serve as a methyl group donator and vitamin B12 as a cofactor. When folate levels, and hence 5-methyl THF, and vitamin B12 levels are low, conversion of homocysteine to methionine decreases, resulting in increased concentrations of homocysteine. Thus, homocysteine levels are a marker for overall folate status. Vitamin B12 is also a requisite cofactor for methylmalonyl coenzyme A mutase, and levels of MMA rise from decreased enzymatic activity resulting from vitamin B12 deficiency.40,41 A multivariate analysis conducted on patients enrolled in studies to identify predictive factors for severe toxicity with pemetrexed found that increased homocysteine and MMA levels were highly correlated with toxicity. 42 Homocysteine correlated significantly with grade 4 neutropenia (P = 0.003), grade 4 neutropenia and grade 3/4 infection (P < 0.001), grade 4 thrombocytopenia (P < 0.001), and grade 4 neutropenia and grade 3/4 diarrhea (P < 0.001). MMA was significantly correlated with grade 3/4 diarrhea (P < 0.001) and grade 3/4 mucositis (P < 0.001). Prevalence of severe toxicities also increased with elevated pre-treatment levels of homocysteine and MMA. Identification of the correlation between poor folate status and increased pemetrexed toxicity in a multivariate analysis led to the requirement of folic acid and vitamin B12 supplementation for patients in all pemetrexed studies. 42 The standard dose for oral folic acid is 300–1000 μg daily, while for vitamin B12 injection is 1000 μg every 9 weeks, beginning at least 1 week before pemetrexed administration and continuing until 3 weeks after the last administration.
CTC (Common toxicity criteria–-version 2) Grade 3 and 4 toxicities by supplementation status (arranged from Scagliotti GV, J Clin Oncol; 2003:1556–1561 [2]).
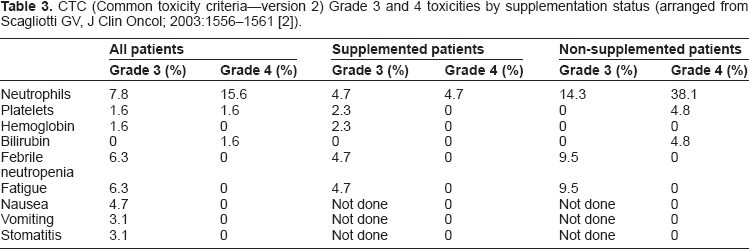
The addition of folic acid and vitamin B12 markedly reduced the incidence of grade 3 and 4 hematologic and non-hematologic toxicities. Grade 3/4 neutropenia was reported in 8% of supplemented patients receiving pemetrexed 500 mg/m2 compared with 46% of non-supplemented patients at the same dose and 54% of non-supplemented patients who received pemetrexed 600 mg/m2. Only 2.6% (7/265) of patients receiving pemetrexed with vitamin supplementation in the Phase III trial of NSCLC required use of G-CSF during treatment of neutropenia or as prophylaxis for subsequent cycles of pemetrexed after a neutropenic episode. 43 Of the total 196 patients enrolled in studies by Miller et al 44 Martin et al 45 John et al 38 and Thodtmann et al 46 and who received pemetrexed 600 mg/m2, 47 (24%) patients required red blood cell transfusions. Development of a pruritic skin rash (all grades) occurred in 60% to 85% of patients receiving pemetrexed 500 to 600 mg/m2 every 21 days and was often described as painful. Prophylactic use of oral dexamethasone (4 mg orally twice daily) given the day before, the day of, and the day after pemetrexed infusion reduced both the incidence and severity of the rash. Pemetrexed has been associated with grade 3/4 elevations (>5 times the upper limit of normal [ULN]) in ALT, AST, alkaline phosphatase, and bilirubin. In all trials, these elevations were reported as transient, and they resolved or improved before the next pemetrexed dose; in some cases, interruption of therapy was required. No cases of liver failure or irreversible liver disease attributed to pemetrexed have been reported in clinical trials. In the MPM Phase III trial of combination pemetrexed/cisplatin versus cisplatin alone, nausea, vomiting, and fatigue were the most commonly reported nonhematologic adverse events in both groups. 5 Significantly more patients in the pemetrexed/cisplatin combination group experienced hematologic toxicities: anemia (4.8% vs. 0%) and thrombocytopenia (5.8% vs. 0%), with grade 3/4 neutropenia (27.9% vs. 2.3%) and grade 3/4 leukopenia (17.7% vs. 0.9%) the most common (all, P < 0.001). A significantly higher incidence of non-hematologic toxicities occurred in the pemetrexed/cisplatin group compared with the single agent cisplatin group: nausea, 14.6% vs. 6.3%; vomiting, 13.3% vs. 3.6%; diarrhea, 4.4% vs. 0%; dehydration, 4.0% vs. 0.5%; and stomatitis, 4.0% vs. 0% (all, P < 0.05). Supplementation with folic acid and vitamin B12 reduced the incidence of toxicities, except diarrhea, in the pemetrexed/cisplatin combination, with statistically significant differences in nausea (11.9% vs. 31.3%; P = 0.012), vomiting (10.3% vs. 31.3%; P = 0.003), and febrile neutropenia (0.5% vs. 9.4%; P = 0.009) between patients who received supplementation throughout the entire treatment course or partially throughout compared with those who did not receive any supplementation. However, a higher incidence of hypertension (11% vs. 3%), chest pain (8% vs. 6%), and thrombosis/embolism (6% vs. 3%) was reported in the fully supplemented patients compared with non-supplemented patients. Age also appeared to influence pemetrexed safety; the manufacturer reported a higher incidence of grade 3/4 neutropenia, fatigue, leukopenia, and thrombocytopenia in patients aged >65 years, despite full supplementation. 13 Rash was also reported to occur more frequently in men, but no other gender or race effects were identified. 13
Future Directions
The combination of cisplatin/carboplatin and pemetrexed represents the standard of care in the first-line treatment of MPM. However, more than one third of patients do not respond to this schedule, receiving useless toxicity. Considering the toxicity profile of this platinum compound containing schedule and the poor performance status of several MPM patients, due to the advanced years and the usual advanced extension of disease at the diagnosis, it represents not only an important medical problem but also an useless expenditure of economical and human resources. Unfortunately, today there are no many data about pemetrexed and/or cisplatin/carboplatin predictors of response in MPM patients. 47 The identification of molecular predictors of effective therapy that are easily detectable in the clinical routine is crucial for maximizing therapeutic efficacy and minimizing useless treatment in patients with cancer. It is well known that all drug treatments have differential effects on patients. Moreover, it is now recognized that the way a patient responds to chemotherapy is a complex trait, influenced by the tumor characteristics and individual genetic constitution. The use of different combinations of predictive markers in order to define optimal treatment regimens represents an intriguing modern challenge. This approach could also allow identification of patients that are unlikely to respond; this information would allow physicians to avoid unnecessary toxicity and hospitalization and preserve economical and human resources.
Recently, Righi et al 48 reported data from an observational study where they retrospectively investigated the correlation between baseline expression of TS and ERCC1, in patients with MPM treated with pemetrexed-based chemotherapy. They found that low TS protein expression, but not TS gene expression, was significantly correlated with a longer TTP and a longer median OS. Conversely, they did not observed a significant correlation between TS expression and outcome in patients who were not treated with pemetrexed. Moreover, in platinum-treated patients they did not find a relationship between survival and ERCC1 levels according to the median-H-score, whereas patients with protein expression in the higher tertile and mRNA expression above median had significantly longer OS. An other retrospective analysis correlated the immunohistochemical expression of excision repair cross-complementing group 1 (ERCC1) and TS with the outcomes of 72 MPM patients treated with carboplatin/pemetrexed in first line setting. 49 Interestingly, the higher TS expression was associated with progressive disease (PD) (odds ratio 1.02; P = 0.061). In particular, the odds ratio of PD for patients with a TS expression ≥20% was 11.7 (P = 0.003), with a risk to progress of approximately 2 times higher than patients with a TS expression <20% (HR 1.90; P = 0.014). This trend was confirmed also for overall survival (HR 1.77; P = 0.044). On the basis of these results, TS expression may be considered as a potential predictor of response to the pemetrexed treatment in MPM patients. However, considering its significant correlation with response, PFS, and OS in a retrospective analysis, adequate prospective studies are needed to confirm its possible predictive and/or prognostic role.
Conclusions
Pemetrexed is a multitargeted antifolate that has demonstrated antitumor activity, as a single agent and in combination with other chemotherapeutic agents, in various tumor types, especially MPM and NSCLC. Myelo-suppression was the predominant dose-limiting toxicity of pemetrexed. However, the addition of folic acid and vitamin B12 markedly improved its safety, also combined with cisplatin. The combination of pemetrexed with cisplatin resulted feasible and effective, without pharmacokinetics interferences. Actually, pemetrexed/cisplatin combination is currently approved for first line treatment of MPM. However, the results are still modest, with an extension of a median survivals by only 3 months and with a median survival of approximately one year. An improvement of the knowledge of the molecular alterations that are specific for MPM will allow the discovery of biomarkers as a useful predictive or prognostic tool and the development and testing of novel targeted agents in this disease in the future.50,51 The TS expression levels seem to predict sensitivity to pemetrexed and the identification of predictors of response will be an interesting field of study to better define the cancer patient population that really benefits from this combination.
Disclosure
This manuscript has been read and approved by all authors. This paper is unique and is not under consideration by any other publication and has not been published elsewhere. The authors and peer reviewers of this paper report no conflicts of interest. The authors confirm that they have permission to reproduce any copyrighted material.
