Abstract
Context
Fibromyalgia syndrome (FMS) is a frequent medical condition characterized by chronic widespread pain and reduced pain threshold. Associated symptoms include fatigue, non restorative sleep, and psychological distress. As usual in medicine, even if the pathogenesis is unclear, some treatments are useful to help patients.
Objectives
Tricyclic antidepressants were the first drugs used to treat FMS. More recently, among serotonin-norepinephrine reuptake inhibitors, duloxetine was approved by US Food and Drug Administration to treat FMS. Duloxetine is used for the management of major depressive disorder, neuropathic pain, generalized anxiety disorder, and stress incontinence. In the pharmacotherapy of fibromyalgia, a focus is presented on the drug duloxetine.
Results
Mechanism of action, metabolism and pharmacokinetic profile are presented. Clinical studies of Duloxetine showed an acceptable efficacy for this chronic condition: Number Need to Treat (NTT) of 4.7 to 9.9, through two 3-month placebo-controlled trials and two 6-month trials. Evaluation criteria are discussed. Safety of this medication has been found to be satisfactory, with nausea as the most common adverse event, in almost 20% of cases.
Conclusion
Treatment algorithm for duloxetine is presented inside FMS treatment strategy. With duloxetine, it is important to start low and increase slowly to prevent or minimize adverse events: 30 mg/day up to 60 mg/day in the second week and if necessary up to 90-120 mg/day. It is possible to treat for 3 to 6 months, possibly up to 12 months. The drug could be decreased 2 to 4 weeks before stopping, with regular assessments during this time. International recommendations insist on multimodal treatments: drug and non drug. Also effective for anxiety and depression, duloxetine ranks among the first place drugs for FMS.
Introduction
Fibromyalgia is a frequent medical condition characterized by chronic widespread pain and a reduced pain threshold with hyperalgesia and allodynia. 1 Associated symptoms include fatigue, increased fatigability, pain due to a daily activity, sleep disturbance, stiffness, cognitive disorders, depression, anxiety, headache, bowel dysfunction, jaw pain, and sensitivity to cold. 23 The result may be disability and a decreased quality of life. The burden of Fibromyalgia Syndrome (FMS) needs effective treatment options with associated pharmacological and non-pharmacological treatments. In FMS these treatments are chosen to target FMS symptoms. Hypothetical and demonstrated pathogenesis may be useful for treatment options.
Main potential pathogenic mechanisms which have been proposed are:
Central sensitization, common with other dysfunctional disorders: Chronic Fatigue Syndrome (CFS), Irritable Bowel Syndrome (IBS), Tension Headache (TH), 4
Sleep disorders 5
Neuroendocrine disorders 6
Learning process (found in conditioning, cognitive errors, coping), common with chronic disorders: pain, fatigue, anxiety, depression 7
Somatization (found in chronic symptoms or syndromes, but also in somatoform disorders)8,9
FMS usually resulting from a chronic local pain may be associated with numerous co-morbidities: Rheumatoid arthritis, Goujerot Sjögren syndrome, hypothyroidism, depression, and anxiety. FMS associated with these co-morbidities decreases quality of life. 3
Management of FMS leads to numerous questions and attempts to establish evidence based or practice based guidelines for treatment options. Inside multimodal and multidisciplinary treatments, it is essential to consider drug therapies.
Tricyclic antidepressants (TCAs) have been the first daily used drugs to treat fibromyalgia. Main trials conducted on amitriptyline between 1986 and 2001 showed efficacy of amitriptyline in FMS, 10 however the number of patients was low and duration of treatment short. Anticholinergic activity limits amitriptyline use. 2
Other drugs have been proposed to treat this medical condition. Tramadol, (a central-acting analgesic that inhibits the reuptake of serotonin and norepinephrine, while having an agonist action on the mu opioid receptors), associated with acetaminophen, demonstrated better pain control and improvement in quality of life compared with a placebo. 11 Among antiepileptic drugs used as analgesics, pregabalin has been the first drug approved in USA by the Food and Drug Administration (FDA) to treat fibromyalgia. 12 Among antidepressants, the efficacy of selective serotonin reuptake inhibitions (SSRIs) is controversial for fluoxetine, citalopram, sertraline or paroxetine. 13 Little data exists on norepinephrine reuptake inhibitors such as reboxetine. 2 Among antidepressants, non-selective serotonin and norepinephrine reuptake inhibitors (SNRIs) have been shown to have a potential benefit in the treatment of fibromyalgia with fewer side effects than tricyclic antidepressants. Venlafaxine and milnacipran have been the two first SNRIs shown to have a potential in the clinical setting but only milnacripran has been developed.14,15 Duloxetine after pregabalin is the second drug approved by FDA, (in June 2008,) for the treatment of fibromyalgia. Since early 2009, milnacipran is the third drug approved in the USA. In Europe, the European League Against Rheumatism (EULAR) has included duloxetine among its evidence-based recommendations for the treatment of fibromyalgia, 16 but the drug has not yet been approved in Europe by the European Medical Agency for this clinical indication.
Duloxetine was first approved in USA and in Europe for the management of, major depressive disorder (MDD) and for diabetic neuropathic pain. Other approved indications of duloxetine are, generalized anxiety disorder (GAD) in USA and in Europe for treatment of stress incontinence in women.
Mechanism of Action, Metabolism and Pharmacokinetic Profile
Duloxetine is a serotonin-norepinephrine reuptake inhibitor as demonstrated by preclinical data. 17 Clinical evidence also largely substantiates this dual neurotransmitter reuptake in humans. 18 Clinical assessment of serotonin reuptake inhibition has been provided indirectly by measuring whole blood serotonin levels which are decreased after duloxetine treatment proportionally to the dose used in depressed patients 19 and in healthy subjects. 20 A direct evidence of serotonin transporter occupancy has been reported in healthy subjects by means of positron emission tomography (TEP) showing more than 80% occupancy of the transporter with doses of 40 and 60 mg of duloxetine. 21
Clinical assessment of norepinephrine reuptake inhibition may be performed by measuring urinary excretion of the catecholamine and its metabolites, which is decreased in healthy subjects. 20 Urinary 3-methoxy-4-hydroxyphenylglycol (MHPG) a biochemical marker of central nervous system norepinephrine breakdown is reduced by duloxetine. The effect of norepinephrine reuptake may also be confirmed by using the tyramine pressor test 22 but discordant results have been reported.19,20
What is known about metabolism and pharmacokinetics?17,23,24 Duloxetine is well absorbed (extent >70%) when orally administered (mean oral bioavailibity of 50%). The time to peak concentration is approximately 6 hours in fasted subjects, with the available form of duloxetine which consists of capsules containing enteric-coated pellets. Duloxetine is highly bound to plasma proteins (>95%) as α1-acide glycoprotein, which may contribute to the inter-individual pharmacokinetic variability. The elimination half-life of duloxetine is 12 hours (8 to 17 hours). Steady-state is about 3 days. The drug elimination is predominantly linked to biotransformation into inactive metabolites via glucuronide and sulfate conjugation and via oxidative metabolism involving CYP1A2 and to a lesser degree CYP2A. Seventy percent appears in the urine as metabolites and 20% is excreted in the feces. Hepatic function impairment may result in a decrease in the formation of metabolites and an increase in plasma concentrations of duloxetine. Renal impairment is not associated with changes in duloxetine concentrations, but only with an accumulation of the metabolites without pharmacological activity.
Some drug interactions with duloxetine have been reported. Fluvoxamine, a strong CYP1A2 inhibitor increases duloxetine peak concentrations by 140% and overall exposure by 460%. 25 Duloxetine is therefore contra-indicated in patients treated with potent CYP 1A2 inhibitors such as fluvoxamine as well as by some fluoroquinolones such as ciprofloxacin or enoxacine. Weaker duloxetine concentration increases may also be observed with CYP 2D6 inhibitors such as paroxetine. 24 Duloxetine does not appear to have a significant effect on the metabolism of drugs that are substrates of CYP1A2 such as theophylline. 25
In smokers, significantly lower concentrations of duloxetine serum levels than those expected in the general population, are found. 26 This may be due to an induction of CYP 1A2 by polycyclic hydrocarbons contained in the tobacco smoke.
Breast feeding should be avoided in women treated by duloxetine. Indeed, in a study in postpartum lactating women who took the drug for 3 days, duloxetine was detected in breast milk with steady-state concentrations of about one-fourth of those in maternal plasma. 27
Clinical Studies
The two first major placebo-controlled trials of duloxetine in the treatment of fibromyalgia, were those of Arnold et al28,29 showing that the drug was effective and safe for reducing pain symptoms during the 3 months of treatment.
The first published multicenter double-blind randomized clinical study 28 was conducted using 207 patients (89% of women, mean age 49 years, 38% suffering from depression). The patients were included according to the criteria of American College of Rheumatology (ACR) and had a score ≥4 on the average pain severity items of the Brief Pain Inventory (BPI).
After one week of treatment with a placebo, the patients took two daily doses of 60 mg of duloxetine (n = 194) or the placebo (n = 103) for 12 weeks. The primary endpoints were the total and pain scores of the Fibromyalgia Impact Questionnaire (FIQ). In this study, duloxetine was found to induce significant overall improvement in their FIQ scores, but not on the pain subscale of this questionnaire. Other outcome measures also showed statistically significant improvements as well as interference with daily activities, number of tender points and stiffness scores. After taking into account the involvement with major depressive disorders, it was concluded that the effect of duloxetine on pain was not due to its antidepressant effect.
In a second multicenter, double-blind, randomized, clinical study, 29 the effect of duloxetine was analyzed in 354 women (mean age 49.6 years, 26% having major depressive disorders) with fibromyalgia defined according to the same criteria. Patients received after randomization one (n = 118) or two daily doses of 60 mg (n = 116) or a placebo (n = 120) during 12 weeks.
The two duloxetine groups’ significantly reduced BPI pain severity scores (the primary outcome in this study) versus the placebo. The efficacy of duloxetine was also demonstrated for daily activities interferences, FIQ, patient global impression of severity and several quality of life measures.
The data concerning women in these two studies were pooled 30 to better assess, the efficacy especially on functional impairment, quality of life, safety and tolerability. It was concluded from these studies that duloxetine was superior to the placebo on all quality of life and functional measures. Duloxetine was found in the two studies to exert its effect on pain independently of its effect on mood.
Duloxetine effect was then evaluated in two other studies with a longer duration.31,32
In the Russel's study, 520 patients meeting the ACR criteria of fibromyalgia were treated during 6 months and received different daily doses of duloxetine of 20 mg increased to 60 mg after 3 months, 60 mg, 120 mg, or a placebo. The primary outcome measures were BPI and Patient Global Impression of Improvement (PGI-I) scores. These two scores were significantly improved at 3 and 6 months in 120 mg/day group versus the placebo. In the 60 mg/day group, the improvement of CGI-I was significant only at 3 months. The safety of duloxetine in this study was confirmed by the lack of change in vital parameters, laboratory test measurements and electrocardiogram.
In the 6-months Chappell's study, duloxetine (60-120 mg/day) was evaluated versus the placebo in 330 patients with fibromyalgia according the ACR criteria. The results of this study were in favor of duloxetine although statistically significant differences were not reached. The number of dropouts was significantly greater with the placebo (14.9%) than with duloxetine (7.4%) (p < 0.05). These results suggest a real benefit of the drug. In this study, a dose-dependency efficacy of duloxetine on PGI-I and pain relief was documented.
These two studies were prolonged with a 6-month open extension phase to assess safety and tolerability of duloxetine when administered on a longer duration. 33 In terms of efficacy, it appears that reduction in pain severity and feelings of improvement were maintained over the entire 12 months of treatment.
As previously written in the introduction, other drugs were used in FMS: analgesics such as tramadol and acetaminophen, TCAs with amitriptyline, SSRIs, other SNRIs as Venlafaxine and milnacipran, and pregabalin. Pregabalin, duloxetine and milnacipran are approved in the USA for FMS. But there are no comparative trials, nor add-on trials in spite of duloxetine being given in combination, or as part of multimodal therapy. 34
Safety
Adverse events associated with duloxetine, as reported in patients treated for major depressive disorder, are generally mild. The clinical conditions for which large numbers of patients have been evaluated include: nausea, headache, dry mouth, constipation, insomnia, dizziness, fatigue, somnolence, decreased appetite and sweating increase. 35 Some of these effects such as constipation, sweating and dry mouth are related to increased norepinephrine availability in the central and peripheral nervous system. Nausea, the most commonly observed side-effect (almost 20%) likewise with selective serotonin reuptake inhibitors, is considered as more directly linked to increased serotonin availability. These side-effects are generally mild or moderate. They appear early during treatment and may disappear even though treatment is still ongoing. In a meta-analysis of safety data in clinical trials, it was concluded that there were no apparent differences in the overall safety of the drug regardless of the indication. 36 Nevertheless it should be considered that in a recently published trial, discontinuation for adverse events at 6 months was 15.3% with duloxetine 60 mg/d and 27% with 120 mg/d, compared with 13.2% with the placebo. 31
Safety considerations need to address detailed cardiovascular and hepatic tolerance.
Cardiovascular Safety
Preclinical studies did not find any data suggesting a cardiotoxic potential of duloxetine. In-vitro studies on human cardiomyocytes found an alteration of transmembrane ionic currents, but experiments in conscious animal models showed no significant changes on blood pressure, heart rate, cardiac rhythm and conduction. 37
A consequence of norepinephrine reuptake by duloxetine may be an increase in blood pressure and heart rate. In a pooled analysis of placebo-controlled studies, 37 a slight and not clinically relevant increase in blood pressure and heart rate was confirmed. No significant changes in the QTc interval or other electrocardiogram parameters have been reported. Cases of hypertension and tachycardia may however have been observed as with other SNRIs.
Palpitations which may be linked to tachycardia have been reported in 1%-10% of patients receiving duloxetine in clinical trials. The first case reports of patients treated by duloxetine consisted in two cases with worsening symptoms of preexisting heart failure associated with marked tachycardia when treated by duloxetine (30-60 mg/day). 38 In one of these cases, the patient presented similar effects when previously treated by venlafaxine. Stevens 39 reported the case of a young patient without cardiac history who presented with episodes of tachycardia, diaphoresis and chest pain occurring two months after initiation of a treatment by duloxetine 20 mg/d. The cessation of the drug intake led to the quick return of the heart rate to normal. The tachycardia reappeared after restarting the drug but could be reduced by the co-prescription of a beta-blocking agent. In all these papers, cardiovascular side-effects of this drug were reversible after withdrawal.
These cardiovascular side-effects may be more frequent with larger supratherapeutic doses of duloxetine. 40 Tachycardia is commonly observed as well as blood pressure increase during duloxetine overdoses. 41
Overall cardiovascular safety of duloxetine appears to be satisfactory in patients with medical conditions for which the drug has been evaluated. Caution is nevertheless required in patients with uncontrolled hypertension or with unstable or advanced heart failure.
Hepatic Safety Profile
The question of a potential hepatotoxicity of duloxetine has been raised since the early development of the drug.42,43
Preclinical rodent and canine studies have demonstrated hepatic effects suggestive of microsomal enzyme induction but failed to indicate a hepatotoxic potential of the drug (no transaminase increases in treated animals and no effect of the drug on mitochondrial beta-oxidation in rat hepatocytes).
In clinical trials, 0.9%-1.7% of patients treated by duloxetine were found to have elevated alanine aminotransferase (ALT) (>3 times the upper limit of normal) versus 0.0%-0.3% of placebo-treated patients. 42 Hepatocellular, cholestatic and mixed hepatocellular-cholestatic forms of hepatic injury have been reported. It appears that in the vast majority of cases elevated ALT was not associated with symptomatic hepatic injury or progression to fulminant hepatitis. Up to now, only one case of fulminant hepatic failure has been reported 44 in the 4 weeks after increase of the daily dose from 30 to 60 mg. However, in this case, the role of another antidepressant, mirtazapine could not be excluded.
It must be considered that the hepatotoxic potential of duloxetine as with other antidepressants may be increased in patients with pre-existing liver disease, with chronic substantial alcohol use or use of other potentially hepatotoxic agents. Routine screening for liver injury is not required with duloxetine use, however it is to be noted that agomelatine, a more recent antidepressant, with a same ALT elevated risk requires liver surveillance at the onset of drug use and at week 2.
Efficacy
The six-month analysis, of a multicenter, double-blind, placebo-controlled, randomized trial, performed in the USA was published recently in Pain. 31 On 520 patients, 82/150 (54.7%) completed the 6-months (64.7% completed 3-months) with 60 mg/d, 79/147 (53.7%) completed 6-months (64.6% completed 3-months) with 120 mg/d and 72/144 (50%) completed 6-months (58.3% completed 3-months) with placebo. Discontinuation for AE at 6-months was 15.3% with 60 mg/d, 27% with 120 mg/d, 13.2% with the placebo. At 6-months, response rate, defined as a ≥50% improvement from baseline to the average pain severity was 35.9% (p = 0.009) with 120 mg/d, 32.6% (p = 0.045) with 60 mg/d, 21.6% with the placebo. It was better with a ≥30% response rate. The Number Need to Treat was 7 for both doses. 31 The direct analgesic effect was stronger than the indirect one to depressive symptoms.
A total of 206 patients from 376 completed the trial. Durability of beneficial response was confirmed concerning pain, mental fatigue and global improvement.
In 2008 the European League Against Rheumatism (EULAR) gave a level of evidence of Ib based on the results of randomized controlled double-blind trials. The strength of recommendation was, A directly based on evidence level I for SNRI including Duloxetine. 16 In 2005 the American Pain Society (APS), gave a level of evidence of II because of the well-designed experimental studies but without meta-analysis and strength of recommendation of B because of level II evidence. 45 It is to be noted that Russell and Chappell's studies were published in 2008 and Arnold's studies pooled in 2007.30–32
A systematic review of randomized trials was undertaken in 2008. 46 Three trials concerning Duloxetine in FMS were involved.28,29,31 The response rate was defined as a ≥50% reduction in the average pain score from baseline to endpoint. The Number Need to Treat (NNT) compared with the placebo was 6.4 (4.7 to 9.9). 46
A meta-analysis for treatment of fibromyalgia syndrome was published in 2009. 10 For SNRIs, duloxetine and milnacipran are efficacious in reducing pain (standardized mean difference SMD, -0.36; 95% confidence interval CI, -0.46 to -0.25; P < 0.001) and sleep disturbances (SMD, -0.31; 95% CI, -0.47 to-0.14; P < 0.001). Duloxetine is effective in improving depressed mood (SMD, -0.26; 95% CI, -0.42 to -0.10; P = 0.001) and quality of life HRQOL (SMD, -0.31; 95% CI, -0.44 to -0.17; P < 0.001). There is no effect of duloxetine on fatigue (weighted mean difference WMD, -0.08; 95% CI, -0.20 to 0.05; P = 0.23).
In the same paper, evidence is strong in favor for the efficacy of the TCA amitriptyline in reducing pain, fatigue and sleep disturbances. Effect on HRQOL is low. SSRIs fluoxetine and paroxetine show efficacy in reducing pain. The effect is minor on HRQOL. There is no effect on fatigue or sleep. 10
What can be said about the evaluation criteria? In daily practice, evaluation must focus on goal symptoms defined both with patient and doctor: mainly pain, pain during activities but also fatigue, and sleep…. Patient Global Impression of Change (PGIC) has a high value. PGIC is always compared with Clinician Global Impression of Change (CGIC). When PGIC and CGIC differ, patient and doctor have to specify their choice about their aims and expected results. Evaluation of a single treatment is not always possible. Usually, treatments are associated and interact together. That is why naturalistic studies are needed. These naturalistic studies may be undertaken after the drug is approved in a country. But it would be more interesting to plan naturalistic studies before the approval to complete data is given to the approval agencies.
In clinical studies, disorders and treatments are modeled. In these studies, the main assessments are: pain on a Visual Analogic Scale (VAS); impact of pain on functions such as daily activity or sleep, and validated combined assessment by the Fibromyalgia Impact Questionnaire (FIQ). None of these are perfect but assessment choices taken in such studies are appropriate to the objectives of the studies considering evolution of scientific thought.
Pain assessment is not enough. One gold standard should be the impact of pain in daily life and another PGIC. In such a complex syndrome as FMS, secondary assessments are needed: fatigue, fatigability due to a standardized activity, sleep, quality of life, burden, anxiety, depression, bowel dysfunction, and sensitivity to cold.
Evaluation of adverse events (AE) is also essential. Today, patients may refuse a drug because of a poorly tolerated AE. Each known AE must be assessed within the clinical studies: the AE symptom and its impact on the patient's quality of life. For duloxetine, it is possible that nausea was not systematically reported by patients in clinical studies. In clinical studies when nausea occurred, it was perhaps better accepted than it would be in daily practice. So phase IV or naturalistic studies are needed.
Patient Preference and Recommended Management of Duloxetine: “Start Low and go Slow”
Duloxetine is efficacious in FMS. 31 Pain improvement begins during the first week and becomes more established after 2-4 weeks. Because discontinuation rate for adverse events, mainly nausea is 15.3% for 60 mg/day and 27.2% for 120 mg/day and because nausea occurs at the onset of treatment, a strategy to prevent or minimize this AE is compulsory.
To give patients the best chance to improve FMS symptoms while reducing the risk of AE, treatment should be started at low doses and increased slowly. Duloxetine should start at 30 mg/day, the drug being taken during the main meal, with 30 mg added after one week up to 60 mg/day, in two separate intakes: 30 mg during breakfast and 30 mg during evening meal and an antiemetic nausea treatment, which may be necessary during the first days.34,47 (Fig. 1).
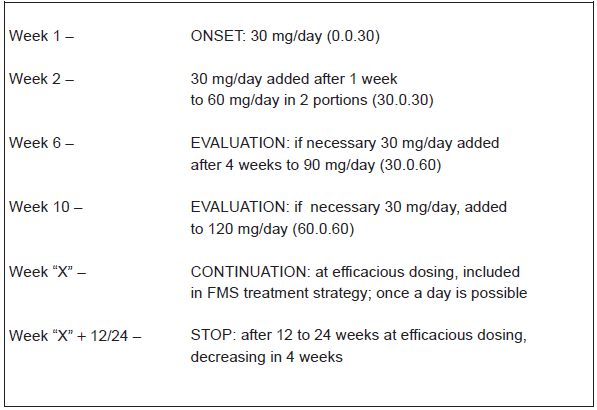
According to evaluation, after 4 weeks, doses can be increased if necessary to improve efficacy if the patient has not experienced a strong adverse event. Because pain efficacy and global impression of improvement were stronger in Russel's study with 120 mg/day and because a dose-dependency of the efficacy using the same criteria was documented in Chappell's study, duloxetine may be increased to 90 or 120 mg/day.31,32 In patients with painful diabetic neuropathy, duloxetine with a dose of 120 mg/day could be appropriate for these patients who require additional pain relief. 48 These doses may be useful in depression or anxiety, for example with 18.28% patients taking 120 mg/day in a general anxiety disorder study with flexible dosing. 49
In cases of efficacious dosing, in agreement with the presented studies, it is possible to stop duloxetine after 3 to 6 months, and eventually 12 months. 33 After this period of remission, the dose may be gradually decreased, with regular assessments. 50 No data about continuation of duloxetine and prevention of relapse of FMS are available. In major depressive disorders recommendations reached 12 to 18 months. 51 The drug could be decreased 2 to 4 weeks before stopping therapy, using the 30 mg daily dose.
Place in Therapy
International recommendations for the treatment of FMS insist on multimodal and multidisciplinary treatments.16,45 A FMS treatment strategy associates different steps: 1. evaluation of FMS with pain and global burden assessment, 2. patient information and education, 3. goals of therapy with symptom reduction for a shared decision, 4. multicomponent therapy including both drug and non-drug, 5. group education or psychotherapy and patient organizations, 6. finally pain clinic consultation in case of major difficulties.
On the one hand non-drug treatments are used: physiotherapy, heated pool, exercise, relaxation or sophrology or hypnosis, cognitive and behavioural therapy, and psychological support mainly with education based upon learning processes. On other hand, associated drug treatments are used: paracetamol and/or tramadol, pregabalin and/or duloxetine or milnacipran 52 (Fig. 2).

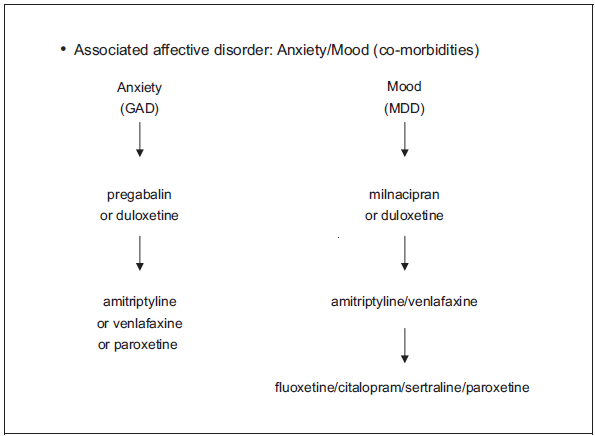
In cases of psychiatric comorbidities, it should be remembered that duloxetine is effective for both anxiety and depression. 34 Duloxetine may be a first choice because of the strong evidence for its efficacy. The same evidence exists for pregabalin in anxiety-FMS co-morbidity and milnacipran in depression-FMS co-morbidity. Amitriptyline may be a second choice drug because of the high level of side effects from this drug. Other drugs have a lower efficacy 10 (Fig. 3). The subgrouping of FMS patients while considering co-morbidities is needed: mood disorders, pain or fatigue or sleep disorders, irritable bowel syndrome… and considering dimensions of pain: active or passive coping, level of self-efficacy, expectancies, anxiety. (Fig. 3). Duloxetine is ranked among the main drug-therapies of FMS as are milnacipran or pregabalin.
Conclusions
Duloxetine as a serotonin norepinephrine reuptake inhibitor is effective in depression, anxiety, neuropathic pain, and fibromyalgia. FMS is a model of chronic pain disorder. 53 Study results demonstrated that duloxetine is efficacious and safe in FMS, at doses of 60 mg/day and 120 mg/day. The Number Need to Treat (NNT) for at least 50% pain reduction is 6. It is a useful drug in FMS, a condition which may be difficult to treat. Adverse events must be explained to the patient and prevented if possible, to avoid withdrawal. Number Needed to Harm (NNH) is 15 for adverse events and 6 for nausea, the most common adverse event.
Also effective for anxiety and depression, duloxetine ranks among the main first place drugs for FMS. Concerning pharmacotherapy of fibromyalgia, more studies are necessary on milnacipran and pregabalin to share knowledge and improve clinical choices. Comparative studies between efficient drugs and naturalistic studies for these efficient drugs are needed.
FMS, being a multidimensional experience with chronic pain, needs a multimodal treatment associating drug and nondrug treatments. Patients must be informed and they must agree to set a realistic therapeutic goal in which duloxetine may play its part in the pharmacotherapy of fibromyalgia.
Disclosures
Eric Serra has served as a consultant, speaker or received funding from Lilly and Boehringer, Pfizer, Pierre Fabre, Grünenthal, Sanofi-Aventis.
Footnotes
Acknowledgments
Rosemary Ancelle-park, Public Health, Paris, France.
