Abstract
Insulin lispro (Humalog®) is a recombinant human insulin analogue with reversed amino acids at position 28 and 29 on the insulin B-chain (Lys(B28), Pro(B29)). It has a faster onset and shorter duration of glucose-lowering activity than regular human insulin (RHI), which is a great advantage. Insulin lispro can be injected immediately before meals or even after meals. This short injection-meal interval contributes greatly to convenience, flexibility, treatment compliance, and patients’ quality of life, which in turn lead to better glycemic control. Numerous clinical trials have demonstrated that insulin lispro, alone or as mixture-25 or -50, controls postprandial blood glucose and stabilizes diurnal variation satisfactorily. The risk of hypoglycemia, including severe or nocturnal episodes, was lower in insulin lispro users than in RHI users. This insulin might be suitable for type 1 diabetic patients, especially when using an insulin pump. Combination therapy of lispro with a sulphonylurea or glargine is safe and efficacious. Several pharmacoeconomic studies have revealed the usefulness of this insulin.
Introduction
The primary goal of treatment of diabetes mellitus is to prevent both micro- and macro-vascular complications and permit the patient to live out their natural life span by maintaining near-normal glycemic control. Past interventional studies like the Diabetes Control and Complication Trial (DCCT), 1 the United Kingdom Prospective Diabetes Study (UKPDS), 2 and the Kumamoto study 3 have demonstrated the importance of intensive therapy in preventing the onset and delaying the progression of diabetic complications in both type 1 and type 2 diabetic patients. Moreover, in UKPDS, intensive therapy was recently shown to reduce total deaths and deaths from myocardial infarction. 4 There is no doubt that insulin is the most effective hypoglycemic agent, but insulin therapy often raises problems in the social activities, physical function and emotional status of patients.5–7 With intensive treatment, multiple injections or severe hypoglycemia may worsen the patient's quality of life (QOL) and may induce compliance problems with insulin therapy. 8 Regular human insulin (RHI) has several limitations in terms of blood glucose control and patients’ QOL. Because of its slow onset of action, patients must inject RHI 30–45 minutes before meals and may find this inconvenient. Patients sometimes might not adhere to the required injection-meal interval. There is a safety concern when patients cannot eat as they plan, as late postprandial hypoglycemia may occur. With RHI, it is difficult to normalize postprandial hyperglycemia without the risk of hypoglycemia. Insulin lispro (Humalog®, Eli Lilly and Company, Indianapolis, USA) is a rapid-acting insulin analogue that can be administered just before meals, and this agent is expected to overcome the difficulties most RHI users experience every day.9,10 This review discusses the properties, therapeutic efficacy, and safety of insulin lispro and its mixtures.
Mechanism of Action, Metabolism and Pharmacokinetic Profile
Mechanism of action
Insulin lispro is a recombinant human insulin analogue, which is synthesized in a special non-pathogenic laboratory strain of Escherichia coli bacteria that has been genetically altered to produce insulin lispro.11,12 Chemically, it is Lys(B28), Pro(B29) human insulin analogue, created when the amino acids at position 28 and 29 on the insulin B-chain are reversed. The primary structure of insulin lispro is shown in Figure 1.
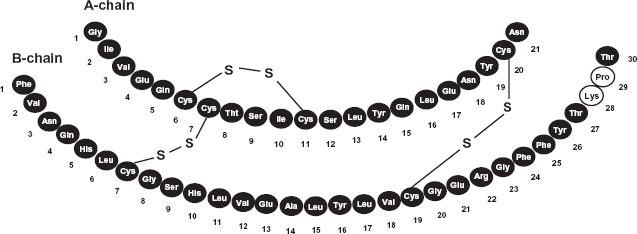
Primary structure of insulin lispro. The amino acids at positions 28 and 29 on the insulin B-chain of human pancreatic insulin are reversed.
Insulin lispro has the empirical formula C257H383 N65O77S6 and a molecular weight of 5808, both of which are identical to those of human insulin. 13 As a result of amino acid transposition, lispro has a significantly lower capacity for self-association into dimers than has RHI,14–16 leading to its more rapid action. 17 The glucose-lowering activity of lispro and RHI is comparable on a molar basis. One unit of lispro has the same glucose-lowering activity as one unit of RHI.
Pharmacokinetic Profile
The bioavailability of insulin lispro is equal to that of RHI, ranging between 55% and 77% with doses of 0.1 to 0.2 U/kg. 13 However, insulin lispro is absorbed more quickly than RHI and has a shorter duration of activity.9,18 In healthy volunteers given subcutaneous doses of lispro ranging from 0.1 to 0.4 U/kg, peak plasma concentration was observed 30 to 90 minutes after administration. When healthy volunteers received equivalent doses of RHI, peak insulin concentration was observed 50 to 120 minutes after administration. 13
Plasma insulin concentration returns to the baseline value faster with insulin lispro than with RHI. Figure 2 shows a comparison of the time-dependent serum insulin concentration profile with insulin lispro and RHI. Patients with type 1 diabetes received subcutaneous injection of 0.2 U/kg insulin lispro (n = 10) or RHI (n = 10) immediately before a high carbohydrate meal.13,19 Insulin lispro was absorbed at a consistently faster rate than was RHI, regardless of the site of subcutaneous injection (abdominal, deltoid, or femoral subcutaneous site). 13 After abdominal administration of lispro, serum insulin level was higher and the duration of action was slightly shorter than with deltoid or thigh administration. 20 The parameters of lispro activity (time of onset, peak time, and duration) as presented in Figure 2 should be considered only as general guidelines. The rate of absorption and consequently the onset of activity are known to be affected by the site of injection, exercise, and other variables.
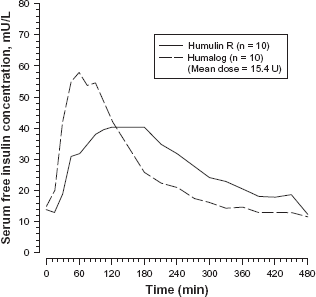
Plasma insulin concentrations of insulin lispro and Humulin R (regular human insulin). Patients with type 1 diabetes mellitus were injected with insulin lispro or Humulin R (0.2 unit/kg) subcutaneously immediately before a high carbohydrate meal.
Recently, several studies comparing the pharmacodynamics and pharmacokinetic properties between insulin glulisine and lispro were performed. Heise et al reported that in subjects without diabetes insulin glulisine showed a faster onset of action compared with lispro (0.2 U/kg: P < 0.05, 0.4 U/kg: P < 0.001), independent of body mass index and dose. 21 Luzio et al reported that glulisine achieved significantly lower glucose excursions (P < 0.01) over insulin lispro when injected immediately before a meal in obese patients with type 2 diabetes. 22 Homko et al compared insulin levels and actions of lispro and aspart in type 1 diabetes and it was concluded that both lispro and aspart were indistinguishable from each other with respect to circulating levels and effectiveness. 23 According to the report of Bode, there were no significant differences in the rate of absorption and glycemic control among 3 rapid-acting insulin analogues-lispro, aspart, and glulisine. 24 In this article, it was stated that insulin glulisine showed a faster onset of action compared with lispro and aspart, however, this advantage lasted only for 1 hour, after which the pharmacokinetic properties were similar among 3 analogue insulin.
Distribution, metabolism, and elimination
The volume of distribution following injection of insulin lispro is identical to that of RHI, with a range of 0.26 to 0.36 L/kg. Human metabolism studies have not been conducted. However, animal studies indicate that the metabolism of lispro is identical to that of RHI. When lispro is given subcutaneously, its t1/2 is shorter than that of RHI (1 versus 1.5 hours, respectively).13,18,25 When given intravenously, lispro and RHI show identical dose-dependent elimination, with a t1/2 of 26 and 52 minutes at 0.1 U/kg and 0.2 U/kg, respectively. 13
Clinical Studies
Insulin lispro
Type 1 diabetes mellitus
Clinical data on the efficacy of insulin lispro compared with RHI for patients with type 1 diabetes is based on several randomized, multicenter trials.26–29 In these trials, premeal bolus lispro or RHI was administered with basal insulin, ultralente or neutral protamine Hagedorn (NPH) insulin. Insulin lispro was generally associated with significantly lower 2-hour postprandial plasma glucose than that with RHI. Mean overall 2-hour postprandial plasma glucose level was significantly lower with lispro in the largest trial, 26 and was also significantly lower with lispro after two or more meals (breakfast and lunch, 27 breakfast and dinner, 28 or after all three meals. 29 Patients using lispro had significantly smaller overall mean 2-hour postprandial blood glucose excursion (difference in blood glucose levels before and after meals) 26 and mean value for two or more meals28,29 than those in patients using RHI. Fasting and bedtime plasma glucose levels were similar between treatment groups.27,28 HbA1c achieved with lispro was also similar to that with RHI. The risk of hypoglycemia with lispro was similar to28,29 or significantly lower than26,27 that with RHI. There was also a significantly lower frequency of both nocturnal and severe hypoglycemia with lispro than with RHI.26,27,29
In intensive treatment for type 1 diabetes mellitus, several clinical trials have investigated how to adjust the dose of rapid-acting insulin, or how to use neutral insulin when switching from conventional RHI. Ebeling et al performed an open-label trial to determine the appropriate intensive treatment regimen to improve glycemic control and reduce hypoglycemic events simultaneously, and they pointed out the necessity of twice a day injection of neutral insulin before breakfast and at bedtime. 30 As premeal insulin, RHI was switched to insulin lispro in 66 type 1 diabetic patients for 5 months. The amount of both lispro and basal insulin could be adjusted according to the results of self-monitoring of blood glucose. The mean daily plasma glucose level, all postprandial glucose values, and HbA1c were all improved, without increasing the frequency of hypoglycemia. The dose of basal insulin was increased by 8 units and that of bolus insulin was decreased by 5 units, leading to an increase in total daily insulin dose of 3 units. The number of daily basal insulin injections significantly increased from 1.4 to 3.1. Thirty two percent and 54% of the patients considered lispro equal to or better than RHI, respectively. Lalli et al performed a longer trial to investigate the possibility that insulin lispro could be successfully implemented in long-term intensive treatment of type 1 diabetic patients. 31 Fifty six patients were randomized to lispro (n = 28) or RHI (n = 28) as premeal insulin for one year. Insulin lispro and RHI were administered at mealtimes and 10–40 minutes before meals, respectively. Bedtime NPH insulin administration was continued in both groups. There was no significant difference in total insulin dose between the two treatment groups. In the lispro plus NPH group, a 30% lower dose of lispro at meals and 30% higher dose of NPH was necessary compared with those in the RHI plus NPH group. There was no significant difference in the amount of NPH injected at bedtime between the two groups. In the lispro plus NPH group, the mean daily blood glucose level (8.0 ± 0.1 vs. 8.8 ± 0.1 mmol/L, P < 0.05), HbA1c (6.34 ± 0.10 vs. 6.71 ± 0.11%, P < 0.002), and the frequency of hypoglycemia were significantly (P < 0.05) lower compared with the comparator.
Combination therapy with insulin lispro and glargine was also shown to be effective in the treatment of type 1 diabetes in several clinical trials. Murphy et al performed a randomized crossover trial in 28 adolescents with type 1 diabetes on multiple injections receiving either glargine at bedtime plus preprandial lispro or NPH insulin at bedtime plus pre-prandial RHI. 32 Compared with RHI plus NPH, lispro plus glargine was associated with lower mean blood glucose level (fasting: 8.0 vs. 9.2 mmol/l, P < 0.0001; 2h post breakfast: 8.1 vs. 10.7 mmol/l P < 0.0005). The incidence of nocturnal hypoglycemia was 43% lower in the lispro plus glargine group compared with the RHI plus NPH group; however, there was no difference in the rate of self-reported symptomatic hypoglycemia. Total insulin dose required to achieve target blood glucose control was significantly lower on lispro plus glargine (1.16 IU/kg) compared with RHI plus NPH (1.26 IU/kg, P < 0.005), but there was no significant difference in HbA1c (8.7 vs. 9.1%, P = 0.13). Raskin et al performed a large-scale, open-label trial to explore the efficacy and safety of combination therapy with insulin lispro and glargine. 33 Patients with type 1 diabetes receiving basal-bolus injection of NPH and lispro were randomized to receive glargine once a day (n = 310) or NPH (n = 309) as basal treatment with continued bolus lispro for 16 weeks. Compared with patients with NPH, patients with glargine had a significant decrease in fasting plasma glucose (−2.33 ± 0.26 vs. −0.69 ± 0.26 mmol/l, P = 0.0001). More patients with glargine (29.6%) than with NPH (16.8%) reached the target fasting plasma glucose level of 119 mg/dl (6.6 mmol/l). However, there was no difference with respect to change in HbA1c (P = 0.8409). Glargine users were also associated with a significant decrease in the variability of fasting plasma glucose compared with NPH users (P = 0.0124). The rate of symptomatic hypoglycemia, including nocturnal hypoglycemia, was not different between the two treatment groups. Weight gain was 0.12 kg in glargine users and 0.54 kg in NPH users (P = 0.034). Rossetti et al performed an open-label trial to compare the basal insulin regimens, NPH injection four times a day or glargine once a day at dinner or at bedtime, as to which is better for blood glucose control in type 1 diabetic patients receiving insulin lispro at mealtimes. 34 Fifty one patients with type 1 diabetes receiving intensive insulin therapy (four times daily NPH as basal and mealtime insulin lispro as bolus) were randomized to three different treatment regimens of basal insulin substitution; four times daily NPH (n = 17), once daily glargine at dinner (n = 17), and once daily glargine at bedtime (n = 17). In all three groups, treatment with lispro as bolus substitution was continued. Mean daily plasma glucose level was significantly lower with dinnertime (7.5 ± 0.2 mmol/l) or bedtime (7.4 ± 0.2 mmol/l) glargine than with NPH (8.3 ± 0.2 mmol/l)(P < 0.05). Compared with baseline, HbA1c was improved in the dinnertime (from 6.8 ± 0.2 to 6.4 ± 0.1%) and bedtime (7.0 ± 0.2 to 6.6 ± 0.1%) glargine group, but did not change in the NPH group (P < 0.04 vs. NPH) at the end of the study. Total daily insulin dose was similar in all three groups, but in the glargine groups there was an increase in basal and a decrease in bolus insulin needed for blood glucose control. The frequency of mild hypoglycemia was lower with glargine than with NPH (P < 0.04). Hamann et al performed an open-label, randomized, parallel group, multicenter trial to investigate whether insulin glargine is equally effective if administered before breakfast, before dinner, or at bedtime. 35 Patients with type 1 diabetes on basal-bolus therapy (n = 378) were treated with once-daily individually titrated insulin glargine in combination with preprandial insulin lispro for 24 weeks. Mean total daily insulin dose was similar at baseline and remained almost constant during the study period in all three treatment groups. Mean HbA1c value was similarly decreased from baseline to the endpoint of the study in all three groups. A similar percentage of patients achieved the target HbA1c value of 7.0% in the three groups. The diurnal blood glucose profile in relation to injection timing of glargine was similar in all groups. The total incidence of hypoglycemia did not differ among the three groups, but nocturnal hypoglycemia was less frequent in the morning injection group (59.5%) compared with the dinnertime (71.9%) and bedtime (77.5%) injection groups (P = 0.005). These data suggest that combination therapy consisting of lispro and glargine is not only effective, but also safe and flexible. All of these four trials suggested the effectiveness of this combination therapy for the treatment of type 1 diabetes mellitus.
Kawamori et al conducted an open, randomized, parallel-group, comparative non-inferiority trial to compare the efficacy and safety between insulin lispro and glulisine in type 1 diabetic patients, using insulin glargine as basal insulin. 36 With respect to change in HbA1c, mean 2h-postprandial hypoglycemia (P = 0.065), hypoglycemia-related events (P = 0.437), and tolerability, there were no significant differences between lispro and glulisine in this trail.
Physicians who treat patients with type 1 diabetes sometimes try to minimize the number of daily injections to reduce the treatment burden and improve compliance despite the manufacturer's caution against mixing insulin glargine with lispro. However, the possibility was also pointed out that mixing these agents might be disadvantageous for the treatment of type 1 diabetes. To examine whether mixing with glargine has an adverse effect on the early pharmacodynamic action of lispro, euglycemic glucose clamp was performed in 11 young people (age 15.1 ± 3 years old) with type 1 diabetes with lispro (0.2 units/kg) and glargine (0.4 units/kg), injected either separately or mixed. 37 Mixing glargine with lispro shifted the dose curve to the right, with a significantly lower glucose infusion rate (GIR) after mixed injection between 60 and 190 minutes and significantly higher GIR between 270 and 300 minutes, lowered GIRmax (separate 7.1 ± 1 vs. mix 3.9 ± 1, P = 0.03) and significantly delayed the time to reach GIRmax (separate 116 ± 8 min vs. mix 209 ± 15 min, P = 0.004). The GIR area under the curve was significantly lower with the mixed injection. These data demonstrate that mixing lispro with glargine markedly flattens the early pharmacodynamics peak of lispro and causes a shift to the right in the GIR curve that might lead to difficulties in controlling postprandial glucose excursions.
Type 2 diabetes mellitus
In randomized, open-label clinical trials of 4–12 months duration, the efficacy of insulin lispro compared with RHI in adults with type 2 diabetes has been examined.38–41 After a run-in period of 2–6 weeks, they received lispro or RHI three times daily at meals in combination with long-acting insulin. Their baseline HbA1c was 7.9%–8.9%. It was demonstrated that 2-hour postprandial blood glucose level40,41 and 2-hour postprandial blood glucose excursion38,40 were significantly decreased with lispro compared with RHI. HbA1c at the endpoint38–40 and the frequency of hypoglycemic episodes during the trial period38,39 were similar between the treatment groups, but in the largest multicenter, cross-over trial with 722 patients, the frequency of nocturnal hypoglycemia (hypoglycemic episodes between midnight and 6 am) was significantly lower in lispro users. 40
In 25 patients with type 2 diabetes who developed secondary failure of sulfonylurea therapy, insulin lispro plus a sulfonylurea significantly reduced fasting (from 10.9 to 8.5 mmol/l, P < 0.0001) and 2-hour postprandial blood glucose levels (from 18.6 to 14.2 mmol/l, P < 0.0001) and HbA1c (from 9.0 to 7.1%, P < 0.0001) compared with sulfonylurea therapy alone. 42 The usefulness of lispro plus sulfonylurea therapy was also demonstrated by Bastyr et al. 43 This combination therapy improved HbA1c with less frequent hypoglycemia and other adverse events compared with NPH plus a sulfonylurea or metformin plus a sulfonylurea. 44 Bretzel et al performed the APOLLO study (A Parallel design comparing an Oral anti diabetic drug combination therapy with either Lantus once daily or Lispro at mealtime in type 2 diabetes patients failing Oral treatment). In this open-label study of 44-weeks duration, 418 patients who developed secondary failure of oral hypoglycemic agents were randomly assigned to either insulin glargine injected once daily at the same time or to insulin lispro administered three times daily. 45 The primary objective was to compare the change in HbA1c from baseline to the endpoint between the two groups. Two hundred and five patients were randomly assigned to glargine and 210 to lispro. Mean decrease in HbA1c was −1.7% (from 8.7 ± 1.0% to 7.0 ± 0.7%) in the glargine group and −1.9% (from 8.7 ± 1.0% to 6.8 ± 0.9%) in the lispro group, which was within the limit of non-inferiority. One hundred and six (57%) and 131 (69%) patients achieved HbA1c of 7% or less in the glargine group and lispro group, respectively. In the glargine group, the decreases in fasting plasma glucose (−4.3 mmol/l vs. −1.8 mmol/l, P < 0.0001) and bedtime plasma glucose (−3.3 mmol/l vs. −2.6 mmol/l, P = 0.0041) were greater than those in the lispro group, whereas insulin lispro better controlled postprandial plasma glucose throughout the day (P < 0.0001). The incidence of hypoglycemia was lower in the glargine group than in the lispro group (5.2 vs. 24.0 events per events per patient per year, P < 0.0001).
Epidemiological studies have shown a strong association between postprandial or post-challenge hyperglycemia and cardiovascular risk.46,47 The Diabetes Epidemiology Collaborative Analysis of Diagnostic Criteria in Europe (DECODE) 48 and the Diabetes Epidemiology Collaborative Analysis of Diagnostic Criteria in Asia (DECODA) 49 studies, which analyzed baseline and 2-hour post-challenge glucose level from large prospective cohort studies, found that 2-hour post-challenge glucose level is a better predictor of all-cause and cardiovascular disease mortality than fasting plasma glucose. Hanefeld et al performed a meta-analysis showing that acarbose, which is an alfa-glucosidase inhibitor that specifically suppresses postprandial plasma glucose excursion, reduced the risk of cardiovascular events. 50 This finding is consistent with findings from the STOP-Noninsulin–Dependent Diabetes Mellitus (NIDDM) trial, which showed that treating people with impaired glucose tolerance with acarbose was associated with a significant reduction (P = 0.02) in the risk of cardiovascular disease. 51 Raz et al performed a multinational, randomized, controlled trial called “Hyperglycemia and its Effects After Acute Myocardial Infarction on Cardiovascular Outcomes in Patients with Type 2 Diabetes Mellitus (HEART2D)”, in order to compare the effect of prandial versus fasting glycemic control on risk for cardiovascular outcomes in patients with type 2 diabetes after acute myocardial infarction (AMI). 52 A total of 1115 patients were randomly assigned within 3 weeks after AMI to the two treatment groups. One was treated with three times daily injection of insulin lispro at meals with a target of 2-hour postprandial blood glucose less than 7.5 mmol/l (n = 557), and the other was treated with NPH insulin twice daily or insulin glargine once daily with a target of fasting/premeal blood glucose less than 6.7 mmol/l (n = 558). The mean duration of patients’ participation after randomization was 963 days. The achieved HbA1c was similar between the treatment groups (lispro group 7.7 ± 0.1 vs. glargine group 7.7 ± 0.1%, P = 0.4). In the lispro group, mean daily postprandial plasma glucose concentration (7.8 vs. 8.6 mmol/l, P < 0.01) and 2-hour postprandial plasma glucose excursion (0.1 vs. 1.3 mmol/l, P < 0.001) were lower than those in the glargine group. On the contrary, mean fasting plasma glucose was lower in the glargine group (7.0 vs. 8.1 mmol/l, P < 0.001). No difference was found between the two treatment groups with respect to risk for first cardiovascular event in type 2 diabetic patients with a recent AMI (hazard ratio 0.98 [95% CI 0.8–1.21]).
Lispro Mixtures
Insulin lispro mixture-50
Insulin lispro mixture-50 is an insulin analogue preparation composed of a 1:1 mixture of insulin lispro and neutral protamine insulin lispro (NPL) suspension. This preparation is therefore expected to have an advantage in suppressing postprandial hyperglycemia. It has been reported that the ratio of the basal insulin requirement to the bolus insulin requirement is approximately 1:1 in healthy individuals. 53 It is therefore rational to administer exogenous insulin while keeping the ratio of basal to bolus insulin at 1:1 in diabetic patients.
Lispro Mixture-50 in Type 1 Diabetes
Randomized cross-over clinical trials have investigated the efficacy of insulin lispro mixtures in type 1 diabetic patients. Hers et al compared treatment with mixture-50 and premeal RHI. 54 Roach et al compared mixture-50 with insulin lispro self-mixed with NPH insulin. 55 Intensive treatment with mixture-50 achieved similar HbA1c to premeal RHI, and lispro self-mixed with NPH insulin. Postprandial plasma glucose levels with mixture-50 were similar to those with comparators (only plasma glucose after supper was lower with mixture-50). Premeal and bedtime plasma glucose levels were similar between the two treatment groups.
Mixture-50 Three Times Daily in Type 2 Diabetes
Mixture-50 three times daily injection is expected to provide an adequate reduction of postprandial blood glucose level and sufficient reduction of pre-prandial blood glucose level, possibly achieving blood glucose control comparable to that with conventional basal-bolus therapy (four or more injections daily). 56 We conducted a study to examine the efficacy of mixture-50 three-times daily injection monotherapy. 57 The subjects were 35 patients with type 2 diabetes, whose average HbA1c was 10.5%. After the initiation of mixture-50, HbA1c improved significantly (8.2 ± 1.1% at 6–8 weeks and 7.2 ± 1.1% at 6 months) (at 6–8 weeks and 6 months vs. the baseline, P < 0.001). The bedtime plasma glucose level correlated with improvement of HbA1c (r = −0.41, P = 0.014). Therefore, it is suggested that bedtime plasma glucose level can serve as a predictor of the outcome of long-term blood glucose control during this insulin regimen and that bedtime plasma glucose level needs to be reduced sufficiently to achieve adequate blood glucose control. Kazda et al conducted a trial which compared mixture-50 three-times daily with glargine once daily therapy or with insulin lispro three-times daily therapy. 58 In this trial, premeal lispro and mixture-50 three-times daily injection was associated with significantly lower postprandial plasma glucose levels (P < 0.001 for both), smaller glucose excursion, and better HbA1c (P = 0.001 and P < 0.001) than glargine at bedtime. On the other hand, lispro and mixture-50 were associated with a significantly smaller reduction in fasting plasma glucose level than was insulin glargine.
Mixture-50 Twice Daily in Type 2 Diabetes
Basal-bolus insulin therapy has been shown to be useful for controlling both pre- and postprandial blood glucose levels and for lowering HbA1c level. In the presence of comparatively preserved endogenous insulin secretion, however, elderly patients or patients who find it difficult to perform self-injection during the daytime often prefer to receive fewer injections. We investigated the effects on diurnal variation of blood glucose levels and postprandial blood glucose elevation of switching from twice daily rapid-acting insulin 70/30 (biphasic insulin aspart 70/30 (insulin aspart 30% and insulin protamine aspart 70%) and biphasic human insulin 70/30) or lispro miture-25 to twice-daily lispro mixture-50. 59 The initial dose of lispro mixture-50 was the same as that of their previous insulin preparation, but could be adjusted. Mixture-50 significantly suppressed the blood glucose elevation from the time before breakfast to the period between breakfast (P < 0.01) and lunch (P < 0.05) and also from the time before supper to the period between supper (P < 0.05) and bedtime (P < 0.01), thereby stabilizing the diurnal variation of blood glucose levels. None of the patients experienced any episodes of hypoglycemia.
Insulin Lispro Mixture-25
Insulin lispro mixture-25 is an insulin analogue preparation composed of a 25:75 mixture of insulin lispro and NPL suspension. This agent has been shown to be a favorable alternative to biphasic human insulin 70/30 in patients with type 2 diabetes. 60 Postprandial plasma glucose levels were lower in patients with mixture-25 than in patients with biphasic human insulin 70/30 (after breakfast 8.95 ± 2.17 vs. 10.00 ± 2.28 mmol/l, P = 0.017; after dinner 9.28 ± 2.51 vs. 10.27 ± 2.76 mmol/l, P = 0.014), although HbA1c was similar with both agents (mixture-25 7.8%, 70/30 8.1%, P = 0.408). Niskanen et al showed that mixture-25 twice daily and biphasic insulin aspart 70/30 were equal with respect to improving HbA1c and pre- and postprandial plasma glucose levels. 61
CSII of Insulin Lispro
The efficacy of insulin lispro as CSII via an external pump has been compared with that of a regimen of multiple daily injections (insulin lispro before meals and NPH as basal insulin62–64 and of CSII with RHI65–68 in randomized trials in patients with type 1 diabetes. A crossover study in patients with type 1 diabetes conducted by Hanaire-Broutin et al showed superior glycemic control (HbA1c 7.89 ± 0.77 vs. 8.24 ± 0.77%, P < 0.001) with lower daily doses of insulin (38.5 ± 9.8 vs. 47.3 ± 14.9 U/day, P < 0.0001) when insulin lispro was given by CSII rather than as part of a multiple dose regimen. 62 This superiority of CSII with lispro over multiple daily injections in type 1 diabetes was also shown in study conducted by Hoogma et al. 63 HbA1c (7.45 vs. 7.67%, P < 0.001) and mean daily blood glucose level (8.6 vs. 9.4 mmol/l, P < 0.001) were significantly lower with lispro via CSII than via multiple daily injections. The frequency of hypoglycemic episodes was significantly lower with CSII with insulin lispro. On the other hand, a parallel-group trial in patients with type 1 diabetes conducted by Tsui et al showed no significant difference in glycemic control after 9 months of therapy with lispro in a CSII regimen (n = 13) versus a multiple daily injection regimen (n = 14). 64 Zinman et al performed a double blind, crossover study in patients with type 1 diabetes. 65 In this study, CSII with lispro was advantageous in improving HbA1c over CSII with RHI (7.66 ± 0.13 vs. 8.00 ± 0.16%, P = 0.0041). One-hour postprandial plasma glucose levels after breakfast, lunch, and dinner were significantly lower with CSII with lispro than with RHI: 8.35 vs. 9.79 mmol/l (P = 0.006), 7.58 vs. 8.74 mmol/l (P = 0.049), and 7.85 vs. 9.01 mmol/l (P = 0.03). Furthermore, the rate of hypoglycemia was also lower with CSII with lispro. Renner et al also reported that CSII with lispro significantly reduced HbA1c (6.8 ± 0.9 vs. 6.9 ± 1.0%, P < 0.02) and 2-hour postprandial plasma glucose levels (P < 0.001) more than did CSII with RHI, without increasing the incidence of hypoglycemia. 66 Campbell et al reported that by switching from CSII with RHI to that with lispro, HbA1c (8.0 ± 1.6 vs. 7.1 ± 1.2, P = 0.0001), daily total insulin dose (45.3 ± 24.9 vs. 41.2 ± 20.2 U/day, P = 0.0001), and the frequency of hypoglycemia (9.6 vs. 7.1 events/month, P = 0.0001) were decreased, improving the patients'QOL. 67 In another study there was no significant difference in HbA1c, postprandial glucose levels, and rate of hypoglycemia between the two treatment groups. 68
Recently, van Bon et al conducted a randomized, open-label, crossover, controlled multicenter study comparing insulin glulisine with aspart and lispro administered by CSII in patients with type 1 diabetes. 69 In this report, there were no significant differences in HbA1c at endpoint, most points of the seven-point glucose curves, severe hypoglycemia, and symptomatic ketoacidosis among 3 insulin analogues.
In a trial in elderly patients with type 2 diabetes, the efficacy of insulin lispro via CSII was also compared with a multiple daily injection regimen (premeal lispro with glargine as basal insulin). 70 In this study, total daily insulin dose and rate of hypoglycemic episodes were similar between CSII with lispro and multiple daily injections of lispro as bolus and glargine as basal substitution.
Special Populations
Pediatric use
The results of two randomized, open, crossover trials have shown no inferiority of postprandial glycemic control with insulin lispro relative to RHI in children with type 1 diabetes mellitus. In trials conducted by Tupola et al in 24 patients (median age of 6 years), 71 and by Fairchild et al in 35 patients (median age of 8 years), 72 no significant differences in glycemic control and rate of hypoglycemic episodes between lispro and RHI were shown. In a trial conducted by Deeb et al in 61 patients (median age of 8 years), no significant difference in glycemic control or rate of hypoglycemia was observed between lispro and RHI either; however, lispro reduced 2-hour postprandial blood glucose levels significantly (P < 0.001) compared with RHI. 73
In a 12-month, randomized prospective study of adolescents aged 13 to 34 years, comparable glycemic control measured by HbA1c was achieved regardless of treatment; insulin lispro 10 to 15 minutes before meals versus RHI 20 to 40 minutes before meals. 74 The 2-hour postprandial glucose level was significantly lower (P < 0.05) in the lispro group. The total insulin dose was similar between the two groups. The incidence of hypoglycemia was lower (P < 0.04) in the lispro group at 6 months. Lower 2-hour postprandial glucose level and rate of hypoglycemia with insulin lispro than with RHI in adolescents (aged 9 to 18 years) were also shown by Holcombe et al. 75 In this trial, patients were randomized to receive 4 months of treatment with lispro administered immediately before meals or RHI administered 30 to 45 minutes before meals. Hypoglycemic episodes (P = 0.023), especially nocturnal hypoglycemia (P < 0.001), were significantly less frequent with lispro.
Rami et al conducted a randomized crossover study to compare plasma glucose levels before breakfast, and 30, 60, and 120 minutes after breakfast among three groups of children and adolescents with type 1 diabetes; RHI 20 minutes before breakfast, lispro immediately before breakfast, and lispro 15 minutes after breakfast. 76 The group administered lispro immediately before breakfast showed significant suppression of postprandial blood glucose elevation compared with the other two groups. The plasma glucose levels at 30, 60, and 120 minutes after breakfast were similar in the group administered RHI before breakfast and the group administered lispro 15 minutes after breakfast, indicating the possibility of postprandial injection of lispro. This would permit flexibility of injection timing and help to improve compliance and QOL of patients.
Geriatric Use
Of the total number of subjects (n = 2834) in eight clinical studies of insulin lispro, 12% (n = 338) were 65 years of age or over. 77 The majority of these were patients with type 2 diabetes. HbA1c and hypoglycemic rate did not differ by age. The frequency of adverse events did not differ significantly between lispro and RHI.
Pregnancy
Reproduction studies have been performed in pregnant rats and rabbits. The results have revealed no evidence of impaired fertility or harm to the fetus due to lispro. 13 There are, however, no adequate and well-controlled studies with lispro in pregnant women. Because animal reproduction studies are not always applicable to humans, this insulin should be used cautiously during pregnancy, only if clearly needed. According to the classification of drug teratogenic effects from the Food Drug and Administration, insulin lispro is categorized as Pregnancy category B. Data for the use of lispro in pregnant women with diabetes are available from small prospective or retrospective studies. Insulin lispro is at least as effective as RHI in not only type 178,79 and type 2 diabetes, but also gestational diabetes.80,81 The effects of pregnancy on the pharmacokinetics and pharmacodynamics of lispro have not been investigated.
Wyatt et al conducted a multinational, multicenter, retrospective study to determine the rate of major congenital anomalies in babies of women with diabetes treated with insulin lispro. 82 The charts of 496 mothers (97% with type 1 diabetes), with 533 pregnancies resulting in 542 offspring, who were treated with lispro before and during pregnancy were examined. The rate of major congenital anomalies was 5.4%, which does not differ from the published major congenital anomaly rates for other insulin preparations.
Various Eating Habits
There is much variation in eating habits among different countries and different cultures around the world. Subsequently, the timing of insulin injections varies from country to country. Many Japanese and Americans eat three times a day. However, Germans have an early breakfast and a midmorning breakfast, and French don't eat breakfast. The typical Italian diet is low in fat and high in complex carbohydrates. The merit of lispro mixture-25 for such diets has been reported. 83 There are some areas where Ramadan is observed, requiring Muslims to fast during the daytime and eat at night for a month. There have been two trials comparing the effectiveness and safety of insulin lispro and RHI in Muslims.84,85 One was a trial in 64 patients with type 1 diabetes 84 and the other in 70 patients with type 2 diabetes. 85 Patients in both studies received NPH insulin as basal substitution. Insulin lispro was administered immediately and RHI 30 minutes before the permitted meals for two weeks. In both trials, lispro suppressed the postprandial elevation of plasma glucose concentration better than did RHI. The rate of hypoglycemia was significantly lower with lispro than with the comparator. The superiority of insulin lispro over RHI under such circumstances was suggested.
Renal or Hepatic Impairment
In case of renal insufficiency, insulin clearance is decreased and circulating insulin lispro concentration is increased. In case of hepatic impairment, limited data suggest the possibility of increase in plasma insulin lispro concentration.86,87 Careful monitoring of blood glucose concentration and hypoglycemic symptoms is mandatory. In some cases, dose reduction of lispro might be necessary.
Safety
Hypoglycemia
As with all insulin preparations, hypoglycemic reactions may be associated with the administration of insulin lispro. The largest trials in patients with type 1 26 and type 2 40 diabetes demonstrated a significantly lower incidence of hypoglycemia in patients with lispro than in those with RHI. A meta-analysis of eight multi center trials demonstrated a reduction in frequency of severe hypoglycemia in patients with type 1 diabetes mellitus using lispro compared with those using RHI. 88 In this meta-analysis, 2756 patients were included; 2327 and 2339 patients were administered lispro and RHI, respectively. Of patients using lispro, 72 (3.1%) had a total of 102 episodes of severe hypoglycemia compared with a total of 131 episodes in 102 (4.4%) patients using RHI (P = 0.024).
Other Adverse Events
The frequency of adverse events in clinical trials was similar for insulin lispro and RHI. Hypersensitivity, pruritus, rash, lipodystrophy, and hypokalemia are potential major clinical adverse events with all insulin preparations.89,90 There were no clinically significant differences in the rates of treatment-related adverse events or progression of retinopathy, neuropathy or cardiovascular events between patients using lispro and those using RHI in a pooled data analysis (n = 3634). 77 In this analysis, both type 1 and type 2 diabetic patients were included. The most common events were headache, infection, flu-like symptoms, rhinitis, and pharyngitis.
Allergy
As with any insulin therapy, patients using insulin lispro may experience redness, swelling, or itching at the site of injection. These minor reactions usually resolve in a few days to a few weeks. Systemic insulin allergy is less frequent, but potentially more serious. It may cause rash (including pruritus) over the whole body, shortness of breath, wheezing, tachycardia, and hypotension. Severe cases of systemic allergy, including anaphylactic reaction, could be life-threatening. In controlled clinical trials, pruritus was seen in 17 patients using RHI (n = 2969) and 30 patients using lispro (n = 2944) (P = 0.053). 13
Antibody Production
In large clinical trials, antibodies that cross-react with human insulin and insulin lispro were observed both in patients treated with RHI and insulin lispro. 91 The overall immunological characteristics of both agents were alike. Similar findings were observed in insulin-naïve patients. 92 There are several case reports in which patients with type 1 diabetes with severe RHI-induced resistance were successfully treated with lispro.93,94
QOL in Patients Using Insulin Lispro and Its Mixtures
Insulin lispro
In a study that randomized 1008 patients with type 1 diabetes to RHI or insulin lispro, 468 patients answered the Diabetes Quality of Life Clinical Questionnaire (DQLCTQ), which is composed of 142 questions covering four primary domains (energy/ fatigue, health distress, treatment flexibility, and treatment satisfaction). 26 There were no differences between treatment groups for the energy/fatigue and health distress domains, but treatment flexibility score (P = 0.001) and treatment satisfaction score (P < 0.001) were significantly higher with lispro than with RHI (higher scores mean better QOL). On the other hand, in a large crossover trial that randomized patients with type 2 diabetes to RHI or lispro, 474 patients answered DQLCTQ, with no significant difference in scores in any of the four primary domains between treatment groups. 40
A study with self-rated QOL as a primary endpoint has shown increased treatment satisfaction and improved QOL when insulin lispro was given in a randomized comparison. 96 The 24-week multi center, open-label, parallel-group study was conducted in Japan in order to evaluate the impact of lispro on patients’ QOL, satisfaction and compliance with insulin therapy. Four hundred and twenty-six diabetic patients (241 type 1 and 185 type 2), who were on multiple injection therapy were randomized to either lispro treatment or RHI and analyzed. 96 Patients’ QOL was evaluated using the insulin-therapy-related QOL (ITR-QOL; a questionnaire developed in Japan), the Diabetes Treatment Satisfaction Questionnaire (DTSQ), and the “Questionnaire on insulin injection”, which is their original questionnaire. DTSQ showed significant improvement in treatment satisfaction (P < 0.001) and frequency of undesired hypoglycemia (P = 0.023) with insulin lispro relative to RHI. ITR-QOL showed significant improvement relative to RHI for a variety of indicators of activity and function in patients receiving lispro. “Questionnaire on insulin injection” revealed significantly higher compliance in patients using lispro.
Ishii et al conducted an open-label, 12-week study of 770 patients whose medication was switched to insulin lispro from RHI in order to evaluate the impact of lispro on patients’ QOL. 97 The primary outcomes were compliance with insulin injection timing, HbA1c, postprandial blood glucose level, frequency of hypoglycemia, and QOL measurements. After switching to lispro, about 95% of patients always or usually complied with the timing of insulin injections as instructed by their doctors. HbA1c was improved from 8.6 to 8.2% (P < 0.001) without increasing the frequency of hypoglycemia. In terms of QOL, a statistically significant improvement (P < 0.001) was observed in the ITR-QOL total score. Statistically significant correlations were observed between compliance with insulin injection timing and glycemic control, as well as glycemic control and QOL (Fig. 3). The improvement in patient convenience obtained by switching to lispro provided better compliance with insulin injection timing, and this in turn led to better glycemic control and improved QOL.
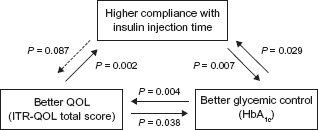
Correlations between high compliance with insulin injection timing, better glycemic control, and better QOL of insulin lispro users.
Lispro Mixtures
The effect of insulin lispro mixture-25 and mixture-50 on QOL of type 1 and type 2 diabetic patients was assessed by means of the ITR-QOL questionnaire. 98 The subjects were patients who were treated with biphasic human insulin 70/30 or 50/50 twice daily. Their insulin regimen was changed to mixture-25 or mixture-50 twice daily injection. In this open-label trial, 215 patients received lispro mixture-25 or mixture-50, and QOL assessment was performed: at the start and after 12 weeks of the study. The average score on the ITR-QOL was significantly higher at the endpoint than at baseline, demonstrating improvement of the patients’ QOL (P < 0.001). Compliance with treatment and adherence to the physicians’ instructions regarding treatment were also significantly improved.
Conclusions
Intensive insulin therapy is considered to be the most effective method for maintaining near-normal glycemic control in patients with diabetes mellitus, but intensive therapy with RHI is limited by its slow onset and long duration of action. Patients have to inject RHI 30 to 60 minutes before meals. Many patients cannot adhere to this injection-meal interval. Its slow onset of action is associated with initial low bioavailability, leading to early postprandial hyperglycemia. Its long duration of action is associated with inappropriate hyperinsulinemia, leading to the risk of hypoglycemia. The pharmacokinetics of RHI does not match the speed at which food (which patients eat as meals) is absorbed.
Insulin lispro was designed for rapid subcutaneous absorption compared with RHI. The rapid onset and shorter duration of action are characteristics of this insulin, which are great advantages over RHI. Because of these advantages, better postprandial glycemic control with a similar or lower rate of hypoglycemia and short injection-meal interval are realized. Insulin lispro can be injected immediately before meals or even after meals. This short injection-meal interval contributes to convenience, flexibility, treatment compliance and patient satisfaction.
Although postprandial plasma glucose was controlled better with insulin lispro than with RHI, available studies show HbA1c to be generally similar between these insulins. In a meta-analysis from 42 studies, short acting insulin analogues, lispro and aspart, and RHI were compared. 99 In adults with type 1 diabetes, the meta-analysis showed a small, but significant decrease in HbA1c using short acting insulin analogues. In patients with type 2 diabetes, no superior effect on HbA1c was observed. However, in subgroup analyses, there was a more pronounced effect on HbA1c in favor of analogues in patients using CSII. In this meta-analysis, almost identical results for trials with lispro and aspart were obtained, which is in accordance with controlled studies using glucose clamp in type 1 diabetic patients.23,100 Insulin lispro might be suitable for type 1 diabetic patients, especially those using insulin pump therapy. Several trials assessing the efficacy of lispro with a long-acting insulin analogue in basal/bolus regimens have been performed. Combination therapy with lispro and glargine seems to be promising.
Not only insulin lispro but also its mixtures -25 and -50 can be administered immediately before or even after meals. This convenient and flexible injection may enable patients to achieve the tight glycemic control required to minimize long-term complications and contribute to patient satisfaction. Three-times daily mixture-50 injection is a promising way to achieve an adequate reduction of both pre-and postprandial blood glucose level, with almost equal blood glucose control as that with conventional basal-bolus therapy. 101 Furthermore, twice-daily mixture-50 injection is reported to be as effective as basal-bolus therapy in controlling blood glucose levels in patients with type 2 diabetes and to provide better QOL as compared with basal-bolus therapy. 102
One pharmacoeconomic study which compared the cost effectiveness of insulin lispro and RHI in type 1 diabetes was published from the United Kingdom. 103 In this analysis, lispro was projected to be superior compared with RHI. Insulin lispro was associated with improved quality-adjusted life expectancy and lower lifetime medical costs than RHI, which derive from the lower incidence of severe hypoglycemic episodes. Although the costs associated with intensive insulin therapy are higher than those of conventional therapy, cost savings might be expected from intensive therapy by delaying or preventing diabetic complications. 104 From another pharmacoeconomic point of view, willingness-to-pay analyses were performed in Australian patients 105 with diabetes and Canadian taxpayers.106,107 The former analysis evaluated insulin lispro in relation to RHI, 105 and the latter evaluated lispro mixture-25 in relation to biphasic human insulin 70/30. 106 In these studies, significantly higher preference for lispro and mixture-25 than for RHI and biphasic human insulin 70/30 was demonstrated. Because of the long-term nature of diabetes mellitus, QOL of diabetic patients is very important. They have to manage their disease on a daily basis for many years. Treatment with insulin poses problems in daily activities and social life, and can be a psychological burden. Since insulin lispro and its mixtures can be administered just before meals, it is expected to reduce the problems most users of RHI experience daily. 108
Footnotes
Disclosure
This manuscript has been read and approved by all authors. This paper is unique and is not under consideration by any other publication and has not been published elsewhere. The authors and peer reviewers of this paper report no conflicts of interest. The authors confirm that they have permission to reproduce any copyrighted material.
