Abstract
Published analyses have noted elevated alanine aminotransferase (ALT) activity in patients taking up to 4000 mg/day of acetaminophen. Data from 3 osteoarthritis trials of acetaminophen 3900–4000 mg/day in which ALT and aspartate aminotransferase (AST) were recorded within approximately 2 weeks of therapy initiation were retrospectively analyzed. Patients with baseline ALT or AST above the upper limit of the reference range (ULRR) were excluded. Among 466 patients, 376 (80.7%) had no ALT elevations within approximately 2 weeks of treatment initiation. Elevations >1.5 and >3.0 times ULRR occurred in 4.5% and 0.9% of patients, respectively. Elevations were transient as most resolved (72.9%) or declined (22.4%) with continued treatment beyond 2 weeks. Within approximately 2 weeks of therapy initiation, no patient had ALT > 5 times ULRR or ALT > 3 times ULRR combined with bilirubin >2 times ULRR. ALT elevations were transient and asymptomatic; almost all resolved or declined while on continued therapy and appear not to be clinically important.
Introduction
Osteoarthritis (OA) is the most common form of arthritis in the United States and is estimated to affect approximately 27 million patients. 1 OA is a leading cause of disability and is associated with substantial activity limitations, reduced quality of life, and high health-care costs. 2 Based on its efficacy and well-established safety profile, the European League Against Rheumatism (EULAR) states that acetaminophen is the first choice oral analgesic for hand OA 3 and is effective in relieving pain arising from OA at any site. 4 The OA Research Society International (OARSI) recommends and the American College of Rheumatology (ACR) conditionally recommends acetaminophen for the initial management of knee and hip OA.5,6
Clinical studies of patients with OA pain evaluating the use of acetaminophen for up to 12 months at doses up to 4000 mg/day have demonstrated efficacy with a favorable clinical safety profile.7–12 However, there have been reports of alanine aminotransferase (ALT) elevations >3 times the upper limit of the reference range (ULRR) among patients with OA receiving acetaminophen 3900–4000 mg/day for up to 12 months. 13 In almost all of these subjects, the ALT elevations either resolved or were declining in subsequent assessments during continued therapy. 13 Watkins and colleagues reported ALT elevations >3 times the ULRR in healthy volunteers who were given daily doses of acetaminophen 4000 mg alone for up to 2 weeks. 14 These ALT elevations continued for up to several days after discontinuing treatment and subsequently declined. In response to the data published by Watkins and colleagues, ALT activity data from McNeil Consumer Healthcare (McNeil)-sponsored OA clinical trials were retrospectively analyzed to assess the prevalence and magnitude of ALT elevations within approximately 2 weeks of treatment initiation with acetaminophen 3900–4000 mg/day and to determine the rates of declining or resolving ALT activity with continued treatment beyond 2 weeks.
Methods
Nine randomized, controlled McNeil-conducted clinical trials were identified in which at least one of the treatments was acetaminophen 3900 – 4000 mg/day alone and the primary objective was evaluating the efficacy of acetaminophen for the relief of OA involving the hand, knee, hip, shoulder, or cervical or lumbar vertebrae. If qualifying with OA of the knee, patients had to have knee pain and radiographic osteophytes, and fulfill at least one of the following 3 criteria: experienced morning stiffness <30 minutes in duration, experienced crepitus on motion, or be at least 40 years of age. 15 If qualifying with OA of the hip, patients must have had hip pain, radiographic femoral and/or acetabular osteophytes, and radiographic joint-space narrowing as established by ACR criteria for idiopathic OA of the hip. 16 If qualifying for OA of the lumbar or cervical vertebrae, hand, or shoulder, patients had to have pain of moderate to severe intensity at rest or on motion, and radiographic evidence of the changes of osteoarthritis, including osteophytes and joint space narrowing. Patients must have had pain in 4 or more interphalangeal joints on the same hand, as well as radiographic evidence of the changes of osteoarthritis, including osteophytes and joint space narrowing, if qualifying with hand OA. 17 Additionally, tenderness, pressure, swelling, or heat could have been present in the involved joint/s. The studies were conducted in accordance with the ethical principles that have their origin in the Declaration of Helsinki. The protocol and amendments received ethics committee review and/or institutional review board approval before study initiation. A total of 1530 patients with OA were treated with long-term acetaminophen therapy lasting 4 weeks to 12 months in these 9 studies. This current analysis includes only those studies in which OA patients were treated with acetaminophen 3900–4000 mg/day and in which an initial on-treatment ALT activity measurement was recorded within 2 weeks of treatment initiation. A total of 3 studies met these criteria and are included in this analysis; 6 studies were excluded because they only included ALT activity measurements at baseline, or no ALT activity tests were performed within approximately the first 2 weeks of acetaminophen treatment. Data from 158 patients who were randomly allocated to OA treatment with less than 3900 mg/day of acetaminophen in these 3 studies were not included in this analysis. In the 3 studies that are included in this analysis, assessments of ALT activity were scheduled at weeks 0, 1, and 2 in 1 study and at weeks 0 and 2 in 2 studies (Table 1). The actual timing of the week 1 and week 2 visits ranged from 3–23 days after acetaminophen treatment initiation. Patients with ALT or AST activity >ULRR before receiving study drug were excluded from the current analysis, as were patients who had no ALT activity measurements while on treatment. Case report forms were available and reviewed as appropriate.
McNeil studies of patients with OA treated with acetaminophen alone and ALT measurements at baseline and within 2 weeks after initiation of treatment.

Dose excluded from analysis because it is below the labeled maximum daily dose of acetaminophen;
Novartis Consumer health, Parsippany, NJ, USA.
This analysis of aminotransferase data evaluates only ALT activity because ALT levels are primarily derived from the liver. 18 To facilitate comparisons of ALT data from different laboratories with varying reference ranges, ALT activity was expressed as multiples of the ULRR. The ULRR for ALT varied across studies from 30 IU/L to 70 IU/L. The ULRR for bilirubin was 1.2 mg/dL (21.0 μmol/L) in all 3 studies. Bilirubin results were also expressed as multiples of the ULRR in this analysis.
Stratification of ALT elevations and clinical importance
The clinical importance of varying levels of transient and asymptomatic ALT alterations has not been established. A US Food and Drug Administration (FDA) working group stated that, although any ALT above the ULRR deserves attention, low-level elevations (≤3 times the ULRR) are common in untreated and placebo-treated patients and are not considered informative for predicting drug-induced liver toxicity. 19 Investigators have noted that isolated elevations of ALT (even up to 10 times the ULRR), in the absence of other laboratory abnormalities, are not specific for liver injury and are poor predictors of serious liver injury or disease.20,21
Consistent with other analyses of aminotransferase activity,22–24 in this analysis, maximum ALT activity measurements for each patient were stratified by degree of elevation into the following categories: >ULRR, >1.5 times the ULRR, >3 times the ULRR, and >5 times the ULRR. The number and percentage of patients in each category were calculated by treatment within each study, as well as for all studies combined. The combination of ALT > 3 times the ULRR and bilirubin >2 times the ULRR (ie, Hy's Law) was captured in another category.20,25 It has been postulated that the combination of ALT > 3 times the ULRR and bilirubin >2 times the ULRR, accompanied by clinical data, better characterizes settings in which anomalies in liver test measurements are potentially clinically important.26,27
Definition of ALT resolution
Among patients who experienced an ALT elevation within approximately 2 weeks of initiating treatment with acetaminophen, all subsequent ALT activity was investigated to determine whether these elevations resolved or were in the process of declining with continued acetaminophen therapy beyond 2 weeks. ALT elevations were considered to have resolved “on treatment” if the ALT activity returned to ≤ULRR at the final blood draw, which could have occurred as late as 2 days after the last dose of the study drug. If the ALT activity at the final blood draw was lower than the maximum observed ALT level but >ULRR, the ALT elevation was considered to be declining. The number and percentage of patients who had ALT elevations that either resolved or were declining while on treatment with acetaminophen were summarized and pooled across studies.
Clinical assessment of adverse events
The adverse event data were retrospectively evaluated to determine if any patients experienced on-treatment adverse events that might suggest hepatic effects (anorexia, nausea, vomiting, abdominal pain, dyspepsia, gastroenteritis, gastritis, jaundice). Adverse events, using the preferred terms from the original coding dictionary for these symptoms, were pooled across studies for patients taking acetaminophen 3900–4000 mg/day. The percentage of patients who had any adverse events that might suggest hepatic effects among those who had ALT elevations >ULRR within approximately 2 weeks of treatment initiation were compared with the percentage of patients who had these adverse events in the patient group with ALT activity ≤ULRR. A similar comparison was made for patients with ALT > 1.5 times the ULRR.
Results
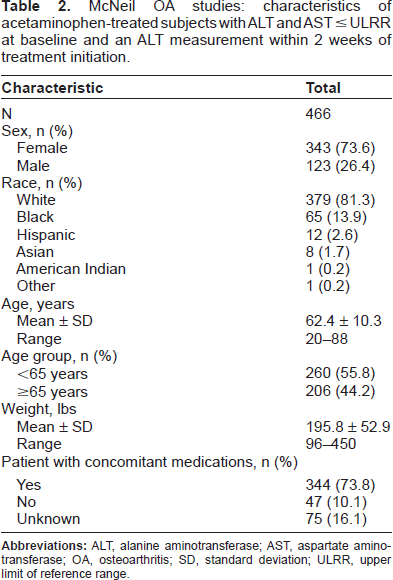
Table 1 summarizes the selection criteria, treatment durations, and scheduled timing of laboratory tests for the 3 studies that met the inclusion criteria. In these 3 studies, 466 patients received acetaminophen 3900–4000 mg/day, had baseline measurements of AST and ALT ≤ ULRR, and had ALT activity recorded within approximately 2 weeks of initiating acetaminophen treatment. The demographic profiles of the 466 patients in these 3 studies who met the inclusion criteria are summarized in Table 2. The majority of patients were female (73.6%) and white (81.3%). The mean age of these patients was 62.4 years and 44.2% of patients were ≥65 years of age. The majority of patients used concomitant medications (73.8%) during the study period. Of the 530 ALT measurements evaluated for these patients, 343 (64.7%) values were obtained between Days 14 and 16. Figure 1 shows the disposition of the patients who received acetaminophen. The incidences of ALT elevations identified within approximately 2 weeks of treatment initiation, stratified by magnitude, are summarized for patients receiving acetaminophen and comparators (ie, placebo or aspirin) in Table 3.
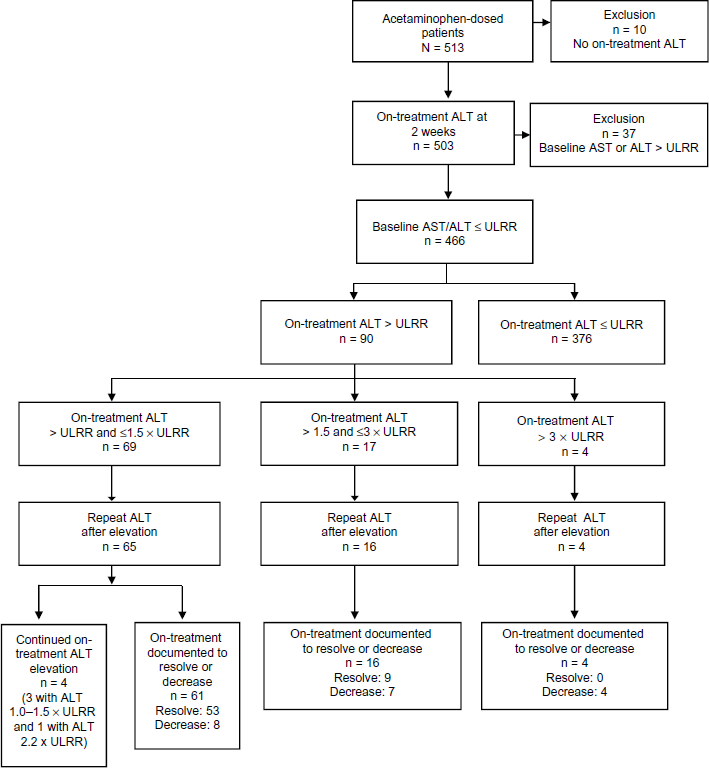
Disposition of OA patients who received acetaminophen.
McNeil OA studies: characteristics of acetaminophen-treated subjects with ALT and AST ≤ ULRR at baseline and an ALT measurement within 2 weeks of treatment initiation.
McNeil OA studies: ALT within approximately the initial 2 weeks of treatment by study and treatment for patients with ALT and AST ≤ ULRR at baseline.
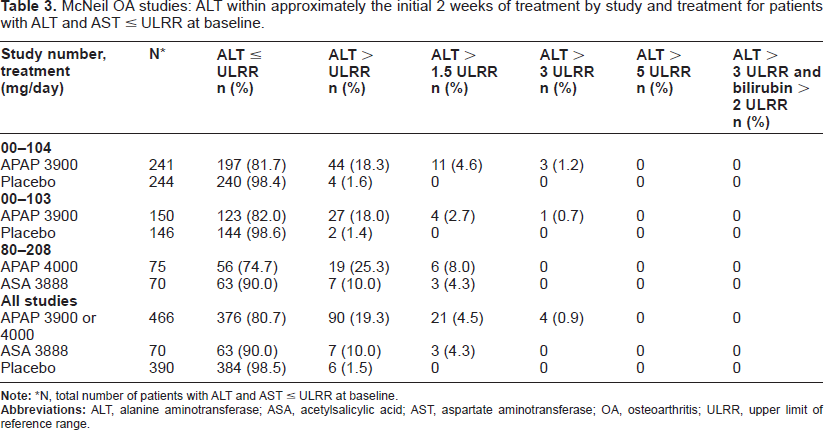
N, total number of patients with ALT and AST ≤ ULRR at baseline.
Of the 466 patients who met the criteria for this analysis, 376 (80.7%) patients had no ALT activity >ULRR within approximately 2 weeks of treatment initiation. A total of 69 (14.8%) patients had ALT activity >ULRR but ≤1.5 times the ULRR within approximately 2 weeks of treatment initiation. 21 of 466 patients (4.5%) had ALT activity >1.5 times the ULRR within approximately 2 weeks of acetaminophen therapy initiation; 17 (3.6%) patients had ALT activity >1.5 times but ≤3 times the ULRR and 4 (0.9%) patients had ALT activity >3 times the ULRR. Within approximately 2 weeks of acetaminophen treatment initiation, no patient had ALT activity >5 times the ULRR or ALT activity >3 times the ULRR with an accompanying bilirubin >2 times the ULRR.
Repeat on-treatment ALT measurements were available for 85 of 90 patients who experienced ALT elevations within approximately 2 weeks of initiating acetaminophen therapy. Of these 85 patients, 81 (95.3%) had either a documented resolution (72.9%, 62/85) or declining ALT activity (22.4%, 19/85) with continued acetaminophen treatment ranging from 22 to 177 days in duration. 4 patients (4.8%, 4/85), all of whom had ALT activity 1.0 to 1.5 times the ULRR within approximately 2 weeks of acetaminophen therapy initiation, had an ALT elevation that was not documented to resolve or decline during continued treatment. 3 of these 4 patients had ALT activity that remained between 1.0 and 1.5 times the ULRR during continued acetaminophen therapy lasting 52, 58, and 77 days, respectively. The remaining patient had a final on-treatment ALT measurement 2.2 times the ULRR after 85 days of acetaminophen treatment. For patients who had ALT elevations >1.5 times the ULRR within approximately 2 weeks of treatment initiation, follow-up ALT activity was available for 20 of 21, with all 20 patients having documented resolution or a declining ALT while continuing therapy for the remainder of the study.
Figure 2 illustrates the time course of ALT activity in each of the 17 patients who had an on-treatment ALT > 1.5 times but ≤3 times the ULRR within approximately 2 weeks of treatment initiation. Repeat ALT measurements were available for 16 of these 17 patients. ALT activity either resolved or declined in all 16 patients with continued acetaminophen therapy beyond 2 weeks. The other patient is also illustrated, but did not have repeat ALT activity testing. Figure 3 illustrates the time course of ALT activity in each of the 4 acetaminophen-treated patients with on-treatment ALT activity >3 times the ULRR within approximately 2 weeks of treatment initiation. Subsequent on-treatment ALT activity was available for all 4 patients during continued acetaminophen treatment beyond 2 weeks. ALT activity declined with continued acetaminophen treatment in all 4 patients. These data depict the transient nature of the ALT elevations. For 1 patient (case 21), after 2 additional weeks of acetaminophen treatment, ALT activity peaked at 9 times ULRR without an elevation of bilirubin. The ALT elevation decreased somewhat while on treatment and resolved after treatment was discontinued. No patient experienced hepatotoxicity or hepatic failure during continued acetaminophen treatment beyond 2 weeks.
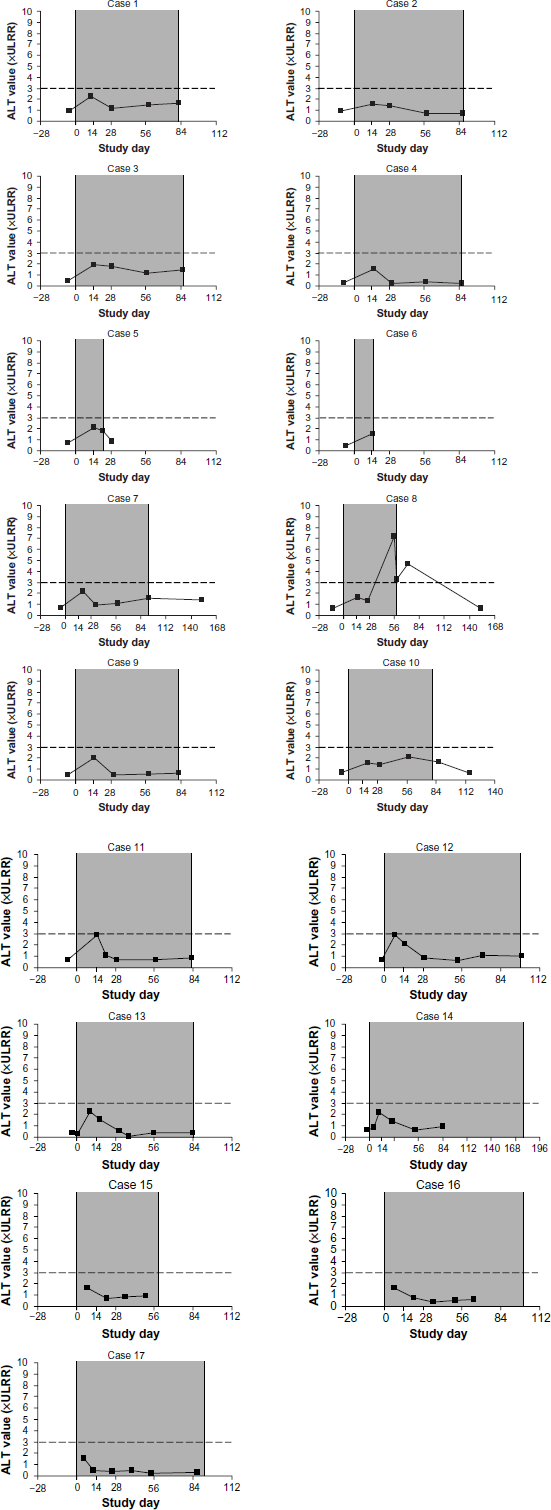
Time course of ALT activity in 17 acetaminophen-treated OA patients with ALT > 1.5 times but ≤3 times the ULRR during the initial 2 weeks of treatment (shaded area indicates time on treatment).
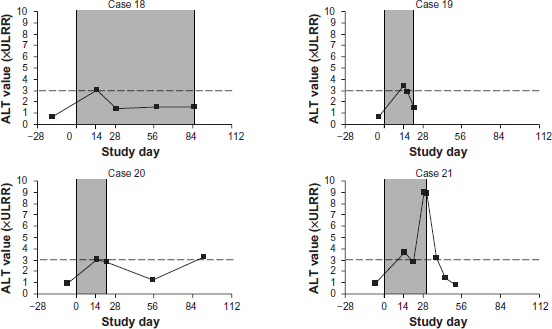
Time course of ALT activity in 4 acetaminophen-treated OA patients with ALT > 3 times the ULRR during the initial 2 weeks of treatment (shaded area indicates time on treatment).
The incidences of adverse events that might suggest hepatic effects (anorexia, nausea, vomiting, abdominal pain, dyspepsia, gastroenteritis, gastritis, jaundice) were similar for patients with an ALT elevation within approximately 2 weeks of acetaminophen treatment initiation (12.2%, 11/90) compared with patients who did not have an ALT elevation within this time period (12.8%, 48/376). The incidences of adverse events of possible hepatic origin were also similar for patients with an ALT elevation >1.5 times the ULRR within approximately 2 weeks of treatment initiation (14.3%, 3/21), compared with patients who did not have an ALT elevation >1.5 times the ULRR within approximately 2 weeks of treatment initiation (12.6%, 56/445).
Discussion
No ALT elevations occurred in 80.7% of OA patients within approximately 2 weeks of initiating treatment with acetaminophen 3900–4000 mg/day and fewer than 1% of patients treated with acetaminophen had ALT elevations >3 times the ULRR during the same period. These ALT elevations were transient and resolved or declined in almost all of the patients for whom follow-up data were available during continued acetaminophen treatment beyond 2 weeks. The incidences of adverse events of possible hepatic origin were similar for patients with ALT elevations >1.5 times the ULRR compared with patients who did not have ALT elevations >1.5 times the ULRR. Adverse events of possible hepatic origin were similar in patients with or without ALT elevations, indicating that the elevations were not clinically important.
The 2-week data presented in this analysis are a subset of a larger body of evidence, consisting of 9 OA studies that demonstrate the safety of longer-term acetaminophen use. 13 Among these 9 McNeil-conducted studies involving 1530 patients treated with acetaminophen 1950–4000 mg/day for 4 weeks to 12 months, there were no cases of hepatotoxicity or hepatic failure. 13 7 of these studies included patients who had ALT and AST measurements obtained at baseline and an ALT measurement at an additional visit after initiating therapy (n = 1039). On-treatment ALT activity >1.5 times the ULRR was observed in 44 (4.2%) patients receiving acetaminophen 1950–4000 mg/day for 4 weeks to 12 months in these 7 studies. 13
Elevations in ALT activity can be caused by a variety of factors, including day-to-day ALT variability of approximately 10% to 30% in healthy volunteers and individuals with stable liver disease. It has also been demonstrated that time of day, gender, race, age, and body mass index can influence ALT activity.18,28–30 A meta-analysis of 13 Phase I studies reported ALT elevations in 22% of healthy male volunteers receiving placebo treatment during in-house studies. 31 Researchers note that dietary factors and the stress of hospitalization may influence aminotransferase activities in healthy subjects.31–33 Compared with subjects on a standard diet, ALT values were 38% and 143% higher in healthy volunteers housed in a controlled clinic environment who consumed a high-fat high-calorie diet or a high-carbohydrate high-calorie diet, respectively. 32 In the National Health and Nutrition Examination Survey (NHANES) conducted from 1999–2002, the overall prevalence of elevated ALT activity in the general population was 8.9%. Higher rates of ALT elevations were seen in males compared with females, Mexican Americans versus non-Hispanic whites, and in adults 30–39 years of age compared with elderly individuals (≥70 years). 29
ALT elevations have been reported in healthy volunteers taking acetaminophen 4000 mg/day during a 2-week in-patient study. 14 In a short-term, single-blind study in young healthy volunteers confined to clinical pharmacology units, Watkins and colleagues observed maximum ALT values > 3 times the ULRR in 38% of patients within 2 weeks of initiating therapy with acetaminophen 4000 mg/day. 14 All ALT elevations >3 times the ULRR resolved after treatment cessation, with the exception of one patient who was lost to follow-up. All patients with these ALT elevations were asymptomatic and there were no abnormalities in total bilirubin or alkaline phosphatase. The authors acknowledged that acetaminophen has an outstanding safety record when taken as directed and that long-term therapy with acetaminophen 4000 mg/day has proven to be safe. Watkins and colleagues postulated that the ALT elevations observed in their study would have likely resolved with continued acetaminophen treatment. 14 At present, the mechanisms for these transient elevations of ALT are unknown.
The incidence of ALT elevations reported by Watkins and colleagues may be higher than those seen in our analysis because of the environmental conditions or heterogeneity of the patient populations in these 2 analyses. While Watkins and colleagues confined patients in clinical pharmacology units, the 3 McNeil OA studies were conducted on an outpatient basis. Whereas their study population was relatively young (mean age, 33.8 years), male (78%), and Hispanic (57%), ours was predominantly elderly (mean age, 62.4 years), female (73.6%), and white (81.3%). Watkins and colleagues reported in their analysis that the relative risk of maximum ALT > 3 times the ULRR was higher for Hispanics versus non-Hispanics (1.9; 95% CI 1.1–3.3) taking either acetaminophen monotherapy or combination therapy. 14 Because of age or cultural diversity, there may have been a higher percentage of patients in the analysis conducted by Watkins and colleagues who were acetaminophen-naïve or without a history of recurrent acetaminophen use. These individuals may have exhibited an adaptive response to acetaminophen therapy. This adaptive phenomenon has been seen in patients taking tacrine, who had ALT elevations during initial tacrine therapy and lower peak ALT levels when tacrine treatment was reintroduced, even when rechallenge was performed many weeks after cessation. 34 Asymptomatic elevations in aminotransferase activity are commonly associated with a variety of medications, including aspirin, tacrine, and statins.22–24,26,35–37
The NHANES III Second Examination study found that, in some instances, initially elevated ALT activity returned to within normal limits upon subsequent retesting. 38 This substudy assessed the magnitude of intraindividual variability in liver test results and evaluated the proportion of adults with persistently elevated activity levels after 1 abnormal result. In the NHANES III First Examination study, 5.9% of participants had elevated ALT activity at the first testing. However, when retested a mean 17.5 days later, more than 30% of the individuals with initially elevated ALT activity levels returned to normal activity. 38 The high intraindividual variability in ALT activity suggests that clinical decision-making should not be based on a single elevated ALT measurement and that asymptomatic patients with an abnormal ALT measurement should be retested. 38 When liver injury is suspected, additional clinical and laboratory data can be obtained to ascertain a differential diagnosis of the cause.
Since elevations in ALT are documented to occur in the general population, can be caused by a variety of factors, and have been shown to resolve spontaneously in some individuals, the relevance of isolated ALT elevations during drug therapy is uncertain. Given the variability of ALT elevations across populations, it is apparent that elevated transaminases alone do not necessarily indicate liver damage. Elevations in bilirubin indicate an inability of the liver to perform its normal functions, such as the clearance of bilirubin from plasma into bile. 22 The combined finding of aminotransferase abnormalities and impaired hepatic function (bilirubin elevation >2 times the ULRR), in the absence of biliary obstruction or some other explanation of injury (eg, viral hepatitis, alcohol hepatitis), indicates a signal of potential drug-induced hepatotoxicity. 26 None of the patients in the current analysis had an ALT > 3 times the ULRR and a serum bilirubin >2 times the ULRR.
Our analysis is the first to evaluate ALT activity within the initial few weeks of acetaminophen therapy and the ALT pattern while remaining on continued acetaminophen therapy in controlled trials of patients with OA pain. The low-level transient ALT elevations observed in these OA trials are quite different from the clinical patterns seen during acetaminophen overdose. Acetaminophen-induced hepatic injury associated with overdose is characterized by gross elevations in ALT activity (ie, >1000 IU/L or approximately 25 times the ULRR) and clinical symptoms, including nausea and vomiting.39,40 In the 3 studies that were evaluated for this analysis, no patient had ALT activity > 5 times the ULRR within approximately 2 weeks of acetaminophen treatment initiation.
The retrospective design of our analysis is a methodologic limitation. The studies from which ALT data were obtained were designed to determine the efficacy of acetaminophen treatment in patients with OA. Allowable concomitant medications were recorded for patients but were not used in assessing possible causes of ALT elevations. Laboratory test results were not a primary outcome measure. Since multiple serial ALT values were not obtained at specific time points, the temporal pattern of ALT elevations within the first few weeks of acetaminophen treatment cannot be fully characterized. In addition, there were differences between these studies with respect to inclusion/ exclusion criteria, use of concomitant medications, timing and frequency of blood collections, and the use of multiple clinical laboratories. There are data to suggest that the ULRR for serum transaminases may vary greatly depending on the clinical laboratory performing the analysis. 41 Therefore, the choice of laboratory may alter the proportion of patients with an elevation in ALT activity and could potentially cause misclassification of patients and misinterpretation of data. Also, there was no standardized process for gathering additional clinical and laboratory data to make a differential diagnosis of the cause of elevated ALT activity in the 3 studies.
This analysis of 3 clinical studies involving 466 patients with OA taking acetaminophen 3900–4000 mg/day demonstrates that asymptomatic low-level ALT elevations occurred within approximately 2 weeks of therapy initiation in approximately 20% of patients. These elevations resolved or declined with continued doses of acetaminophen 3900–4000 mg/day beyond 2 weeks in almost all of the patients. The incidences of adverse events of possible hepatic origin were similar for patients with an ALT elevation >1.5 times the ULRR within approximately 2 weeks of acetaminophen therapy initiation compared with patients who did not have ALT activity >1.5 times the ULRR within the same time period. These low-level transient ALT elevations were not associated with signs or symptoms of liver toxicity and, as such, were not clinically important. When taken at daily doses up to 4000 mg, acetaminophen is a safe and efficacious analgesic and antipyretic.
Author Contributions
Edwin Kuffner and Joseph M Lynch take responsibility for the integrity of the work as a whole, from inception to finished article. Conception and design: ART, EK, JML, JB, KMC, BAZ. Acquisition of data: KMC, BAZ, KBF, LAB. Analysis and interpretation of data/statistical expertise: KMC, BAZ. Drafting of manuscript: ART, EK, JML, JB, KMC, BAZ, KBF, LAB. Critical revision for important intellectual contribution: ART, EK, JML, JB, KMC, BAZ, KBF, LAB. All authors were involved in the drafting of the article and revising it critically for important intellectual content. All authors approved the final version to be submitted for publication.
Funding
The individual clinical studies, analyses of clinical data, and preparation of the manuscript were funded by McNeil Consumer Healthcare, Fort Washington, PA.
Competing Interests
EK, JML, BAZ and LAB are employees of McNeil Consumer Healthcare. KMC was an employee of McNeil Consumer Healthcare at the time the analyses were conducted and became an employee of J&J PRD during preparation of the manuscript. JB was an employee of McNeil Consumer Healthcare at the time the analyses were conducted and the manuscript was prepared. ART and KBF receive consulting fees from McNeil Consumer Healthcare.
Disclosures and Ethics
As a requirement of publication the authors have provided signed confirmation of their compliance with ethical and legal obligations including but not limited to compliance with ICMJE authorship and competing interests guidelines, that the article is neither under consideration for publication nor published elsewhere, of their compliance with legal and ethical guidelines concerning human and animal research participants (if applicable), and that permission has been obtained for reproduction of any copyrighted material. This article was subject to blind, independent, expert peer review. The reviewers reported no competing interests.
Footnotes
Acknowledgements
Editorial and technical assistance preparing the draft manuscript was provided by Rosanne Degnan, who was employed by Scientific Connexions, a division of KnowledgePoint360 Group LLC, at the time of this work. Editorial and technical assistance preparing the manuscript for submission were provided by Kathleen Boyle of KE Boyle Consultants, LLC.
