Abstract
Generalized anxiety disorder (GAD) is a common disorder that is chronic, disabling, and often goes undiagnosed. Currently, four distinct classes of medications have demonstrated efficacy in the treatment of GAD: benzodiazepines, serotonin and/or norepinephrine reuptake inhibitors (SSRIs/SNRIs), histamine H1 receptor blockers (hydroxyzine), and pregabalin. Pregabalin acts via binding to an α2-δ subunit presynaptic membrane protein that inhibits neurotransmitter release in excited neurons. Pregabalin is renally excreted and undergoes minimal (<2%) hepatic metabolism, thus limiting the risk of drug–drug interactions. The efficacy of pregabalin for the treatment of GAD has been established based on the results of 8 double-blind, placebo-controlled, short-term led trials, and one 6-month relapse prevention study. The current review summarizes data showing that pregabalin has a significantly different safety profile from the benzodiazepines (eg, less sedation, less cognitive and psychomotor impairment, less risk of dependence and withdrawal), and SSRI/SNRI anxiolytics (eg, less gastrointestinal side effects and sexual dysfunction). The review also summarizes efficacy data showing that pregabalin is a broad spectrum anxiolytic that with a speed of onset similar to the benzodiazepines.
Introduction
GAD is a common disorder that is chronic, disabling, and often goes undiagnosed. Approximately 1 out of every 10 patients evaluated in the primary care practice setting meets DSM-IV criteria for GAD.1–3 Despite its high prevalence, GAD frequently goes undiagnosed.3,4
Over more than a decade of follow-up, GAD has been shown to have a lower recovery rate than major depressive disorder (58% vs. 73%), 5 and among patients who do recover, the rate of recurrence is high (45%). 5 GAD is also associated with work and social impairment at levels comparable with those observed among MDD patients. 6
As summarized in a meta-analysis that included clinical trials up to 2005, 7 five classes of drugs have effect sizes (ES) that have been shown to be significantly greater than placebo: the α2-δ pregabalin (ES, 0.50), the piperazine H1 receptor antagonist hydroxyzine (ES, 0.45), the SNRI venlafaxine-XR (ES, 0.42), benzodiazepines (ES, 0.38), and the SSRIs citalopram, escitalopram, paroxetine, and sertraline (ES, 0.36). The effect size (0.17) of the 5-HT1A partial agonist buspirone was not found to be significantly different than placebo. Since the publication of this meta-analysis, the SNRI duloxetine has also been approved for the treatment of GAD, 8 however, based on available data, its effect size appears to be smaller than venlafaxine-XR.
The last review of pregabalin in the treatment of GAD was published 3 years ago. 9 The current review will provide an update, primarily summarizing data that have been published since that review.
Mechanism of Action
Currently available treatments for GAD act via one of four mechanisms: either by increasing inhibitory activity in the benzodiazepine-GABAergic receptor complex (benzodiazepines); by modulating monoaminergic neurotransmission, typically via inhibition of serotonin and/or norepinephrine reuptake proteins (SSRIs/SNRIs); by blocking the neuronal excitatory effects of histamine at H1 receptors (hydroxyzine); or by binding to an α2-δ subunit presynaptic membrane protein that inhibits neurotransmitter release in excited CNS neurons (pregabalin).
At the time of the previous review it was known that pregabalin, despite its name, had no activity at benzodiazepine or GABA receptor sites. Evidence from mutant and knockout mouse models has now conclusively shown that α2-8 binding (specifically to the type 1 Cav α2-δ protein) is the mechanism of action of pregabalin.10–12 In animal models, a crush injury to a peripheral nerve (a neuropathic pain model) results in an upregulation α2-δ binding sites. This upregulation is significantly correlated with the onset of allodynia. 13 It is not yet known whether a similar stress-related upregulation in α2-δ binding sites also occurs in the neural substrates of anxiety in patients who develop GAD. More research is needed to further characterize the activity of pregabalin on these neural substrates.
Metabolism, Pharmacokinetic Profile
Pregabalin is rapidly absorbed (Tmax, 1 h) and has high bioavailability (>90%). 14 When taken with food, the Cmax is decreased by 25%–30%, and the Tmax is delayed to ~2.5 h, but the elimination half-life is unchanged (~6.3 h), and the overall AUC remains the same. Pregabalin is a highly hydrophilic compound that crosses the blood brain barrier via a specialized transporter system (system L). 11
Since pregabalin is predominantly excreted unchanged in the urine, undergoes minimal hepatic metabolism in humans (<2% of a dose recovered in urine as metabolites), does not inhibit drug metabolism in vitro, and is not bound to plasma proteins, it is unlikely to produce, or be subject to, pharmacokinetic drug-drug interactions. However, it should be noted that pregabalin is pharmacodynamically additive in its impairment of cognitive and psychomotor function when administered with CNS-depressant drugs such as oxycodone, benzodiazepines, or alcohol.
Due to its renal elimination, dose adjustments are required in patients with renal impairment who are treated with pregabalin. A starting-to-maximal dose of 75–300 mg/day is recommended in patients with a creatinine clearance of 30–60 ml/min; and a dose range of 50–150 mg/day (once or twice daily) in patients with a creatinine clearance of 15–30 ml/min. Dose adjustments are not required in patients with moderate-to-severe hepatic impairment. Gender and age have no significant effect on plasma concentrations.
Clinical studies
Methodology
The efficacy of pregabalin for the treatment of GAD has been established based on the results of 8 short-term (4–8 week), randomized, double-blind, placebo-controlled trials15–21 and one 6-month relapse prevention study. 22 All patients met DSM-IV criteria for GAD, with a minimum HAM-A severity score of 20. The mean HAM-A severity score at baseline across all studies was approximately 26, which indicates that at least 50% of patients were experiencing severe anxiety. It should be noted that a concurrent diagnosis of major depressive disorder was reason for exclusion, though a significant proportion of patients reported mild-to-moderate levels of depressive symptoms.
Four of the short-term trials of pregabalin included a high potency benzodiazepine as a comparator (lorazepam or alprazolam), while one early trial included venlafaxine, and one more recent trial included venlafaxine-XR as comparators.19,21 One of the eight short-term trials evaluated the efficacy of pregabalin in an elderly patient population. 20
The therapeutic dosage range for pregabalin is 150–600 mg/day, typically administered in two divided doses. All but 2 of the 8 short-term trials19,21 were fixed-dose studies, which permitted a dose-response analysis to be performed. 23 The results of this analysis indicated that response rates were lower on the 150 mg daily dose, but that there was no dose-response effect across the recommended dosing range of 300–600 mg/day.
The use of fixed-dose studies, which required some patients to titrate to higher doses regardless of initial improvement, very likely resulted in a higher incidence of adverse events, and higher discontinuation rates. Subsequent uncontrolled trials conducted in clinical practice settings have reported high levels of efficacy and tolerability using lower doses, in the range of 150–300 mg. 24
Efficacy
The efficacy of pregabalin in GAD, based on data available up through January, 2006 has previously been summarized by one of the authors. 19 This review summarized the results of 6 short-term, placebo-controlled GAD treatment studies. In 5 of the 6 studies, pregabalin demonstrated significant efficacy compared to placebo. The sixth study was considered to be a failed study because neither pregabalin, nor lorazepam (the active comparator) achieved superiority over placebo. Since that date, a double-blind, placebo-controlled study evaluating the comparative efficacy of pregabalin and venlafaxine-XR has been reported. 21 The results of this study demonstrated significant efficacy in favor of pregabalin on both the HAM-A total score (the primary outcome), and on the psychic and somatic factor scores (Fig. 2). Improvement on venlafaxine-XR was not significantly different than placebo. This latter finding is consistent with the results of the largest previously reported double-blind, placebo-controlled comparison of venlafaxine-XR to diazepam. 25 Both the study by Kasper et al 21 and the study by Hackett et al 25 had similarly high placebo response rates (HAM-A change score, -11.7 for both studies), which has become an endemic problem in clinical trials in mood and anxiety disorders. Similar levels of improvement on venlafaxine-XR were also reported in the study by Kasper et al 21 (-12.0 on a mean dose of 155 mg) and in the study by Hackett et al 25 (-12.8 on a fixed dose of 150 mg; 14.0 on the 75 mg dose).
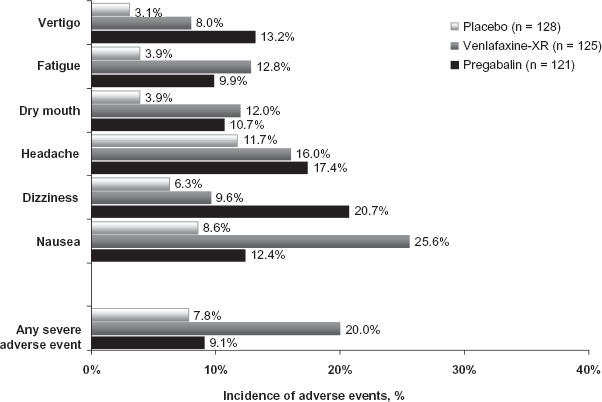
Treatment-emergent adverse events during 8 weeks of treatment for generalized anxiety disorder.
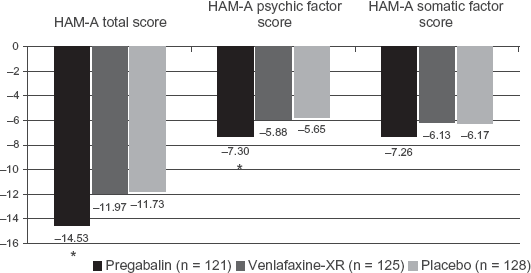
Mean change in HAM-A total, and psychic and somatic anxiety factor scores at LOCF-endpoint *P < 0.05. LOCF endpoint results based on ANCOVA.
Efficacy in psychic and somatic anxiety symptoms
DSM-IV criteria for GAD privilege psychic symptoms over somatic symptoms because of their greater discriminatory validity. Nonetheless, somatic symptoms are a prominent part of the clinical presentation of anxiety, and represent the chief complaint in 48% of individuals with GAD who present in the primary care setting. 3 Furthermore, somatic symptoms are typically (but not always) less responsive to treatment with SSRI and SNRI anxiolytics.26–28 In contrast, treatment with benzodiazepines is associated with more consistent improvement in somatic anxiety symptoms.
A pooled analysis of short-term studies was recently reported that evaluated the comparative efficacy of pregabalin and benzodiazepines in treating the full range of psychic and somatic symptoms. Treatment with pregabalin 300–450 mg/d was associated with significant improvement on 13 out of 14 HAM-A items (except intellectual), while treatment with pregabalin 600 mg was associated with significant improvement in 6 out of 7 psychic factor items (except intellectual), and 4 out of 7 somatic factor items (except somatic sensory, GU, autonomic; Fig. 3). Patients treated with high potency benzodiazepines demonstrated significant improvement in 3 out of 7 psychic factor items (except fears, intellectual, depressed mood, behavior at interview) and 2 out of 7 somatic factor items (except somatic sensory, respiratory, GI, GU, autonomic). These findings suggest that pregabalin has broad spectrum efficacy across the full range of psychic and somatic anxiety symptoms that is comparable, if not superior, to benzodiazepines.
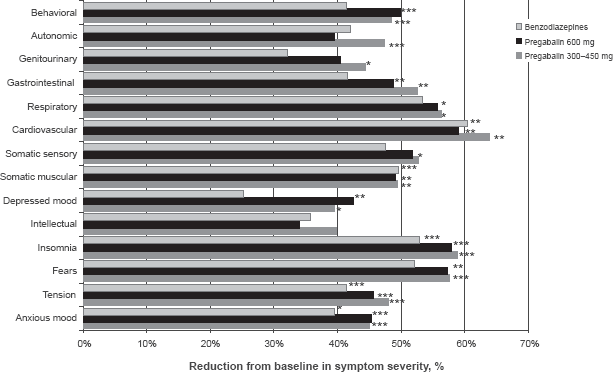
Percent reduction in HAM-A Item scores from baseline to 4-week LOCF-endpoint. Comparison with placebo:
P < 0.05;
Speed of onset of response
One of the most prominent efficacy differences between the benzodiazepines and SSRI and SNRI anxiolytics is speed of onset of treatment effect. In patients with GAD, significantly greater improvement in anxiety versus placebo emerges after 2–4 weeks of treatment with the SSRI/SNRI anxiolytics, but occurs by the end of the first week of treatment with benzodiazepines. Results from a double-blind comparator trial (Fig. 4) 21 demonstrate that significant improvement versus both placebo and venlafaxine-XR occurs by Day 4 on pregabalin in both the HAM-A total score and the HAM-A psychic and somatic factor scores. In this study, pregabalin was started at a dose of 300 mg/d, and venlafaxine-XR was started at 75 mg/d.
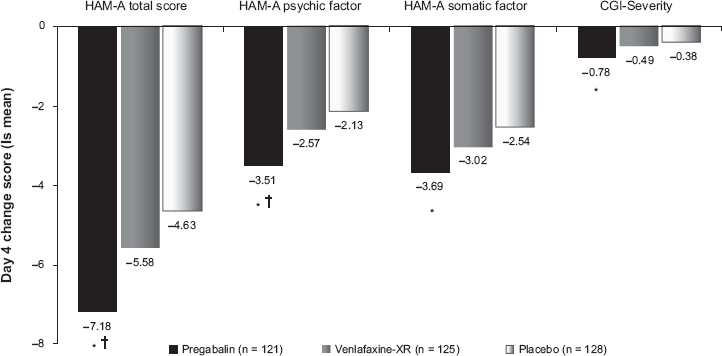
Change from baseline to day 4: results of MMRM analysis comparing pregabalin and venlafaxine-XR.
P < 0.01 vs. placebo;
Improvement in anxiety symptoms appears to be early and sustained in a large proportion of patients treated with pregabalin. In a pooled analysis of all Phase 2 and 3 clinical trials, treatment with pregabalin was associated with early and sustained improvement in anxiety symptoms that was fully comparable to the improvement observed on the high potency benzodiazepines lorazepam and alprazolam (Fig. 5). For both pregabalin and the benzodiazepines, more than 25% of patients met responder criteria by Week 1 (Fig. 5).
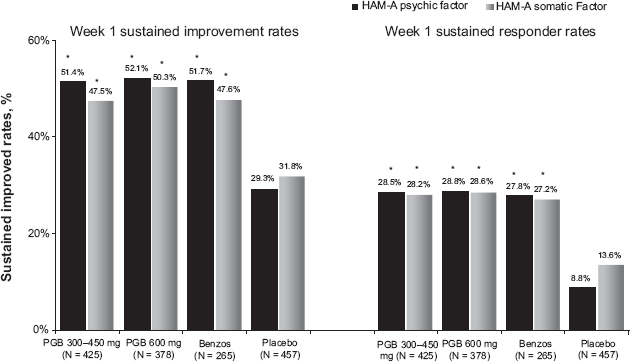
Proportion of patients achieving early-and-sustained improvement and response by week 1: HAM-A psychic and somatic anxiety factor scores from pooled study data. Early and sustained improvement criteria: ≥30% reduction in HAM-A factor scores by week 1, and sustained at that level or greater until endpoint; early and sustained response criteria: ≥50% reduction in HAM-A factor scores by week 1, and sustained at that level or greater until endpoint.
P < 0.001.
For most chronic illnesses, speed of onset of response is not a key efficacy parameter. However, GAD symptoms tend to exhibit notable fluctuations in severity, either due to environmental stressors or endogenous factors. As a consequence, a large proportion of patients feel the need to supplement SSRI and SNRI therapy with as-needed benzodiazepines. 29 A recent study, using a dental model of anxiety 30 suggests that a single low dose (150 mg) of pregabalin achieves moderate-to-marked improvement in anxiety within 2 hours post-dose that is comparable to 0.5 mg of alprazolam (in 59% vs. 61% of patients). As-needed use of benzodiazepines is complicated by well-characterized effects on memory, cognitive function, vigilance and attention, and psychomotor function that may result in impaired performance in complex tasks such as operating a motor vehicle. As summarized previously, data from a double-blind crossover study in healthy volunteers has confirmed that alprazolam is associated with significant cognitive and psychomotor impairment, but suggests that pregabalin has a more favorable cognitive and psychomotor profile that was minimally different from placebo. 31
Anxiety and insomnia
One of the key diagnostic criteria for GAD is sleep disturbance, 32 and not surprisingly, clinically significant insomnia occurs in at least 50% of patients diagnosed with GAD. 33 Conversely, the gender-adjusted odds ratio that a patient will have a diagnosis of GAD if insomnia is present is 7.0. 34
Compared to MDD, insomnia is less frequently a prodromal symptom for GAD, but has an onset concurrent with, or after, the onset of GAD in >70% of cases.35,36 This is consistent with the hypothesis that insomnia and GAD share, at least in part, a common pathophysiology related to hyperarousal. 37
Polysomnography has not identified consistent or pathognomonic findings in GAD. However, the most commonly reported findings are increased sleep latency, reduction in total sleep time and sleep efficiency, and a lower percent of slow wave (stage 3/4) sleep.33,38–40
Given their diffuse CNS depressing activity, benzodiazepines effectively treat both GAD and insomnia. However, their main effect on sleep architecture is a tendency to reduce restorative slow wave sleep. 41 In contrast, SSRI and SNRI anxiolytics typically increase REM latency, decrease REM density, and increase sleep fragmentation. 42 In a polysomnography study of healthy volunteers, pregabalin increased total sleep time and sleep efficiency. 43 In addition, pregabalin significantly increased slow wave sleep compared to alprazolam. 43 Comparative clinical studies are needed in patients with GAD to confirm the differential effects of pregabalin and benzodiazepines on sleep architecture.
The effect of pregabalin in treating insomnia in patients with a primary diagnosis of GAD has recently been evaluated using the validated Medical Outcome Study (MOS) sleep scale. 44 Treatment with pregabalin resulted in a significant endpoint improvement in the MOS sleep disturbance factor compared to placebo (Fig. 6). Despite a somewhat higher incidence of treatment-emergent sedation on pregabalin compared to venlafaxine-XR (9.1% vs. 4.8%), treatment with pregabalin was associated with numerically greater reduction in daytime sleepiness (Fig. 6).
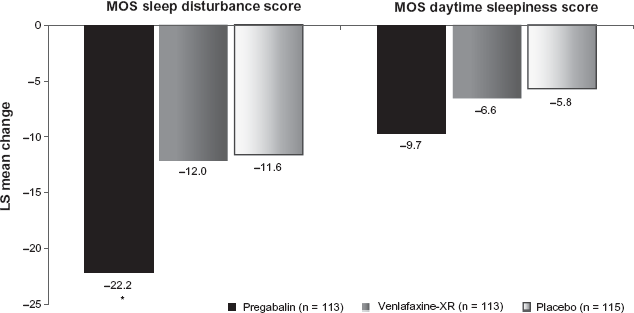
LOCF-endpoint change in MOS sleep disturbance and daytime sleepiness: comparison of pregabalin, venlafaxine-XR, and placebo.
P < 0.001.
A recent mediational analysis was performed in an attempt to evaluate the extent to which treatment with pregabalin had a direct effect on reducing insomnia, or an indirect effect that was mediated by an antecedent improvement in anxiety. The results of this mediational analysis indicated that 53% of the effect of pregabalin on sleep disturbance was due to a direct effect, and 47% was due to an indirect effect, mediated through reduction in anxiety symptom severity (Fig. 7). 45
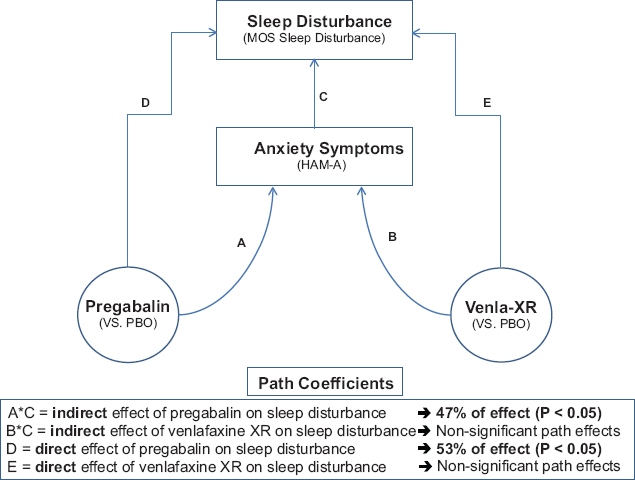
Is the effect of pregabalin on sleep a direct effect, or an indirect effect due to improvement in anxiety? Results of a mediational analysis.
Anxiety and functional GI symptoms
Moderate-to-severe gastrointestinal (GI) symptoms are commonly reported by patients diagnosed with GAD. 46 GI symptoms may include both upper GI (nausea, vomiting, globus, burning sensations, borborygmi, dyspepsia) and lower GI symptoms (constipation, abdominal fullness, gas pains). Patients diagnosed with irritable bowel syndrome (IBS) have been reported to have high rates of GAD comorbidity, in the range of 30%–50%. 47 However, the incidence of comorbid IBS in patients with a primary diagnosis of GAD has not been established.
Treatment-emergent worsening of GI symptoms is commonly reported during treatment with SSRI and SNRI anxiolytics. GI side effects have not been reported as commonly after benzodiazepine therapy. A recently published analysis 48 examined the comparative efficacy of pregabalin and benzodiazepines in treating GAD patients who presented with severe GI symptoms. The results of this analysis found that pregabalin significantly improved HAM-A total scores at endpoint in patients presenting with both high and low levels of GI symptoms (Fig. 8). The effect size (vs. placebo) was notably higher on pregabalin compared to the benzodiazepines. Similarly, treatment with pregabalin, but not benzodiazepines, was associated with significant improvement in the GI symptom item on the HAM-A (item #11). No patients treated with pregabalin reported treatment-emergent worsening of GI symptoms.
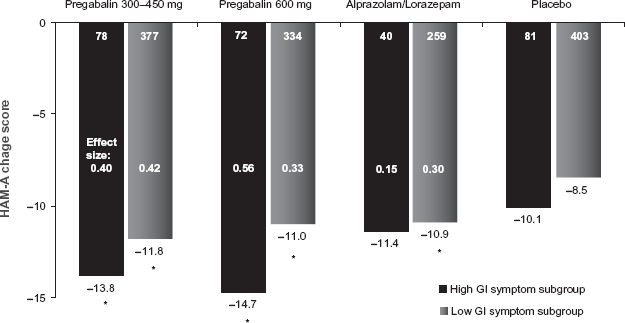
Mean change in HAM-A total score at LOCF-endpoint: comparison of patients presenting with high vs. low levels of gastrointestinal (GI) symptoms.
P < 0.01 vs. placebo.
Anxiety and pain
Chronic pain disorders such as neuropathic pain and fibromyalgia are relatively commonly occurring disorders with prevalence rates in the range of 1% (for neuropathic pain) to 2% (for fibromyalgia). 49 Anxiety frequently complicates the clinical presentation of chronic pain. For example, the odds ratio for an anxiety disorder diagnosis is 4.3 in patients with a diagnosis of fibromyalgia, and GAD occurs in at least 25% of cases.50–52 The presence of anxiety is associated with greater pain severity, functional impairment, and chronicity. 52 Pregabalin has demonstrated significant efficacy in multiple placebo controlled trials in treating both neuropathic pain and fibromyalgia.53–55 Unlike the results obtained for insomnia, a mediational analysis found that only a relatively small (25%) proportion of pain relief was an indirect effect attributable to antecedent improvement in anxiety and/or depressive symptoms. 56
GAD in the elderly
GAD has a later median age of onset (31 years) compared to other anxiety disorders, with 20% of individuals developing the disorder after the age of 50. 57 Furthermore, given the high rates of chronicity, GAD is more likely to persist into the sixth decade, resulting in an estimated prevalence rate in the range of 2%-7%. 58
Pregabalin has demonstrated significant efficacy in treating GAD in a large, double-blind, placebo-controlled trial, with 64% of patients achieving a response by Week 8. 20
Treatment of anxiety in the elderly is complicated by a greater likelihood of drug-drug interactions, due to higher rates of polypharmacy in this age group. Since pregabalin is primarily renally excreted (>90%), there are no CYP-related drug-drug interactions.
Treatment of GAD in the elderly is also complicated by a greater vulnerability to adverse events, especially sedation and cognitive-impairment. Treatment with benzodiazepines is associated with significant cognitive impairment and sedation in the elderly. In contrast, treatment of elderly patients with pregabalin was not associated with any impairment on a cognitive battery that included DSST, digit span, and the set test. 59 In the same elderly sample, sedation was reported as an adverse event in 13.0% of patients compared to 7.3% on placebo (NNH, 17.5). However the median duration was relatively short (16 days), and no patients rated their sedation as severe.
Safety
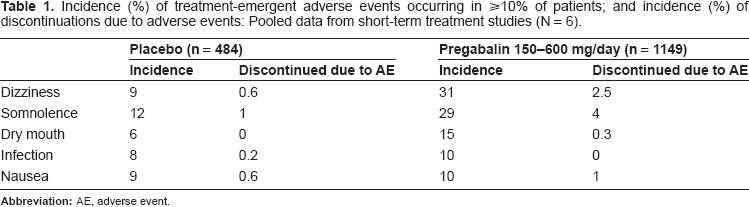
Treatment with pregabalin was well-tolerated in the dosage range of 150–600 mg per day. For the pooled 6-study short-term data set (not including the trial in the elderly; or the 8-week trial vs. venlafaxine-XR), only two adverse events occurred significantly more frequently on pregabalin compared to placebo, dizziness and somnolence (Table 1). The overall discontinuation rate due to adverse events was relatively low on pregabalin compared to placebo (11.3% vs. 9.3%; NNH = 50.0). Most adverse events, including dizziness and somnolence, were mild-to-moderate in intensity, and typically remitted within 2–3 weeks, regardless of whether the study was 4 weeks or 8 weeks in duration. Adverse events rates were notably higher on the 600 mg dose, which >80% of patients were titrated to by Day 7 as part of a fixed-dose regimen.
Incidence (%) of treatment-emergent adverse events occurring in ≥10% of patients; and incidence (%) of discontinuations due to adverse events: Pooled data from short-term treatment studies (N = 6).
Safety/tolerability vs. benzodiazepines
Because of its different mechanism of action, the safety and tolerability profile of pregabalin differs from the benzodiazepines in three important ways: (1) less sedation, and less impairment in arousal and attention; (2) less impairment in cognitive, memory and psychomotor function; (3) less risk of physical dependence and withdrawal.
As has been noted above, sedation is one of the most commonly reported adverse events on pregabalin. However the discontinuation rates due to intolerability were low for both dizziness (0.6%) and somnolence (2.5%), and thus do not appear to compromise treatment with pregabalin. Furthermore, the results of a pooled analysis found that the relative risk of sedation was significantly higher on alprazolam and lorazepam compared to the highest (600 mg) dose of pregabalin, both in a GAD subgroup presenting with moderate-to-severe insomnia (RR = 1.44) and a subgroup presenting with mild-to-no insomnia (RR = 1.72). 60 Overall, attrition due to adverse was higher in the pooled six short-term GAD studies for the benzodiazepine treatment group (28.1%) compared to the discontinuation rate on both the 300–450 mg dosages of pregabalin (8.1%) and the 600 mg dosage (18.0%). 61
Consistent with these findings, a crossover study of non-anxious volunteers found significantly less reduction on pregabalin compared to alprazolam on both vigilance and critical flicker fusion tests (a measure of CNS arousal). 31 Across a battery of cognitive and psychomotor tests, acute dosing with pregabalin was not associated with impairment relative to placebo. In fact, brake reaction time was significantly improved on pregabalin compared to placebo. In contrast, acute dosing with alprazolam was associated with significant impairment across almost all psychomotor measures, including break reaction time, choice reaction time, critical flicker fusion, vigilance and divided attention and tracking tasks. 31
Physical dependence and risk of withdrawal are major liabilities associated with benzodiazepine therapy. However the dependence and withdrawal risk appear to be notably less of a risk with pregabalin therapy. In animal studies, pregabalin did not produce the subjective and reinforcing effects associated with drugs of abuse (Pfizer, data-on-file). Pregabalin did not produce benzodiazepine-like discriminative stimulus effects in rhesus monkeys trained to discriminate midazolam from vehicle, nor did it maintain IV self-administration in rhesus monkeys. Unlike morphine, pregabalin did not induce conditioned place preference or drug discrimination in rats, suggesting low reinforcing effects.
In a long-term (6 month) treatment study utilizing pregabalin 450 mg/day, minimal-to-no withdrawal symptoms were observed upon discontinuation. The mean Physician Withdrawal Checklist (PWC) change score in the week after discontinuation was 2.0 for patients treated with pregabalin compared to -0.63 for placebo-treated patients. The PWC scores observed in after discontinuation of long-term benzodiazepine therapy are typically in the range of 12–25.
Finally a class warning on the small increased risk of suicidal ideation and behaviour has recently been added by the EU to all anti-epileptic drugs including pregabalin. The number of such events with pregabalin is too small to confirm or exclude the possibility of an increased risk.
Safety/tolerability vs. SSRIs and SNRIs
Head-to-head trials comparing pregabalin with SSRIs have not been reported. One trial has been published that compared pregabalin to venlafaxine-XR. 21 In this 8 week study, dizziness was reported more commonly during treatment with pregabalin, while nausea was reported more commonly during treatment with venlafaxine-XR (Fig. 1). The most salient difference was the lower incidence on pregabalin of adverse events rated as severe (9.1% vs. 20.0%). In addition, treatment with pregabalin was associated with a lower discontinuation rate due to adverse events (12.4% vs. 17.6%). This latter finding is consistent with a recent meta-analysis that found a higher relative risk of attrition due to adverse events on venlafaxine-XR compared to SSRIs (RR = 1.38). 62
The comparative safety profile of SSRIs and SNRIs is well-characterized by now. For example, a recent meta-analysis of clinical trials in depression 63 reported a 20%–40% higher attrition rates due to adverse events on venlafaxine-XR, duloxetine and paroxetine compared to escitalopram and sertraline. With the exception of escitalopram, which appears to have the most favorable side effect profile, sexual dysfunction and upper and lower GI complaints are characteristic complaints that occur in a small but significant subgroup of patients. Pregabalin offers a side effect profile that is distinctively different from both SSRI and SNRI drugs. Pregabalin lacks prominent GI side effects or sexual dysfunction. Instead, the two most prominent adverse events are dizziness and somnolence.
Long-term treatment with venlafaxine-XR, paroxetine and duloxetine are associated with a higher risk of withdrawal symptoms upon discontinuation than fluoxetine or sertraline. As summarized above, discontinuation of long-term pregabalin therapy appears to elicit minimal withdrawal symptoms.
Safety/tolerability in the elderly
Treatment of anxiety in the elderly is complicated by a greater likelihood of drug-drug interactions, due to higher rates of polypharmacy in this age group. Since pregabalin is primarily renally excreted (>90%), there are no CYP-related drug-drug interactions.
Treatment of GAD in the elderly is also complicated by a greater vulnerability to adverse events, especially sedation and cognitive-impairment. Benzodiazepines continue to be one of the most frequently prescribed treatments for GAD, 64 are associated with significant cognitive impairment and sedation in the elderly. 65 In contrast, the results of large, double-blind clinical trial of pregabalin in GAD in elderly outpatients found that treatment with pregabalin was not associated with any impairment on a cognitive battery that included DSST, digit span, and the set test. 59 In the same elderly sample, sedation was reported as an adverse event in 13.0% of patients compared to 7.3% on placebo (NNH, 17.5). However the median duration of sedation was relatively short (16 days to resolution), and no patients rated their sedation as severe.
Weight gain on pregabalin
Clinically significant weight gain is an uncommon adverse event that occurs in approximately 1 out of 70 patients treated with pregabalin. In short-term clinical trials in GAD, weight gain was reported as an adverse event in 1.2% of patients on placebo and 3.7% of patients on pregabalin. No patient discontinued because of weight gain. The mean change in weight was 1.4 kg for patients treated with pregabalin compared to 0.4 kg for placebo-treated patients. Four percent of patient treated with pregabalin experienced a clinically significant (>7%) weight gain compared with 1.4% on placebo. Pooled data from 30 controlled trials across the indications of GAD, epilepsy, and neuropathic pain combined, suggest that weight gain is dose related, but is not related to age, gender or baseline BMI. Weight gain is reversible upon drug discontinuation.
Conclusion and Place in Therapy
Pregabalin has proven itself to be a significantly efficacious treatment for GAD, regardless of illness severity, and across a range of clinical presentations. In countries where it is licensed, pregabalin is already widely used as a first line treatment or GAD. Based on available data, no other drug has demonstrated a larger effect size than pregabalin. 7 The efficacy of pregabalin is notable for its broad spectrum coverage of the range of psychic and somatic symptoms characteristic of GAD.
Illnesses such as GAD, for which there are multiple treatment options available, afford the opportunity to offer the patient a choice based on between-drug differences in key efficacy and safety parameters that are weighted according to patient preference. Multi-attribute decision modeling66,67 (MADM) is one method that has been used to match drug characteristics to weighted patient preferences, and thus systematically arrive at an optimal choice of drug for a given patient. Unfortunately, the MADM methodology has not been applied to drug treatments for GAD. Nonetheless, an informal version of this patient-centered approach may be employed. For example, patients will likely prefer pregabalin if they present with one or more of the following (Table 2): (1) prominent complaints of insomnia; (2) prominent GI symptoms; (3) severe anxiety and/or prominent somatic symptoms; (4) fluctuations in anxiety severity during the course of a week, and prefers a drug that can rapidly control these fluctuations; (5) concerns about developing physical dependence and/or withdrawal symptoms; (6) concerns about drug-drug interactions; (7) concerns about sexual dysfunction; and (8) concerns about drug-related cognitive and psychomotor impairment.
Clinical variables that may influence choice of pregabalin.

Even though pregabalin has demonstrated efficacy in treating GAD presenting with depressive symptoms, 68 still, the presence of comorbid depression favors the use of SSRIs or SNRIs, either as monotherapy, or add-on therapy to pregabalin in resistant cases.
More research is needed to fully characterize the place of pregabalin (and other anxiolytics) in the therapy of GAD. Currently, the number of available double-blind, head-to-head comparisons in GAD is relatively small, and no effectiveness studies, similar to the STAR*D program, have yet been published. As a consequence, evidence-based decision-making about choice of drug must largely rely on inferences from double-blind (non-comparator) clinical trials that do not include the heterogeneity of patient populations found in real world settings. Furthermore, it should be noted that the studies demonstrating the acute efficacy of pregabalin in GAD have been shorter in duration than comparable studies of SSRIs and SNRIs (4–6 weeks versus 6–10 weeks). This is largely due to the more rapid onset of improvement in anxiety symptoms among patients treated with pregabalin. Nonetheless, more long-term studies, of both pregabalin and other anxiolytics, are needed to more fully characterize clinical outcome after acute treatment (i.e. what proportion of patients progress from response to remission, and what proportion lose their initial response). Finally, relatively little is known about what treatment strategy should be used to treat patients who are treatment-resistant, or who achieve only a partial response to an initial course of therapy.
With these caveats in mind, it can be concluded that pregabalin is a safe and efficacious drug for GAD that offers a pharmacologically novel therapeutic approach.
Disclosures
Dr. Montgomery has served as a consultant for AstraZeneca, Bionevia, Bristol-Myers Squibb, GlaxoSmithKline, Johnson & Johnson, Eli Lilly, Lundbeck, M's Science, Merck, Merz, Neurim, Pierre Fabre, Pfizer, Richter, Roche, Sanofi, Sepracor, Servier, Shire, Synosis, Pharmaneuroboost, Takeda, Targacept, Theracos, Transcept, UBC, Wyeth, Xytis.
Dr. Kasper has received grant/research support from Eli Lilly, Lundbeck, Bristol-Myers Squibb, GlaxoSmithKline, Organon, Sepracor and Servier; has served as a consultant or on advisory boards for AstraZeneca, Bristol-Myers Squibb, GlaxoSmithKline, Eli Lilly, Lundbeck, Pfizer, Organon, Schwabe, Sepracor, Servier, Janssen, and Novartis; and has served on speakers’ bureaus for AstraZeneca, Eli Lily, Lundbeck, Schwabe, Sepracor, Servier, Pierre Fabre, and Janssen.
Footnotes
Acknowledgements
Some of the studies discussed in this manuscript were funded by Pfizer Inc. All authors were actively involved in the analytical method of the study, the selection and review of the scientific content and had full editorial control during the writing of the manuscript. Editorial support was provided by Edward Schweizer, M.D. of Paladin Consulting Group and was funded by Pfizer.
