Abstract
Atazanavir is the first azapeptide protease inhibitor. As a consequence of metabolism by the Cytochrome P450 system and excretion by drug-transporters such as P-Glycoprotein, drug interactions are considerable. They can be used to improve efficacy (ritonavir boosting) but may also cause adverse effects. Efficacy of ATV/RTV has been shown to be comparable to lopinavir/ritonavir in antiretroviral naïve patients, providing even better results in patients with high viral load. Efficacy has also been demonstrated in maintenance therapy in antiretroviral-experienced patients, and in patients with previous virologic failure, providing the best virologic response when the virus harbors less than four resistance PI mutations. The gastrointestinal tolerability and the lipid profile are better than with other PIs. The major side effect is a jaundice caused by unconjugated hyperbilirubinemia that rarely leads to discontinuation. ATV/RTV simple administration as well as tolerability may be linked with better treatment adherence. ATV/RTV is simple, potent and well tolerated. Thus it takes an important place in the treatment of HIV-infected patients, preferentially in antiretroviral-naïve or moderately pretreated populations.
Introduction
Since 1995, when saquinavir, the first human immunodeficiency virus (HIV) protease inhibitor (PI) was approved by the United States Food and Drug Administration (FDA), improvements have been made in the treatment of HIV-infected patients. 1 Two years later, a randomised controlled trial demonstrated the superiority of highly active anti-retroviral therapy (HAART) including one PI and two nucleoside reverse transcriptase inhibitors (NRTI), compared to dual NRTI therapy. 2 With such regimens, virologic and immunologic responses were obtained. The difficulty was that first generation PIs were associated with high pill burdens, gastrointestinal side effects, perturbation of lipid levels and glucose metabolism, and, in some cases, food and hydration requirements. 3 Marketed in 2003, atazanavir (ATV) is considered as a member of the second generation of PIs. Its characteristics make it different from other drugs of the same family. It is administered once daily, and has a good tolerability with few gastrointestinal disturbances and better lipid profile compared with other PIs. 4 All these factors can improve the therapeutic adhesion and the quality of life of patients infected by HIV, whereas providing good virological efficacy.
Pharmacodynamic Properties
These properties can be divided in the intended pharmacological effects on HIV (virologica response) and the unintended toxicological effects on the human body (side effects).
The HIV genome is composed of three genes, gag, pol and env. Translation of the gag and pol genes results in two precursor polyproteins, p55 (gag) and p160 (gag-pol). The HIV protease enzyme is responsible for the cleavage of these polyproteins to structural proteins (p55: matrix, capsid, nucleocapsid) and replicative enzymes (p160: protease, reverse transcriptase, integrase). 5 The anti-HIV PIs act by binding to the catalytic site of HIV protease, and by inhibiting proteolytic processing. The PIs prevent the production of infectious HIV and infection of new cells, but have no effect on cells with integrated proviral HIV DNA. 5 The antiretroviral PIs have to enter the cell to be active against HIV.6,7
ATV is the first aza-dipeptide PI. 8 A peptide (linkage CO-NH) in which the carbon atom was switched for a nitrogen atom, aza, contributes to provide the inhibitor effect on the VIH1 protease. 9 The fact that ATV is an aza-dipeptide (versus peptidomimetic PIs) explains in part its resistance profile. 10
The main side effect known is the elevation of indirect bilirubin. ATV is an inhibitor of the uridine diphosphate glucuronosyltransferase (UGT1A1), enzyme responsible for bilirubin glucuronidiation and liver clearance of bilirubin. The inhibition of UGT1A1 results in excess of unglucuronidated bilirubin. 11
Pharmacokinetic Properties
Absorption
ATV is rapidly absorbed with a median maximum time (Tmax) of 2.5 hours (range: 2-4 hours). The molecule demonstrates non-linear pharmacokinetics with greater than dose-proportional increases in area under the plasma concentration-time curve (AUC) and maximum plasma concentration (Cmax) values over the dose range of 200-800 mg once daily. Reduced plasma concentrations of ATV are to be expected if antiacids, H2-receptor antagonists, or proton-pump inhibitors are administered with ATV, due to a decrease in solubility correlated with a pH increase.
ATV must be taken with food. Administration with food enhances bioavailability and reduces pharmacokinetic variability. 12 Co-administration of a single 300- mg dose of ATV and a 100 mg-dose of ritonavir with a light meal (357 kcal, 8.2 g fat, 10.6 g protein), resulted in a 33% increase in the AUC and a 40% increase in the Cmax. 13 Large inter-patient and intra-patient variability has been reported. 14
Distribution
ATV is bound to human proteins (86%) and this protein binding is independent of plasma concentration. 15 It is bound in the same proportion to alpha-1-glycoprotein acid (89%) and to albumin (86%). The concentration of ATV in semen is high enough to expect good antiviral activity in the genital sanctuaries. 16 A weak correlation has been observed between the seminal plasma and the blood plasma concentration of ATV in 15 patients and it appears that ATV, in contrast to most other protease inhibitors, penetrates into seminal plasma. However, this penetration is limited and variable. 17 The diffusion in the central nervous system seems to be more limited. In a recent study, observed cerebrospinal fluid (CSF) concentrations were lower than the estimated free concentration in plasma (~210 ng/mL), suggesting an active transport out of the CSF. This study has shown highly variable CSF ATV concentrations, 100-fold lower than plasma concentrations, even with ritonavir boosting. CSF concentrations of atazanavir do not consistently exceed the half maximal inhibitory concentration (IC50) of atazanavir and may not protect against HIV replication in the CSF. 18
Metabolism/Excretion
ATV is metabolised in the liver by the cytochrome P450 complex and unchanged drug is excreted for approximately 20% and 7% in the feces and urine, respectively. 19 ATV inhibits CYP3A4 and UGT1A1 at clinically relevant concentration (Ki CYP3A4 = 2.35 μM and Ki UGT1A1 = 1.9 μM). The drug should not be administered concurrently with medications with narrow therapeutic windows that are substrates of CYP3A or UGT1A1. ATV inhibits competitively CYP1A2 and CYP2C9.
Drugs that induce CYP3A4 activity may increase the clearance of ATV, resulting in lower plasma concentrations. Co-administration of ATV with other drugs inhibiting CYP3A4 may increase ATV plasma concentrations. Ritonavir (RTV) is a more potent inhibitor of CYP3A4 than ATV. When co-administered, the ritonavir induced interactions could be more important. 20
ATV/RTV pharmacokinetics parameters are shown in Table 1.
Atazanavir 300 mg in association with 100 mg ritonavir: pharmacokinetic parameters.
Relations between Efficacy, Toxicity, and Plasma Concentration Efficacy
The association between the ATV trough concentration (Ctrough) and efficacy was described in 51 patients from the ATV-expanded access program. Virological response (plasma HIV RNA < 50 copies/ml) after 24 weeks was observed in 58%, 75% and 100% of patients with ATV Ctrough of 150 ng/ml, 150-850 ng/ml and 850 ng/ml, respectively. It is suggested that Ctrough has to be between 150 and 850 ng/ml to achieve the highest probability of virological response and the lowest probability of unconjugated-bilirubin increase. 21
Toxicity
In the same study, an association between high plasma concentrations and elevation of total bilirubin was also demonstrated. Elevations of total bilirubin were usually modest, causing low-grade adverse events. Another study confirmed this association and showed that MDR1 3435 CC genotype (associated with high P-Glycoprotein expression) was associated with high ATV concentration and total bilirubin elevations. 22
Drug-Drug Interactions
As a consequence of metabolism by the Cytochrome P450 enzyme and excretion by drug-transporters such as P-Glycoprotein, drug interaction is relevant. The drug-drug interactions can be used to improve efficacy but may also cause adverse effects.
Use of RTV in low non-therapeutic doses increases bioavaibility and decreases clearance of PIs, by inhibition of intestinal and hepatic CYP3A4 and P-Glycoprotein. 23 ATV, which is dosed once daily, is only licensed by the European Medicines Agency (EMEA) with RTV co-administration (http://www.emea.europa.eu).
According to the more potent RTV CYP3A4 inhibition, ATV/RTV must not be prescribed with CYP3A4 substrates with narrow index therapeutic as astemizole, cisapride, terfenadine, pimozide, quinidine, bepridil, triazolam, midazolam, and dihydroergotamine. For example, torsade-de-pointe, a life-threatening ventricular arrhythmia associated with QT prolongation, can occur when ATV/RTV are co-administered with terfenadine, astemizole, cisapride or pimozide.24,25 Symptomatic hypotension may occur when CYP3A4 inhibitors are given with dihydropyridine calcium antagonists. 24 Ataxia can occur with carbamazepine, and ergotism with ergotamine, following the addition of a CYP3A4 inhibitor. 24
Significant decrease (79%) in ATV Cthrough was reported with co-administration of omeprazole. 26 Co-administration of tenofovir, a nucleotide reverse transcriptase inhibitor, has also been shown to decrease the ATV AUC24 by 25%. 27 The mechanism of this drug-drug interaction is unknown.
A pharmacokinetic study performed in healthy subjects assessed the effect of multiple-dose ATV or ATV/RTV on raltegravir (a new HIV integrase inhibitor) plasma levels. 28 ATV alone or in combination with RTV increases the plasma concentration of raltegravir. This finding is attributed to the known inhibition effect of UGT1A1 by atazanavir, 11 the primary isoenzyme responsible for the metabolism of raltegravir. Increased toxicity when co-administered with other drugs has been related. Main known interactions can be found on the website www.hiv-druginteractions.org.
Applications of Therapeutic Drug Monitoring (TDM)
ATV therapeutic drug monitoring is encouraged in HIV-infected patients in case of toxicity, new co-administered drugs, un-boosted use, or gastrointestinal disease.29,30 Because of lack of data in some specific population as pregnant women, children, or patients with renal or hepatic impairment, TDM seems to be important. The FDA has recently recommended new paediatric dosing and TDM follow-up for patients between 6 and 18 years old. A recent study showed that ATV exposure at steady state during the third trimester of pregnancy is similar to that observed in the non pregnant state. 31 ATV crosses the placenta (maternal blood/umbilical cord ratio: 0.10) and may contribute to reduce the risk of vertical transmission.
Efficacy
First Line Therapy
CASTLE study (BMS AI424138) is the first large-scale study to evaluate ATV/RTV in first line therapy. This randomized, open-label, non-inferiority study compared the clinical efficacy of once-daily ATV/RTV (300/100 mg) and twice-daily lopinavir (LPV)/RTV (400/100 mg), both given with once-daily, fixed dose tenofovir and emtricitabine, in 883 treatment-naïve HIV-1 infected adults. The primary endpoint was the proportion of patients with HIV RNA below 50 copies/mL at week 48. Similar proportions of patients have achieved the primary endpoint with 78% and 76% of patients with HIV RNA below 50 copies/mL in the ATV/RTV and in the LPV/RTV group, respectively. In the sub-group of patients with high baseline viral load (greater than or equal to 100 000 copies/mL), 74% of the subjects in the ATV/RTV group achieved undetectable viral load, versus 72% in the LPV/RTV group. In a post hoc analysis, the lower response rates were associated with lower CD4 cell count at baseline in the LPV/RTV group but not in the ATV/RTV group. Gain in CD4 cell count from baseline to week 48 was similar in the 2 groups (+203/μL in the ATV/RTV group, +219/μL in the LPV/RTV group). Virologic failure occurred in 6% of the patients in each group. The two patients with emergence of non polymorphic PI resistance mutations at failure were taking ATV/RTV. In one case, the N88S, an ATV-associated resistance mutation, emerged. 32
At Week 96, virologic efficacy of ATV/RTV remained non-inferior to LPV/RTV. By intent-to-treat analysis, the rate of patients with HIV RNA below 50 copies/mL was significantly higher with ATV/RTV (74% and 68%, respectively). The mean change in CD4 cell count from baseline to week 96 was +268 in the ATV/RTV group, and +290 cells/mm3 in the LPV/RTV group. A higher rate of discontinuation has been observed in the LPV/RTV group versus the ATV/RTV group (21% vs. 16%, respectively). 33
The prospective, randomized, open-label, 96-week BMS AI424-089 study compared 2 ATV-based treatment regimens in 200 treatment-naïve patients. 34 Patients received either once daily ATV/RTV (300/100 mg) or once daily ATV (400 mg) both combined with lamivudine and stavudine. At week 48, 86% and 75% of the patients on the ATV/RTV regimen achieved HIV RNA below 400 copies/ml and HIV RNA below 50 copies/ml, respectively, whereas 85% and 70% achieved the same endpoints with the ATV regimen. The median CD4 cell count increases from baseline through week 48 were comparable, +174 cells/mm3 in the ATV/RTV group, versus +213 cells/mm3 in the ATV group. Virologic failures occurred more frequently in the ATV group (10 patients) than in the ATV/RTV group (3 patients). Major substitutions emerged in the genotype testing of 3 ATV-treated patients: 2 had a mixed I50I/L substitution and the third had pure I50L and N88S substitution. The latter patient's isolate was the only one that demonstrated phenotypic resistance to ATV. No emergence of major PI substitutions was observed in ATV/RTV treated patients. Seven patients with virologic failure in the ATV group and only one patient with virologic failure in the ATV/RTV group had a detectable M184V substitution, associated with lamivudine resistance. The small difference in virologic failure rates and the different emergent resistance patterns likely reflect the higher ATV plasma concentrations that are achieved with RTV boosting. After 96 weeks of therapy, the ATV group remains non-inferior to the ATV/RTV group in term of virological efficacy with 55% and 65% of the patients with a viral load below 50 copies/ml, respectively. However the rate of virological failure was higher in the ATV group (20 patients, versus 5, respectively). 35
Elion et al. have evaluated ATV/RTV in first line therapy in 2 single-arm, open label, multicenter 48-week study. In the COL102060 study, 112 treatment-naïve patients received once-daily abacavir/lamivudine plus ATV/RTV. 36 At week 48, the proportion of patients achieving HIV RNA below 50 copies/ml was 77% by intent-to-treat. Four patients met virologic non-reponse. Treatment-emergent drug resistance was rare, and no patient had virus that developed reduced susceptibility to ATV. In the BATON study, ATV/RTV was combined to tenofovir/emtricitabine in 100 treatment-naïve patients. At week 48, 81% achieved HIV RNA below 50 copies/ml. Neither K65R, tenofovir-associated resistance, nor any mutation associated with ATV resistance emerged in patients with virologic failure; M184V developed in one patient. 36
Treatment-Experienced Patients with Detectable Viral Load
ATV/RTV has been compared to LPV/RTV, both in combination with tenofovir and one nucleoside reverse transcriptase inhibitor, in treatment-experienced patients with previous virologic failure of at least two HAART regimens. 37 Over 96 weeks, the ATV/RTV regimen demonstrated similar virologic efficacy to the LPV/RTV regimen, with mean HIV RNA reductions from baseline of 2.29 log10copies/ml in the ATV/RTV arm and 2.08 log10copies/ml in the LPV/RTV arm. Median increases in CD4 cell count for patients treated with ATV/RTV and LPV/RTV were +160 and +142 cells/mm3, respectively. The analysis demonstrated a more favorable virologic response in patients with less than four PI mutations on baseline genotyping than in those with four or more mutations.
In the prospective NADIS cohort, virologic efficacy of ATV/RTV-containing regimens has been assessed in 424 patients with persistent viral replication. 38 Virological failure, defined by HIV RNA > 400 copies/ml at week 24, was described in 24% of the patients. Previous use of LPV, number of new drugs in the regimen, and baseline viral load were independently related with virological failure.
Maintenance Therapy
ATV/RTV monotherapy has been evaluated as maintenance therapy but is not a recommended option as the rate of viral rebound seems greater than would be expected in a highly compliant treatment population using traditional triple-HAART combination.39,40 In the SWAN study (AI424-097), virologically suppressed patients who were receiving stable PI-based regimens (with or without RTV) were randomized to continue to receive their existing PI or to switch to ATV (400 mg once daily) or ATV/RTV (300/100 mg once daily) if they were receiving tenofovir. The proportion of patients who experienced virologic rebound was significantly lower among those who switched to ATV-containing regimens than it was among those who continued to receive a comparator PI regimen with 7% and 16%, respectively. 41 In the subgroup of patients receiving LPV/RTV, a switch to ATV (+RTV)-based regimens resulted in comparable efficacy and improvements in plasma lipids, compared to patients who continued on LPV/RTV. 42
Safety and Tolerability
First Line Therapy
In the Castle study, clinical tolerance has overall been good.33,32 At week 48, nausea has been experienced by 4% of the patients receiving ATV/RTV versus 8% of those receiving LPV/RTV, and did not increase through week 96. Diarrhea has been experienced by 2% versus 11% of the patients respectively at week 48, and remained stable through week 96. Jaundice has been experienced by 4% of the ATV/RTV receiving patients and none of the LPV/RTV receiving ones, due to elevated levels of indirect and unconjugated bilirubinemia. Grade 2-4 hyperbilirubinemia was described in 34% of the ATV-treated patients at week 48% and 44% at week 96. The elevation of indirect and unconjugated bilirubinemia in patients receiving ATV/RTV has been described since the early development of the drug. 43 ATV is in part an inhibitor of the UGT1A1. This enzyme is involved in the liver clearance of bilirubine. It is dose related, reversible with treatment discontinuation and has not been found to be correlated with AST nor ALT elevations–-which are rare (<5%).
Appearance of cutaneous rashes has also been described early in ATV development, as frequently as in 22% of the patients of phase II studies. 43 However, it has only been described by 3% of the ATV/RTV receiving patients versus 2% of the LPV/RTV receiving ones in the Castle study at week 48 and has never lead to a treatment discontinuation. 32 Recent reports have highlighted occurrences of nephrolithiasis in patients using ATV-based regimens.44–46
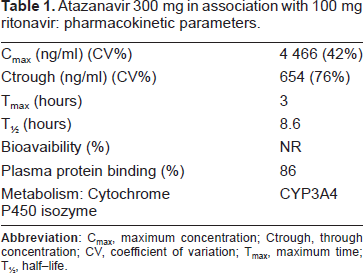
The potential risk of long-term adverse effects associated with PI-related dyslipidaemia is of great concern for physicians when choosing a PI-based regimen as first-line therapy. In the Castle study, changes from baseline in lipid parameters were therefore important secondary endpoints. Their evolution is shown in Figure 1.33,32 The changes in lipid parameters with treatment duration are less severe with ATV/RTV than with LPV/RTV.
Treatment-Experienced Patients with Detectable Viral Load
In treatment experienced patients with virologic failure, ATV/RTV has been compared to LPV/RTV, both in combination with tenofovir and one nucleoside reverse transcriptase inhibitor. 37 In this population, diarrhea was described by 3% of the ATV/RTV treated patients versus 13% of the LPV/RTV treated ones (p < 0.01), jaundice by respectively 7% and 0%, and hyperbilirubinemia in 53% versus <1%. Regarding dyslipemia, total cholesterol decreased by 7% in the ATV/RTV treated group whereas it increased by 9% in the LPV/RTV receiving group (p < 0.0001). The same profile was observed for HDL-cholesterol (-5% versus +7%), LDL-cholesterol (-11% versus +10%), and triglycerides (-2% versus +30%, p < 0.0001). The proportions of patients receiving lipid lowering drugs were of 6% and 5% respectively at baseline. At week 96, these proportions were 9% in the ATV/RTV treated group and 20% in the LPV/RTV one (p < 0.05).
In the prospective NADIS cohort, virologic efficacy of ATV/RTV-containing regimens has been assessed in 424 patients with persistent viral replication. 38 Treatment interruption due to an adverse event was related in 5% of the patients, predominantly hyperbilirubinemia (4% of the population), or cutaneous rash (4 cases). In this study, women gender has been shown to be related with increased rate of treatment interruptions (Adjusted OR = 1.91; 95% CI = 1.1-3.4), mainly because of poor tolerance. This gender issue has previously been studied in a small pharmacokinetic study 47 which showed that ATV concentrations were higher in women than in men. Gender driven differences in drug concentrations may be due to different pharmacokinetic parameters such as bioavailability, distribution, or metabolism. 48
Maintenance Therapy
In the SWAN study (AI424-097) the adverse-related discontinuation rate was comparable (6%) for subjects who maintained their current PI-based regimen and for subjects who switched to ATV or ATV/RTV. 41 There was a significantly lower rate of anti-diarrheal drug use for patients in the the ATV/RTV group, compared with the comparator PI group (2% versus 7%, p = 0.04). The proportion of patients who achieved lipid levels below the National Cholesterol Education Program specified thresholds for intervention with lipid-lowering therapy was increased, mainly in the ATV/RTV receiving group.
Atazanavir is not yet recommended during pregnancy and should be used only if potential benefit justifies the potential risk to the fetus. There are neither adequate, nor well controlled studies of atazanavir use during pregnancy. It is not known whether atazanavir administered during pregnancy will exacerbate physiological hyperbilirubinemia or increase the risk of kernicterius in neonates and young infants. If ATV/RTV is to be used during pregnancy, TDM is highly recommended.
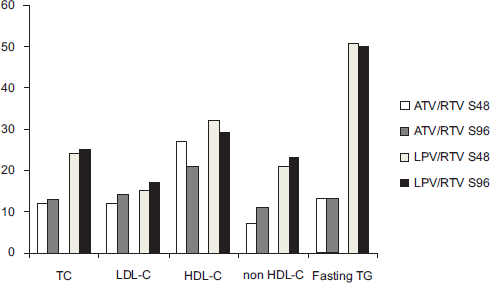
Efficacy and tolerability of the atazanavir-based regimens in the different populations are summarized in Table 2.
Efficacy and tolerability of atazanavir-based regimens in different populations.
Adherence
Since the first years of HAART utilization, it has been proven that continuous high treatment adherence is required to achieve the best and longest term virologic response. 49
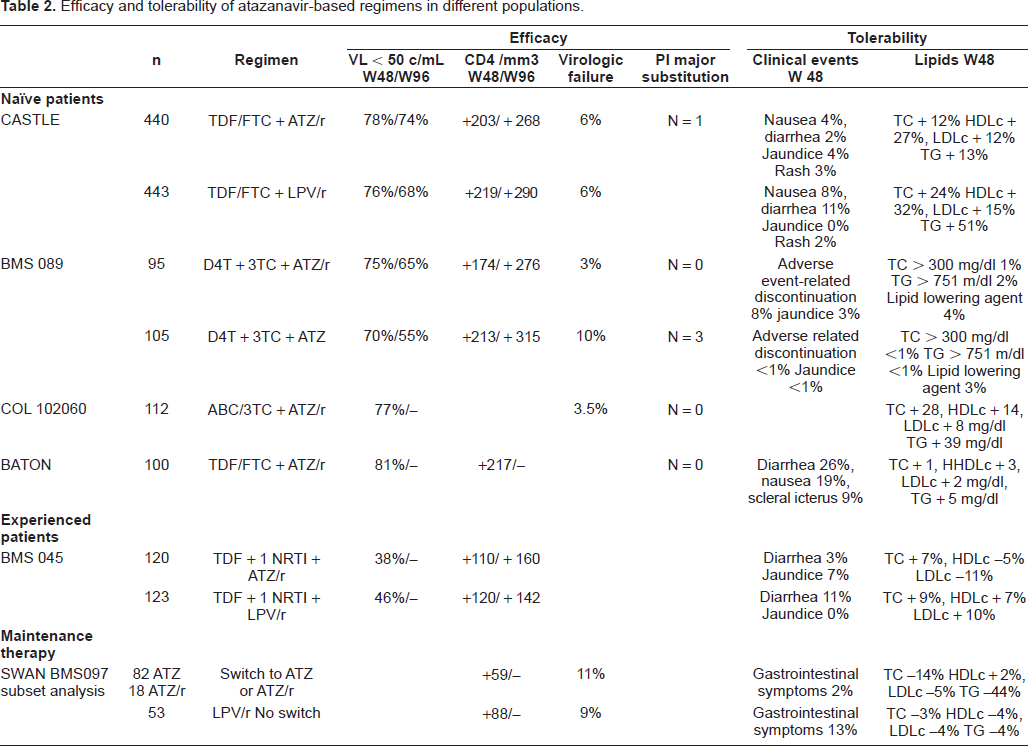
In an observational study performed in 2001, patients with once-a-day regimens have been found to be much more compliant with their treatment than patients with twice-daily or more frequent regimens. 50 JJ Parienti et al confirmed these results in a recent meta-analysis. 51 Even if the simplest available HAART is a one pill daily NNRTI-based regimen, it has been shown that adherence-resistance relationships were much weaker for protease inhibitors and nucleoside reverse transcriptase inhibitors compared with lamivudine and NNRTI in a large study including 1191 HIV-infected, antiretroviral-naive adults initiating highly active antiretroviral therapy. 52 In the Maggiolo's study, the 3 main reasons for missing doses are “problem with the schedule”, to be “away from home”, and to “have forgotten” (Fig. 2). Regimens based on one capsule of ATV 300 mg in association with one capsule of RTV 100 mg and one tablet of combined NRTIs may be simple enough as to minimize those reasons and facilitate adherence.
Reasons for missing antiretroviral medication dose,% of responding patients. 50
In the BATON study, 92% of treatment-naïve patients receiving an ATV/RTV-based once daily regimen reported complete adherence by 1-week recall, and 90% reported being “very satisfied” with the regimen at 48 weeks. 36
Conclusion
Ritonavir boosted atazanavir is a simple, tolerable and effective choice in combination with 2 NRTIs. ATV/RTV monthly costs are in the range of the other PIs. It can be used in first line therapy as well as in patients experiencing previous HAART failures, as long as the failures are due to lack of adherence or side effects rather than HIV genotypic mutations related with PI resistance. As ATV/RTV based regimens allow once-a-day schedule with minor side effects, they can improve therapeutic adherence and quality of life of HIV-infected patients.
Disclosure
The authors report no conflicts of interest.
