Abstract
Narcolepsy is a complex disease with multiple symptoms that include excessive daytime sleepiness, cataplexy, hypnagogic hallucinations, and sleep paralysis. Pharmacologic management of patients with narcolepsy is based on treating these symptoms. Sodium oxybate, a neurotransmitter product of γ-amino butyric acid (GABA), is a Schedule III controlled substance in the US, approved for the treatment of excessive daytime sleepiness (EDS), and cataplexy in patients with narcolepsy. The purpose of this review is to summarize the current knowledge about treatment of narcolepsy with sodium oxybate and to familiarize readers with the safety and efficacy of sodium oxybate, thus enabling clinicians to optimize their management of narcolepsy. A review of the pharmacokinetics, pharmacodynamics, efficacy in different symptoms of narcolepsy, adverse effects, tolerability, and dosing of sodium oxybate is presented. Sodium oxybate administered as an oral solution has a rapid onset of effect, a short duration of action, and nearly complete metabolism with negligible unchanged γ-hydroxybutyrate (GHB) appearing in the urine. There is mounting evidence suggesting that GHB may function as an endogenous neuromodulator/neurotransmitter. The mechanism by which sodium oxybate has a therapeutic effect in narcolepsy is unclear. The efficacy of oral sodium oxybate on the treatment of narcolepsy was evaluated in two randomized, double-blind, placebo- controlled, multicenter, US and International trials. Treatment of patients with narcolepsy-cataplexy with sodium oxybate leads to reduction in the frequency of cataplexy attacks in a dose-related manner, improved EDS, increased level of alertness and ability to concentrate. The major problem with sodium oxybate remains in its potential for non-medical use to elicit altered states of consciousness; however, abuse and misuse of sodium oxybate has been rare over the 7 years since its introduction in the market. Other issues revolve around twice nightly dosing, and salty taste. The most commonly reported adverse events associated with the use of sodium oxybate and occurring with at least 5% greater frequency than seen in placebo-treated patients were dizziness, headache, nausea, pain, night terrors, enuresis, confusion, infection, vomiting, and urinary incontinence. In conclusion, sodium oxybate significantly improves all symptoms in patients with narcolepsy and is well tolerated.
Introduction
Narcolepsy is an uncommon life-long condition that is often under-recognized, especially in children, and is associated with significant impairment in quality of life.1,2 It is a disorder of Rapid Eye Movement (REM) sleep that is characterized by excessive daytime sleepiness, cataplexy, hypnagogic hallucinations, sleep paralysis, and fragmented night sleep.1,3 Of the varied symptoms that may compromise the narcolepsy syndrome, excessive daytime sleepiness is ubiquitous. Central nervous (CNS) alerting or CNS neuromodulator agents are the mainstay of treatment of narcolepsy. 4 Stimulant medications are used with the goal of providing optimal alertness during working or school hours and other social situations.5,6 Modafinil, a non-amphetamine wakefulness-promoting medication, is now considered the first-line treatment for EDS in adults with narcolepsy. 7 REM-suppressant medications such as tricyclic antidepressants or serotonin reuptake inhibitors can be added to control cataplexy or hypnagogic hallucinations. 5 Sodium oxybate is a gamma amino butyric acid (GABA) B receptor agonist currently approved by the U.S. Food and Drug Administration (FDA) for the treatment of both cataplexy and EDS in patients with narcolepsy with age more than 16 years.7,8 The focus of this article is the use of sodium oxybate in narcolepsy.
Narcolepsy
Narcolepsy is a chronic and potentially disabling rapid eye movement sleep disorder resulting from the dys-regulation of sleep-wake cycles, 1 that affects 0.03 to 0.16% of the general population in various ethnic groups. 9 Under the term narcolepsy, the International Classification of sleep disorders (ICSD-2) includes: 10
Primary narcolepsy with cataplexy
Primary narcolepsy without cataplexy,
Narcolepsy due to medical conditions
Unspecified narcolepsy.
Narcolepsy with cataplexy is the most widely recognized and best characterized disorder among the narcolepsy syndromes. Cases of narcolepsy without cataplexy are thought to represent between 10% and 50% of all narcolepsy cases. 4
Clinical features of narcolepsy
The core symptoms of narcolepsy in children are similar to those in adults, but the clinical expression may be different because of maturational factors.5,11 Historically, narcolepsy was considered a disorder of adulthood, but accumulating evidence indicates that signs of narcolepsy may start during childhood.12–14
The characteristic tetrad of narcolepsy includes daytime hypersomnia, cataplexy, hypnagogic hallucinations, and sleep paralysis. 5 The initial and sole manifestation of childhood narcolepsy usually is hypersomnia followed by development of other symptoms months to years later.5,12,15–17 In addition, sleep fragmentation has been recognized as a fifth but commonly present criterion in the diagnosis of narcolepsy. 10
Hypersomnia
Besides daytime sleepiness, patients also experience “sleep attacks”, during which, patients may suddenly feel overwhelmingly tired, and appear transiently “unaware” of their surroundings. 10 Such sleep attacks are refreshing on awakening.
Cataplexy
Cataplexy is a unique feature of narcolepsy. 5 Attacks of cataplexy are episodes of abrupt and reversible decrease or loss of muscle tone which can manifest from a barely perceptible slackening of the facial muscles to head dropping, knee buckling, or total collapse onto the ground. 5 During a cataplexy attack, which may last up to several minutes, the patient is unable to move, although the diaphragm and ocular muscles are unaffected. Preservation of consciousness and intact memory are distinctive features, thus differing from sleep attacks. Cataplectic attacks are typically triggered by intense emotions such as exhilaration and surprise, and by the act of laughter.18,19 The frequency of cataplexy ranges from only one or two episodes per year to dozens of episodes per day. 17 The onset of cataplexy may be delayed for years after the onset of hypersomnia. Attacks of cataplexy may be confused with atonic seizures (“drop attacks”), especially in children under age 5 years. 18
Hypnagogic/hypnopomping hallucinations and sleep paralysis (HH and SP)
Sleep paralysis are brief nocturnal episodes of an inability to move, generally occurring during awakening, and relieved by touching or speaking to the child. Sleep paralysis can be accompanied by hypnagogic (during the transition from wakefulness to sleep) or hypnopomping (during the transition from sleep to wakefulness) hallucinations which consist of vivid visual or auditory experiences, usually of a benign content.1,10 However, both of these symptoms can be unrecognized or misinterpreted by young children or their parents. They may seem to resemble nightmares in children and may be frightening. Both symptoms can occur in isolation in normal people also, without the person having narcolepsy.
Etiology and pathophysiology of narcolepsy
The neurophysiologic hallmark of narcolepsy is the abnormal transition from wakefulness directly into rapid eye movement sleep. Signs such as cataplexy and sleep paralysis are thought to be related to “intrusion” of rapid eye movement sleep into wakefulness. 20 However, this hypothesis does not explain the presence of daytime sleepiness and the short latency to both non-REM and REM sleep during nocturnal and nap recordings. 21 A complementary hypothesis is that narcolepsy results from the disruption of the control mechanism of both sleep and wakefulness. 21
Animal and human studies have suggested that a deficiency in the hypothalamic orexin/hypocretin system underlies the pathogenesis of narcolepsy with cataplexy.22–25 The orexin system is a major excitatory system that affects the activity of monoaminergic and cholinergic systems with major effects on vigilance. 26 The loss of hypocretin excitatory projections creates a cholinergic-monoaminergic imbalance consistent with sleepiness and abnormal REM sleep in narcolepsy.
Diagnostic criteria and laboratory assessment of narcolepsy
The most recent diagnostic criteria for narcolepsy with and without cataplexy are summarized in Table 1. 10
Criteria for narcolepsy.*

International Classification of Sleep Disorders-second edition. 1
Nocturnal polysomnography is obtained to rule out other sleep disturbances such as sleep disordered breathing (SDB) and periodic limb movements of sleep, and for confirming a sleep efficiency above 75%.10,27
The multiple sleep latency test is generally performed on the following day to assess the degree of sleepiness and timing of rapid eye movement sleep onset. The combination of two sleep-onset rapid eye movement periods and a mean sleep latency of <8 minutes is necessary for the diagnosis of narcolepsy10,28
Laboratory tests such as human leukocyte antigen typing and cerebro-spinal fluid hypocretin-1 analysis are useful as adjuncts, but at present are not considered mandatory for a diagnosis of narcolepsy. 29 Genetic studies indicates a strong association between narcolepsy and human leukocyte DQ antigens, specifically DQB1*0602 and DQA1*0102, which are present in 95% to 100% of the narcoleptic population with cataplexy, respectively, but also present in 25% of the normal population. 29 Cerebrospinal-fluid hypocretin-1 measurements may be especially useful when multiple sleep latency testing is difficult to conduct or to interpret in complex cases, and in children. Lower cerebro-spinal fluid levels of hypocretin-1 (< 110 pg/mL) in narcoleptic patients are found, 30 especially in subjects with human leukocyte B1*0602 antigen positivity 30 or signs of cataplexy. 31
Treatment of Narcolepsy
The general approach to the treatment of childhood narcolepsy and other hypersomnias of central origin is based on the adult experience. Recently, the American Academy of Sleep Medicine (AASM) 32 published the practice parameters for the treatment of these disorders.
Non-pharmacological treatment (life style changes)
Education support and counseling of both family and school officials are cornerstones of management. According to the AASM, “scheduled naps can be beneficial to combat sleepiness, but seldom suffice as a primary therapy to narcolepsy (guideline)”. 32 In our experience, short naps during school time (e.g. during lunch break) have been effective in improving daytime functioning. 12 The combination of regular bedtimes and two 15-minute regularly scheduled naps reduced unscheduled daytime sleep episodes and sleepiness when compared to stimulant therapy alone. 33
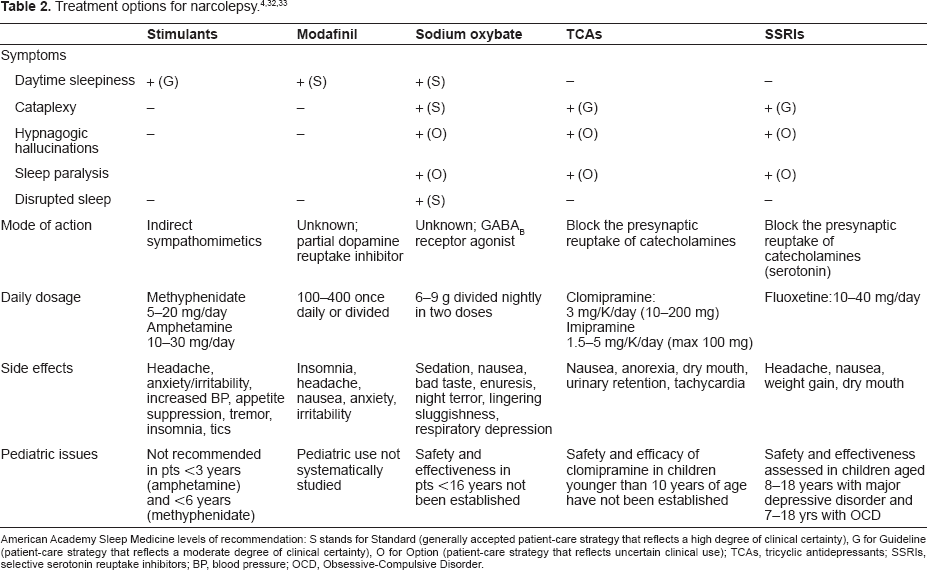
Pharmacological treatment is summarized in Table 24,32,33
American Academy Sleep Medicine levels of recommendation: S stands for Standard (generally accepted patient-care strategy that reflects a high degree of clinical certainty), G for Guideline (patient-care strategy that reflects a moderate degree of clinical certainty), O for Option (patient-care strategy that reflects uncertain clinical use); TCAs, tricyclic antidepressants; SSRIs, selective serotonin reuptake inhibitors; BP, blood pressure; OCD, Obsessive-Compulsive Disorder.
The rest of the article will focus on the pharmacology and use of Sodium Oxybate in narcolepsy.
Sodium Oxybate
From club drug to orphan drug
Sodium oxybate (γ-hydroxybutyrate; Xyrem®) is a neurotransmitter product of GABA. 8 Note that γ-hydroxybutyrate refers to the endogenous substance found in mammalian brain and other tissues while sodium oxybate is the official generic name of a pharmaceutical agent. The term ‘illicit GHB’ is used to refer to illegally manufactured GHB and/or congener substances such as industrial 1,4-BD (butanediol) and GBL(gamma-butyrolactone), substances manufactured as unfit for human consumption. 34
Three decades ago, GHB was introduced in the health-food market as a food supplement for body builders with claims of anabolic effects by stimulating growth hormone release. 35 In the 1990s, GHB was misued as a recreational drug at music venues.36–38 It has been suggested that the common illicit formulation of GHB as a colorless odorless liquid facilitates the unsuspected addition of GHB to the drinks of individuals in bars and clubs. 39
Abuse, addiction, withdrawal syndrome, and overdoses have been documented with GHB, which led to its 1990's ban and placement as a Schedule I agent under the Controlled Substances Act.40,41 Concerns regarding the history of abuse of GHB were taken into consideration by the Food and Drug Administration (FDA) and the manufacturer in the development of sodium oxybate.40,41
Sodium oxybate (γ-hydroxybutyrate; Xyrem®) was approved in 2002 by the Food and Drug Administration as an orphan drug for the management of narcolepsy. 42 It is a federal schedule III controlled substance in the US, which is available only through a centralized pharmacy because of earlier diversion of the drug for recreational use. Rigid guidelines have therefore been established for the prescription of sodium oxybate, which has to occur in conjunction with a detailed patient education program, subsequent to which the drug is shipped directly by the centralized pharmacy to the patient's home address.
Chemistry
The chemical name for sodium oxybate is sodium 4-hydroxybutyrate.34,43 The molecular formula is C4H7NaO3 (Fig. 1) and the molecular weight is 126.09 grams/mole. 43 Sodium oxybate is a white to off-white, crystalline powder that is very soluble in aqueous solutions. 44 GHB, an endogenous short chain fatty acid, is present in the normal brain and serves as both a precursor and a degradation product of the inhibitory neurotransmitter gamma-aminobutyric acid (GABA). 45 GABA is formed from glutamate by action of the glutamic acid decarboxylase (also called glutamate decarboxylase) and is degraded to succinic semialdehyde (SSA) by GABA transaminase. GHB may be formed from SSA by action of succinic semialdehyde reductase or can be converted back to SSA via the GHB dehydrogenase. Succinic semialdehyde is metabolized in the mitochondria by succinic semialdehyde dehydrogenase (SSADH) to succinic acid, which enters the Krebs cycle or can be converted back to GABA. 8 Additionally, GHB is an active metabolite, but to a much lesser extent, of 1, 4-butanediol (1, 4-BD) and gamma-butyrolactone (GBL). 8 The latter two substances are also commercially produced synthetically for a wide range of industrial purposes.
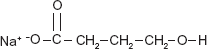
Chemical structure of sodium oxybate.
GHB is thus an interesting and unique compound in that it is an endogenous molecule, a product of abuse (illicit GHB), and a marketed therapeutic drug (GHB sodium salt; or sodium oxybate; Xyrem®, Orphan Medical). 39
Pharmacokinetics
In mammals, GHB is present in micromolar concentrations in all brain regions investigated, although the concentration differs by region. 8 The highest relative concentration is found in the striatum of humans and monkeys. 46 Localization of GHB within cytosolic and synaptosomal fractions 47 suggests a mechanism for presynaptic accumulation. The concentration of GHB is higher in the developing brain than in the adult brain. 48 GHB is also found in micromolar concentrations in peripheral tissues such as heart, kidney, liver and muscle, with the highest concentration being found in the brown fat. 49
The oral pharmacokinetics of sodium oxybate has been studied in healthy volunteers, 50 patients with liver impairment, 51 alcohol-dependent patients, 52 and narcoleptic patients. 53 The pharmacokinetics of oxybate is nonlinear and dose dependent50,52 and demonstrates capacity-limited absorption and elimination. 50 Gender does not appear to affect the drug's kinetic parameters. 54
Absorption and distribution
Due to the highly hydroscopic nature of sodium oxybate, the drug has been approved for use orally as a solution. Following oral administration, sodium oxybate is rapidly absorbed with plasma concentrations generally reaching a peak within 30 to 60 minutes and then falling rapidly with an elimination half-life of 0.5 to 1.0 hour.50,54 It then undergoes extensive first-pass metabolism.54,55 In fasted male and female volunteers 50 the mean maximum plasma concentration (Cmax) after a single oral dose of sodium oxybate appeared to be dose dependent and was reached in mean time (tmax) of 25–75 minutes.50,54,55 Administration of a second dose 4 hours later produced a 1.4–2.2 fold increase in mean Cmax. 53
The mean area under the plasma concentration curve from zero time to infinity (AUC) was nonlinearly dose dependent with a 2.5 to 2.8 fold increase in mean AUC in response to a 2-fold increase in dose, which may reflect capacity-limited absorption. 50
The ingestion of food with sodium oxybate has been shown to significantly reduce peak plasma concentrations and overall systemic absorption. 54 Ingestion of sodium oxybate after consuming a high-fat meal provided a maximum concentration of less than 50% of what is observed in a fasting state. 54 The time to reach maximum concentration was also significantly prolonged, and the AUC significantly decreased in the non-fasting group. Therefore, to obtain maximum peak effect, high-fat meals should be preferably avoided before bedtime when sodium oxybate is administered.
Although the maximum concentration and AUC are slightly increased upon repeated dosing, the changes seen are considered clinically insignificant.44,55
An open-label, two-period study 55 was designed to compare acute and chronic treatment on the pharmacokinetics of sodium oxybate oral solution in narcoleptic patients. Following 8 weeks of nightly administrated of sodium oxybate in a split dose, mean Cmax was increased from baseline by 16% and mean AUC increased 12% in 13 patients with polysomnographically confirmed narcolepsy. The differences were not considered to be clinically relevant. Thus, the pharmacokinetics of sodium oxybate in narcoleptic patients were not changed in any clinically significant manner when the drug was chronically administered.
The absolute bioavailability of oxybate was determined to be about 27% when a dose of 100 mg/kg was compared after intravenous and oral administration. 55 No appreciable plasma protein binding occurs with oxybate. 50
Metabolism and elimination
Metabolism initially involves conversion to succinic semialdehyde by the enzyme GHB dehydrogenase. 44 Succinic semialdehyde dehydrogenase then converts the compound to succinic acid, which subsequently enters the Krebs cycle. It is ultimately metabolized to carbon dioxide and water. 34 Minimal amounts of unchanged drug are eliminated in the feces or urine. 52
In vitro, sodium oxybate does not significantly inhibit the activity of cytochrome P450 (CYP) isoenzymes up to concentrations of 3 mmol/L. 8
The elimination of sodium oxybate from the circulation is rapid, and the relatively short half-life necessitates twice-nightly administration. 53 Elimination half-life and apparent clearance (CL/F) of sodium oxybate did not appear to be influenced by gender, food intake, or multiple-dose administration.54,55 Mean residence time (MRT) varied with dose from 45–249 minutes.50,53
Special populations
In hepatically impaired patients receiving sodium oxybate, it is considered necessary to half the starting dose and to closely monitor potential adverse events during dose titration. 8
In alcohol-dependent patients, consistent with its rapid onset and short pharmacological effect, the data indicates that both GHB absorption into and elimination from the systemic circulation were rapid processes. 52
Potential drug interactions
Drug interactions have not been observed with sodium oxybate. However, an increased rate of adverse events was observed during co-administration with protriptyline. 56
In conclusion, sodium oxybate administered as an oral solution has a rapid onset of effect, a short duration of action, and nearly complete metabolism with negligible unchanged GHB appearing in the urine.
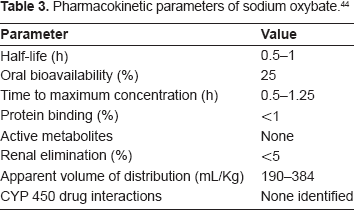
Table 3 summarizes the pharmacokineticdata for sodium oxybate. 44
Pharmacokinetic parameters of sodium oxybate. 44
Pharmacodynamic
GHB is a natural neurotransmitter/neuromodulator that may act through its own receptors and via stimulation of GABAB receptors; 57 however, its precise functions are unknown. 58 The pharmacodynamic properties of sodium oxybate are not expected to differ significantly from those of GHB. 8 An increasing number of studies over the last several years have focused on examining the mechanism of action of GHB. 57 A GHB receptor has been cloned, 59 new receptor binding and functional assays for the GHB receptor have been characterized, 60 new ligands that bind selectively to GHB receptors have been synthesized, 61 and new behavioral effects have been developed. 62 The behavioral effects of GHB are mediated predominantly by GABAB receptors. However, there are also data that suggest that other mechanisms such as other GHB receptors and different subtypes of GABAA and GABAB receptors might contribute to the effects of GHB. 57
GHB as a neurotransmitter: effects on neurotransmitter systems
There is mounting evidence suggesting that GHB may function as an endogenous neuromodulator/ neurotransmitter. 63 Under endogenous conditions and concentrations, and depending on the cell group affected, GHB may increase or decrease neuronal activity by inhibiting the release of neurotransmitters that are co-localized with GHB. In pharmacological concentrations, GHB also may act as a GABAB receptor agonist; most of the observed behavioral effects appear to be mediated via the activity of GHB at GABAB receptors, as long as the concentration is sufficient to elicit binding, which does not happen at endogenous concentrations. 64 Pharmacological doses of GHB increase serotonin turnover and interact with endogenous opioid systems. 65 Dopaminergic regions such as the substantia nigra and ventral tegmental area contain high concentrations of GHB, suggesting that endogenous GHB may modulate the activity of dopamine neurons.
GHB and sleep: effects on sleep architecture
The mechanism by which sodium oxybate has a therapeutic effect in narcolepsy is unclear. 66 One proposed mode of action involves improving nocturnal sleep, which then leads to improved alertness the following day. Sodium oxybate reduces cataplexy within days of intake, and has an effect on daytime sleepiness after approximately 6 to 8 weeks.67,68 Sodium oxybate acts through GABAB receptors and perhaps through its own receptors. 59 It suppresses dopaminergic neuronal activity65,69 with a consequent increase in slow-wave sleep and a decrease in the number of nighttime awakenings, with a corresponding increase in sleep efficiency.70,71
Clinical trials have shown that GHB increases delta sleep and improves the continuity of sleep in normal and narcoleptic subjects. A variety of neuropharmacologic mechanics of action have been reported, but none has been conclusively established. 53 Important targets of GHB are the thalamocortical circuits involved in the sleep-wake cycle and susceptibility to generalized seizures. 58 In vitro studies indicate that GHB, acting via GABAB receptors, reduces neuronal activity in thalamic, hippocampal, and neocortical neurons. GHB administered to healthy volunteers 71 or to patients with narcolepsy/cataplexy 6 elicits an increase in slow-wave sleep (stage N3), which is characterized by the presence of delta waves in the EEG. GHB affects thalamocortical circuits via both presynaptic and postsynaptic GABAB receptors in a dose-dependent manner. 45 It is noteworthy that at very high doses, GHB may elicit loss of excitability in thalamocortical networks, as may occur during overdose or coma. 45
A very recently published double-blind, placebo-controlled study enrolled 278 patients with narcolepsy taking modafinil 200–600 mg daily for the treatment of excessive daytime sleepiness (EDS). 72 Following a baseline polysomnogram (PSG) and Maintenance of Wakefulness Test (MWT), patients were randomized to receive treatment with: (1) placebo, (2) sodium oxybate, (3) modafinil, or (4) sodium oxybate + modafinil. PSGs and MWTs were repeated after 4 and 8 week. The authors concluded that in addition to its established efficacy in the treatment of cataplexy and EDS, nightly sodium oxybate administration significantly reduces measures of sleep disruption and significantly increases slow-wave sleep in patients with narcolepsy.
It is somewhat paradoxical that although most of the behavioral effects of GHB appear to be mediated by GABAB receptors, the prototypical GABAB receptor agonist baclofen is not used as a therapeutic for narcolepsy. The therapeutic effects of GHB in narcoleptic patients do not appear to be due to a general sedative/ hypnotic effect per se, rather, it may be due to a specific increase in delta power and duration of slow wave sleep. 34 A recent clinical trial showed that although GHB and baclofen increased EEG delta activity during nocturnal sleep, only GHB reduced subjective daytime sleepiness and attacks of cataplexy. 66
GHB and seizures
The effects of GHB in the thalamus may also predispose to development of generalized seizures. Absence seizures elicited by GHB administered either systemically or locally in the thalamus serve as good pharmacologic models of human typical absence seizures. 73
GHB and drug addiction
GHB affects the mesolimbic dopaminergic circuit, which is critically involved in mechanisms of drug addiction. 66 At concentrations typically seen with recreational use, GHB preferentially inhibits local GABA-ergic neurons in the ventral tegmental area (VTA), thereby increasing dopamine release in the nucleus accumbens, which is typically seen as a reinforcing effect of many drugs of abuse. At higher concentrations, GHB hyperpolarizes the dopaminergic VTA neurons, resulting in decrease in dopamine output, which may explain the anti-craving properties of GHB. 66 Chronic GHB administration also induces a compensatory up-regulation of dopaminergic D1 and D2 receptors and may also desensitize GHB and GABAB receptors in dopaminergic neurons; this could contribute to the addictive properties of GHB. 74
GHB and neuroprotection
There is in vitro evidence that GHB may exert neuroprotective effects by several mechanisms. 75 GHB may spare energy utilization, reduce oxidative stress, block excitotoxicity, and maintain vascular integrity. The ability of GHB to shift the intermediary glucose metabolism to generate the reducing equivalents such as nicotinamide adenine dinucleotide phosphate (NADPH), which is required for the elimination of reactive oxygen species and lipid peroxides, may provide a basis for the effects of GHB against oxidative stress.
Efficacy
The evaluation of sodium oxybate in narcolepsy include effects on cataplexy, excessive daytime sleepiness, hypnagogic hallucinations, sleep paralysis and measures on global impression of change. The clinical findings are fairly consistent and are discussed in the following section.42,76–80
The efficacy of oral sodium oxybate on the treatment of narcolepsy was evaluated in two randomized, double-blind, placebo- controlled, multicenter, US 42 and International trials. 80 The first one was used in its FDA approval. 42 The use of sodium oxybate in addition to modafinil was evaluated in a randomized, double-blind, double-dummy, pararell-group, placebo-controlled, multicenter combination-therapy trial. 81
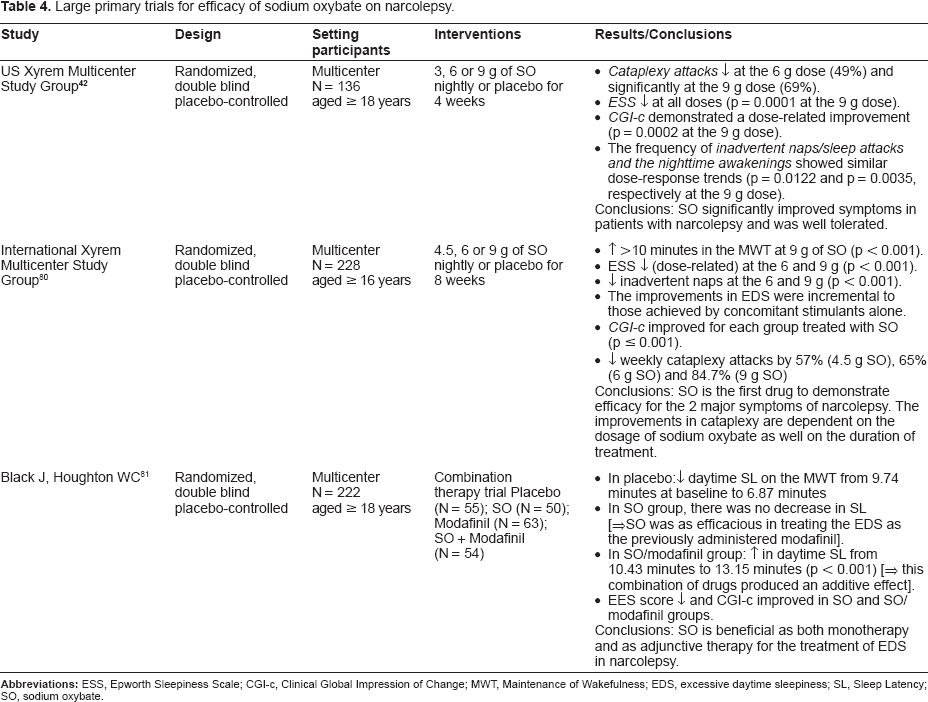
These trials have been summarized in Table 4.
Large primary trials for efficacy of sodium oxybate on narcolepsy.
Effects on cataplexy
The frequency of cataplexy attacks was reduced in a dose-related manner in patients with narcolepsy-cataplexy after 4 (US) or 8 weeks (International) of treatment with sodium oxybate.42,80 During the 12- month open-label extension of the US trial, the frequency of cataplexy attacks continued to decline with a mean reduction of 24 attacks per week after 4 weeks of treatment, to a mean reduction of 35 attacks per week (90%–98% reduction from baseline) by trial end. 82 Additional data supplementing the US trial have been drawn from a double-blind withdrawal study. 83 Fifty-five narcoleptic patients with cataplexy who had received continuous treatment with sodium oxybate for 7–44 months (mean 21 months) were enrolled in a double-blind treatment withdrawal paradigm. A 2-week single-blind sodium oxybate treatment phase established a baseline for the weekly occurrence of cataplexy. This was followed by a 2-week double-blind phase in which patients were randomized to receive unchanged drug therapy or placebo. During the 2-week double-blind phase, the abrupt cessation of sodium oxybate therapy in the placebo patients resulted in a significant increase in the number of cataplexy attacks. The investigators noticed that in contrast with antidepressant drug therapy, there was no evidence of rebound cataplexy upon abrupt discontinuation of treatment.
Excessive daytime sleepiness (EDS)
A growing body of evidence indicates that nightly administration of sodium oxybate to patients with narcolepsy reduces EDS. The results of US and International Xyrem trials have shown that sodium oxybate reduced EDS, increased the level of alertness and the ability to concentrate.82,84 Additionally, the results of the combination-therapy trial has shown that sodium oxybate and modafinil to be equally efficacious for the treatment of EDS, producing additive effects when used together. 81
In the international trial, after 8 weeks of treatment, subjective sleepiness showed significant improvement, manifested as reduction in ESS (improved) with all doses of sodium oxybate (p < 0.001) compared to baseline. 84 In the US trial, 41 EDS was a secondary measure and was also assessed using the ESS, which improved in all of the sodium oxybate treatment groups in a dose-related manner, becoming significant at the 9 g dose when compared to placebo. This improvement in wakefulness occurred even though the majority of patients remained on stable doses of stimulant medications. During the extension study of the US trial, mean ESS score decreased a further 30% and were significantly lower after 2 months on treatment than observed at entry into the study. 82
The results of the combination therapy trial 81 demonstrated that sodium oxybate and modafinil are both effective for treating excessive daytime sleepiness in narcolepsy, producing additive effects when used together.
Thus, sodium oxybate is beneficial as both monotherapy and adjunctive therapy for the treatment of excessive daytime sleepiness in narcolepsy. 81
Effects on hypnagogic hallucinations and sleep paralysis
In both the US and International trial, changes in HH and SP attacks occurred on a background of low baseline frequencies. Only the reduction in SP attacks in the 9g/night group in the International trial was significantly different form placebo. 85 In the study of Mamelak et al 79 when patients with narcolepsy received 4.5 to 9 g per night doses, the proportion of those reporting a decrease in HH and in SP ranged from 29 to 76%, and 38% to 76% respectively.
Effects on clinical global impression (CGI) and health–-related quality of life (HR-QOL)
In the US study, the proportion of patients who responded very much or much improved differed significantly across doses suggesting a dose-related response. 42 During the extension of this study more than 80% were classified as very much/much improved after 2 months of treatment, a level of response that was maintained at the end of the 12 month period. 82 In the International study, the proportion of very much/ much improved was significantly higher than the response of the placebo at all three doses of sodium oxybate. 84 In the combination therapy trial, the proportion of very much/much improved patients was 22% in the placebo group, 19% in modafinil, 48% in sodium oxybate monotherapy group, and 46% in the combination group. 79
In the International study, 84 HR-QOL was improved by treatment in a dose-related manner. The change in total FOSQ score and activity level, vigilance, general productivity and social outcomes subscales were all significantly different at 9 g dose from placebo. However, the intimacy and sexual relationships subscale scores did not change significantly at any dosage.
Side Effects
The major problem with sodium oxybate remains in its non-medical use to elicit altered states of consciousness. Although adverse events (including death) associated with popular abuse of GHB and its analogs have been widely reported, and although the Xyrem package insert has related warnings, to date there have been few reports in the clinical literature on adverse effects associated with therapeutic use, misuse, and abuse of pharmaceutical GHB sold as Xyrem.35,85,86
According to the FDA-approved package insert, the most commonly reported adverse events (≥5%) in placebo controlled clinical trials associated with the use of sodium oxybate and occurring more frequently than seen in placebo-treated patients were: nausea (19%), dizziness (18%), headache (18%), vomiting (8%), somnolence (6%), urinary incontinence (6%), and nasopharyngitis (6%) 42,80–84,86
Selected adverse events by Coding Symbols for the Thesaurus of Adverse Reactions (COSTART) and sodium oxybate treatment group are summarized in Table 5.43,82
Selected adverse events by Coding Symbols for the Thesaurus of Adverse Reactions (COSTART) and sodium oxybate treatment group.
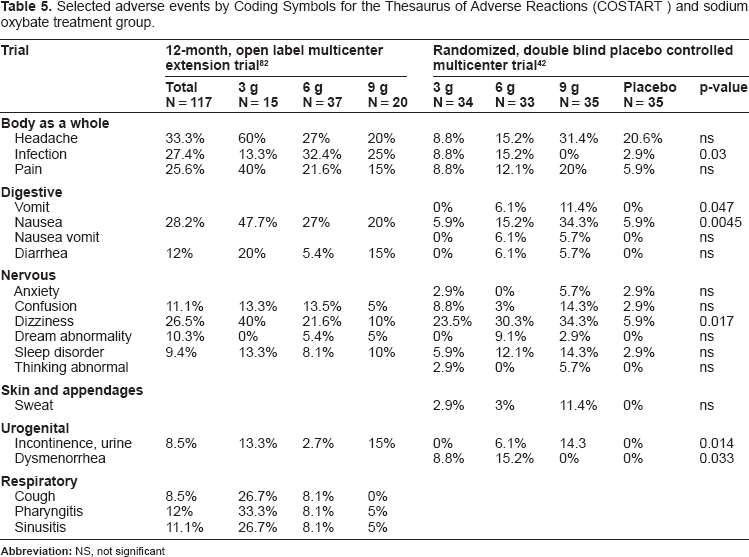
Several patients with pre-existing obstructive
The treatment of patients with narcolepsy with sodium oxybate can result in weight loss. 89 Patients with narcolepsy tend to be overweight due to impaired energy homeostasis. 90 This weight loss in patients with narcolepsy, especially those with cataplexy, on treatment may be possibly related to normalizing or correcting disordered energy homeostasis.
The safety of sodium oxybate in
Abuse Potential, Withdrawal and Fatalities
The abuse of illicit forms of sodium oxybate (GHB) led to concerns of diversion and
GHB
A review of the forensic literature reveals that accurately establishing gamma-hydroxybutyrate-related deaths using laboratory means is a complex matter, requiring samples from cardiac and femoral blood, urine, vitreous humor and stomach contents, and documenting the amount of time that elapsed between the time of death and when fluid samples were obtained. 96
Contraindications and Warnings
Contraindications to sodium oxybate use include patients receiving sedative/hypnotics or those with succinic semialdehyde dehydrogenase deficiency44,64 which is a rare autosomal recessive inborn error of metabolism. 97 Characteristics of the disorder are psychomotor retardation, childhood-onset hypotonia, and ataxia. The enzyme deficiency results in significant accumulation of GHB and GABA. Patients with succinic semialdehyde dehydrogenase deficiency may develop increased adverse effects if they receive sodium oxybate. 63
The occurrence of suicidal ideation has been reported in a case of sodium oxybate abuse. 63 Its occurrence in 1 of 8 subjects was confounded by the fact that this patient also had a known anxiety disorder and oppositional disorder. Hence sodium oxybate should be used with caution in patient with history of preexisting psychiatric symptoms.
Due to the rapid onset of action and strong sedative effect, sodium oxybate should only be ingested at bedtime, and while in bed. 64 Because of the risk of residual sedation, patients should preferably not engage in hazardous activities or activities requiring complete mental alertness or motor coordination, such as driving or operating machinery for at least 6 hours after ingesting sodium oxybate. As with all sedating medications, consideration should also be given to individual patient situations, such as being solely responsible for a small child or being ‘on call’ for any task requiring full mental alertness.
Clinical trials are lacking in the evaluation of the drug in pregnant women; 86 therefore, it should be used only when necessary. Sodium oxybate should be administered with caution in nursing mothers because it is unknown whether the drug is excreted in breast milk. 43
Warning against consumption of alcohol when using sodium oxybate due to the risk of respiratory depression has been adequately emphasized as a black box warning in the package insert.
Administration and Dosages
The recommended initial dose of sodium oxybate is 4.5 g/day divided into 2 equal doses.43,44 The first dose is administered at bedtime, with the patient in bed, due to the drug's rapid absorption and pharmacologic action. The second dose is to be taken within 2.5–4 hours of the first dose because of the elimination half-life of 30–60 minutes. The dosage can be increased by 1.5 g/day every 1–2 weeks until the desired clinical effect has occurred. The maximum dose is 9 g/day given in divided doses. The majority of patients have a clinical response within the dosage range of 6–9 g/day.
Modifications of dosing based on gender are not necessary. 54 The initial dosage of sodium oxybate should be decreased by one-half in patients with liver insufficiency. 43 Sodium oxybate has extensive hepatic first-pass metabolism, which can prolong the elimination half-life in patients with cirrhosis. 98 With liver cirrhosis, the drug has an increased mean AUC, markedly reduced oral clearance, and significant prolongation of the half-life. Repetitive dosing in cirrhotic patients did not result in drug accumulation. Patients with congestive heart failure, hypertension, or renal insufficiency will need to have their daily sodium intake assessed.43,86 Three grams of sodium oxybate contains 0.5 g of sodium, while the 9 g dose contains 1.6 g of sodium.
High-fat meals can significantly reduce the bioavailability of sodium oxybate; therefore, it is advisable to administer the drug on an empty stomach. 54
Sodium oxybate (Xyrem) is available as an oral solution with a concentration of 500 mg/ml.44,86 The kit contains a press-in-bottle adaptor, a 10 mL oral syringe, two 90 mL dosing cups with child-resistant caps, and a medication guide. Each dose requires dilution with 60 mL of water. The patient is instructed to mix both doses before bedtime and to use the diluted solutions within 24 hours. Due to the history of drug abuse with GHB, the Xyrem Success Program was developed for physicians and patients to minimize drug diversion with sodium oxybate.44,86
The program is to be contacted by the prescriber to gain access to a central pharmacy that distributes the medication. The physician is supplied with educational materials before the drug is dispensed. The process then proceeds to specialized prescription forms, physician and patient registration and approval, solving issues of reimbursement, and contacting patients for delivery of the medication along with counseling. Long-term management includes close monitoring of refills and potential adverse effects.
Approved Labeling and Possible Off-Label Uses
Sodium oxybate is a Schedule III controlled substance in the US, approved for the treatment of EDS and cataplexy in patients with narcolepsy. 44 Investigational studies have evaluated its use for management of alcohol withdrawal symptoms and opiate detoxification, 99 fibromyalgia, 100 stimulus-induced paroxysmal drop attacks in Coffin-Lowry syndrome, 101 tardive dyskinesia and bipolar disorder. 102 GHB may have neuroprotective effects in neurodegenerative disorders such as Alzheimer disease. 103 Currently, sodium oxybate is used in various European countries as an adjunct to anesthesia, for treatment of alcohol withdrawal, and for maintenance of alcohol abstinence. 104
Place in Therapy
Rational pharmacotherapy of childhood narcolepsy implies targeting the symptom that is most bothersome to the patient. If daytime sleepiness is severe and cataplexy is less prominent, the initial prescription of modafinil or preparations of methylphenidate and amphetamine seems appropriate, with sodium oxybate as back-up if daytime sleepiness and fragmented sleep persist. When cataplexy is the more disabling symptom, however, consideration should be given to initiating treatment with sodium oxybate at the onset, provided that there are no medical, social, and family contraindications
Future
Despite significant advances in understanding the pathophysiology of narcolepsy, current treatments for human narcolepsy are symptom based. However given the major developments in understanding the neurobiological basis of the condition, new therapies are likely to emerge such as symptomatic endocrine/ transmitter modulating therapies, hypocretin-based therapies and immune-based therapies including steroid therapy, intravenous gamma-globulin, and plasmapharesis.105–108 Development of a long acting sodium oxybate preparation will be helpful in better patient compliance regarding the second dose. Eventually, treatment of narcolepsy–cataplexy is likely to involve hypocretin cell transplant or gene therapy technology. 109 However, these therapies are many years away; it is imperative that neurologists remain aware of future developments, not only out of interest but also because of the implications for treating a lifelong debilitating disease.
Disclosures
The authors report no conflicts of interest.