Abstract
Purpose
The intracytoplasmic morphologically selected sperm injection (IMSI) procedure has been associated with better laboratory and clinical outcomes in assisted reproduction technologies. Less information is available regarding the relationship between embryo aneuploidy rate and the IMSI procedure. The aim of this study is to compare the clinical outcomes and chromosomal status of IMSI-derived embryos with those obtained from intracytoplasmic sperm injection (ICSI) in order to establish a clearer view of the benefits of IMSI in infertile patients.
Methods
We retrospectively analyzed a total of 11 cycles of IMSI and 20 cycles of ICSI with preimplantation genetic diagnosis. The fertilization rate, cleavage rate, embryo quality, blastocyst development, aneuploidy rate, pregnancy rate, implantation rate, and miscarriage rate were compared between the groups.
Results
Similar rates of fertilization (70% and 73%), cleavage (98% and 100%), and aneuploidy (76.9% and 70.9%) were observed in the IMSI and ICSI groups, respectively. The IMSI group had significantly more good quality embryos at day 3 (95% vs 73%), higher blastocyst development rates (33% vs 19%), and greater number of hatching blastocysts (43% vs 28%), cycles with at least one blastocyst at day 5 (55% vs 35%), and blastocysts with good trophoectoderm morphology (21% vs 6%) compared with the ICSI group (P < 0.001). Significantly higher implantation rates were observed in the IMSI group compared with the ICSI group (57% vs 27%; P < 0.05). Pregnancy and miscarriage rates were similar in both groups (80% vs 50% and 0% vs 33%, respectively).
Conclusion
The IMSI procedure significantly improves the embryo quality/development by increasing the implantation rates without affecting the chromosomal status of embryos. There is a tendency for the IMSI procedure to enhance the pregnancy rates and lower the miscarriage rates when compared with ICSI.
Introduction
Since the introduction of the intracytoplasmic sperm injection (ICSI) in 1992, 1 it became clear that the morphology of spermatozoa used for injection was related to fertilization and pregnancy rates. 2 Nowadays, ICSI is used as a mechanism to overcome male factor infertility by selecting motile spermatozoa with normal morphology and motility under a magnification of ×400. However, several studies have shown that subtle morphological malformations of the sperm nucleus are related to low fertilization rates, 3 reduced blastocyst formation, 4 and poor clinical outcomes.5–7 Bartoov et al 8 introduced the motile sperm organelle morphology examination (MSOME) using high-power differential interference contrast optics, allowing the implementation of a new method: the intracytoplasmic morphologically selected sperm injection (IMSI). This high magnification allowed the identification of spermatozoa with a morphologically normal nucleus, which has a symmetrical and oval shape with homogeneous chromatin mass and does not demonstrate more than one vacuole involving less than 4% of the nuclear area. 3 Vacuoles are a possible indicator of sperm DNA damage (fragmentation or denaturation)9,10 and negatively affect human embryo development.
Paternal influences cause about half of the infertile couples to turn to assisted reproduction technology (ART) procedures. Potential predictors of male factor infertility include age, method of fertilization, reactive oxygen species, sperm quality parameters, and DNA fragmentation. Furthermore, recent studies have suggested factors such as body mass index, smoking, male age, and stress as additional causes of infertility. 11 The male factor exerts its influence primarily after embryo genome activation, causing lower blastocyst quality and affecting clinical outcomes. 12 The adverse effects of sperm quality are evident as early as in the pronuclear zygote stage, embryo morphology, and low cleavage speed (early paternal effect) and later related to failure in blastocyst formation (late paternal effect). 13 Paternal effects have been shown to be related to repeated failure of assisted reproduction treatment attempts.14–16 Since the sperm DNA represents half of the offspring's genomic material, a genetically normal sperm is necessary for a successful fertilization, embryo development, and clinical outcomes to produce healthy offspring.17,18 Damage and severe fragmentation of the sperm DNA can lead to errors in the reproductive process that are associated with male infertility.19,20 Various studies have reported a negative effect of high levels of sperm DNA fragmentation on pregnancy rates in ART.21–25 Sperm DNA fragmentation or errors in the first meiotic divisions of the embryo, which are not under the DNA correction mechanisms because the embryo's genome is yet to be activated, can lead to embryo aneuploidy, 26 which is one of the most important contributors to poor laboratory and clinical outcomes of ART. Several studies have independently concluded that human embryos intrinsically contain substantial chromosomal errors27–29 and that this problem is exacerbated as maternal age increases. 30 Souza Setti et al 31 compared the IMSI and ICSI procedures and reported a significantly higher percentage of top quality embryos, implantation and pregnancy rates, and reduced miscarriage rates in patients from the IMSI group compared with those observed in patients from the ICSI group. Furthermore, Knez et al 32 found, in a comparative study between IMSI and ICSI procedures, a trend of higher number of blastocysts per cycle in the IMSI group, but their quality was independent of the procedure of sperm selection.
On the other hand, there is insufficient information regarding the effect of IMSI, compared to ICSI, on the aneuploidy rates in embryos. The study by Figueira Rde et al 33 reported that the total aneuploidy rate was not statistically different between IMSI and ICSI procedures, but there was a significantly increased incidence of sex chromosome aneuploidy in the ICSI embryos. Therefore, this study was conducted to evaluate the overall effect of IMSI compared to ICSI by evaluating the laboratory (fertilization, cleavage, blastocyst development, embryo morphology) and clinical outcomes (pregnancy and implantation rates), as well as the embryo aneuploidy rate, in order to establish a clearer view of the benefits of IMSI in couples with male infertility and its impact on embryo aneuploidy.
Materials and Methods
Patients
A total of 31 cycles of IMSI (n = 11) and ICSI (n = 20) procedures with preimplantation genetic diagnosis (PGD) performed between July 2011 and February 2015 at FERTILAB Laboratory of Assisted Reproduction (Lima, Peru) were analyzed. Cases of azoospermia and egg donation were not included.
The causes for infertility among the patients in the IMSI and ICSI groups were female factor (18% and 45%), male factor (9% and 10%), female and male factor (63% and 45%), and unexplained factor (9% and 0%, respectively). Furthermore, the ICSI and IMSI procedures were recommended by the physician in charge of the patient's fertility treatment and are based on the patient's medical chart, sperm DNA fragmentation, and previous in vitro fertilization (IVF) or ICSI failures.
All cycles and procedures were approved by the Institutional Review Board and the associated Ethics Committee. Written consent was obtained from all patients and their partners included in this study. All experimental procedures were performed according to the Declaration of Helsinki of 1975 and its modifications.
Assessment of sperm DNA fragmentation
Prior to the hormonal stimulation, sperm DNA fragmentation values were evaluated with the sperm chromatin dispersion test 34 using Halosperm® Kit (Halotech DNA). Briefly, sperm samples from each patient, containing not <5 million and >10 million spermatozoa per milliliter after dilution, were used. The kit contains aliquots of agarose gel in Eppendorf tubes. Each semen sample was processed after the agarose gelled (by immersion in a water bath at 90°C for five minutes). When the Eppendorf tubes reached a temperature of 37°C (five minutes at 37°C in a dry atmosphere), 25 μL of the sperm samples was added and gently mixed. Then, 20 μL of this mixture was placed on precoated slides and covered with 22 × 22-mm coverslides. The slides were maintained at 4°C for five minutes to produce a microgel containing embedded spermatozoa. The coverslides were gently removed, and the slides were immersed in a previously prepared acid solution (80 μL of HCl added to 10 mL of distilled water) for seven minutes. After removal from this solution, the slides were incubated for 25 minutes in 10 mL of lysing solution (provided in the Halosperm kit). The slides were then rinsed in distilled water, dehydrated in three concentrations of alcohol (70%, 90%, and 100% vol) for two minutes in each concentration, and were either stored (storage was possible for several months in optimal conditions) or processed immediately with a staining solution for 10 minutes with continuous airflow. Staining was performed with 1:1 (vol/vol) Wright's solution (Merck) and phosphate-buffered saline solution (Merck). The slides were rinsed in tap water, allowed to dry at room temperature, processed for upright or inverted bright-field microscopy at ×100, and covered with 22 × 22-mm coverslides. Operators scored ≥500 spermatozoa for each patient according to the patterns established by Fernández et al. 34 Strong staining is preferred to visualize the dispersed DNA loop halos. Removal of sperm nuclear proteins results in nucleoids with a central core and a peripheral halo of dispersed DNA loops. The sperm tails remain preserved. The acid treatment produces DNA unwinding that is restricted in those nuclei with high levels of DNA strand breakage. After the subsequent lysis, sperm nuclei with fragmented DNA produce very small or no halos of dispersed DNA. However, nuclei without DNA fragmentation release their DNA loops to form large halos.
Ovarian stimulation and oocyte collection
The menstrual cycles of patients were stimulated using recombinant follicle-stimulating hormone (rFSH; Gonal®, Merck Serono Laboratories) according to previously established stimulation protocols, 35 and starting on day 2 of the menstrual cycle until when at least three follicles reached ~18 mm in diameter. The oocyte recovery was performed by vaginal ultrasound under general anesthesia with intravenous injection of 200 mg of propofol (Diprivan® 1% P/V; AstraZeneca Laboratories) 36 hours after the intramuscular application of human chorionic gonadotropin (Pregnyl®, Ferring Pharmaceutical).
During the follicular aspiration procedure, the oocytes were recovered in Global® N-(2-Hydroxyethyl)piperazine-N'-(2-ethanesulfonic acid)) (HEPES)-buffered medium (LifeGlobal) supplemented with 10% vol/vol serum substitute supplement (SSS; Irvine Scientific). After retrieval, cumulus–oocyte complexes were manually trimmed of excess cumulus cells and cultured in ~200 μL drops of Global® Fertilization medium (LifeGlobal) supplemented with 10% vol/vol SSS under mineral oil (Lite Oil®, LifeGlobal) at 37°C and an atmosphere containing 6% CO2, 5% and 89% N2 for five hours before the IMSI or ICSI procedure.
Evaluation and preparation of sperm samples
On the day of the ICSI or IMSI procedure, semen samples were collected by masturbation into sterile cups after three to five days of abstinence. After semen liquefaction for 30 minutes at room temperature, the concentration, progressive motility, and morphology of spermatozoa were assessed according to the World Health Organization criteria. 36 Motile spermatozoa were separated from the seminal plasma by centrifugation at 300× g for 10 minutes through 1.0 mL of 95% and 45% isolate gradients (Irvine Scientific). The pellet was washed once by centrifugation for three minutes and later resuspended in pregassed Global® Fertilization medium supplemented with 10% vol/vol SSS for the microinjection procedures.
Sperm selection for IMSI procedure
For sperm retrieval and immobilization, a glass bottom dish (Willco Wells) with one 5-μL microdroplet of polyvinylpyrrolidone (PVP; Irvine Scientific) and another microdroplet of Global® HEPES-buffered medium supplemented with 10% vol/vol SSS under mineral oil were prepared. Spermatozoa were selected under ×10,160 magnification in an inverted microscope (IX71; Olympus) equipped with a Normarski differential interference contrast optics, ×100/1.30 oil objective lens, and a variable zoom lens. Spermatozoa were retrieved from the medium and then transferred to the PVP microdroplet to be immobilized by tail crushing. Afterward, they were classified according to their morphology and grade of vacuolization pattern (types I–IV) as described by Vanderzwalmen et al 4 (Fig. 1). The spermatozoa with a normal morphology and presenting a vacuolization pattern of type I or II were used for injection into the oocytes.
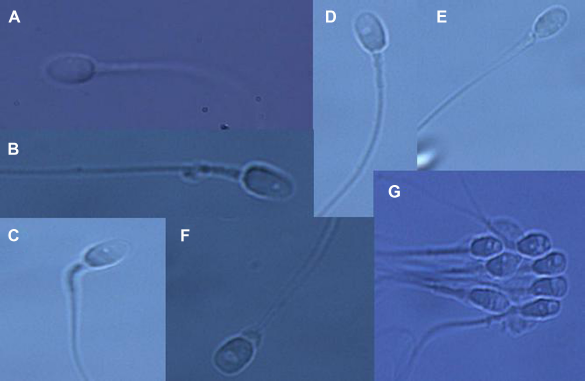
Grading of spermatozoa into four groups according to the presence or size of vacuoles. Grade I: normal form and no vacuoles (A). Grade II: normal form with less than or equal to two small vacuoles (B, C). Grade III: normal form with more than two small vacuoles or at least one large vacuole (D, E). Grade IV: large vacuole and abnormal head shapes or other abnormalities (F, G).
Oocyte preparation and microinjection
For IMSI and ICSI procedures, the oocytes were denudated by incubation for ~30 seconds in 80 UI/mL of hyaluronidase (LifeGlobal). The oocytes were then aspirated in and out of a glass pipette to allow the complete removal of the cumulus and corona cells. Only the metaphase II oocytes were selected for sperm injection, which was performed five hours after oocyte retrieval. For sperm selection, a number of suitable spermatozoa according to the number of retrieved oocytes were selected by the MSOME method for IMSI or by morphology for ICSI.
Microinjection was performed in a plastic culture dish containing four microdroplets of 10 μL of Global® HEPES-buffered medium supplemented with 10% vol/vol SSS around a 20 μL microdroplet of PVP at the center and covered with mineral oil. The selected spermatozoa were transferred into the PVP microdoplet, and the oocytes were placed on the surrounding droplets. A single spermatozoon was aspirated by the injection pipette and, after crushing the tail one more time, injected into the oocyte as described elsewhere. 37 Subsequently, all injected oocytes were cultured at 37°C in an atmosphere of 6% CO2, 5% O2, and 89% N2.
Fertilization, embryo culture, and transfer
Fertilization was evaluated 16-18 hours post injection (day 1) by the presence of two pronuclei. The zygotes were individually cultured under mineral oil, in 10-μL droplets of Global® medium supplemented with 10% vol/vol SSS from day 1 to day 3. On day 3, the embryos were moved to fresh 10-μL droplets of Global® medium supplemented with 10% vol/vol SSS and cultured for two more days up to the transfer day in blastocyst stage.
On day 3, the embryos were evaluated for cell number, fragmentation, and multinucleation. Good quality day 3 embryos were defined as those with six to eight cells and ≤10% of fragmentation. Good quality blastocysts were defined as having an inner cell mass (ICM) and trophoectoderm (TE) type A or B, according to Gardner's scoring system. 38 The ICM score was evaluated as follows: type A = compact area, many cells present; type B = cells are loosely grouped. The TE was scored as follows: type A = many cells forming a tight epithelial network of cells; type B = few cells forming a loose network of cells.
The embryos were transferred on day 5 using an Emtrac embryo transfer catheter (Gynétics) that had been previously washed with the culture medium. The catheter was completely filled with the culture medium, and the embryos were filled in the last 10 μL of the catheter. All transfers were performed according to the methods previously described by Mansour. 39 The blastocysts that were not transferred were cryopreserved or discarded according to their morphology.
Embryo biopsy, fixation, and FISH analysis
In patients with PGD indication, one cell per embryo was biopsied at the third day after insemination following a protocol described elsewhere. 40 Individual embryos were placed into calcium/magnesium-free media (PGD Biopsy Medium; LifeGlobal), and one nucleated blastomere was mechanically removed through a hole in the zona pellucida opened using Tyrode's acid (LifeGlobal) solution. After biopsies, the embryos were rinsed thoroughly and returned to culture under mineral oil, in 10-μL droplets of Global® medium supplemented with 10% vol/vol SSS.
The blastomeres were fixed individually following routine protocols to minimize signal overlap and loss of micronuclei. 41 PGD analysis was performed by fluorescence in situ hybridization (FISH) using probes specific for 12 chromosomes 8, 13, 14, 16, 18, 20, 21, 22 (Abbott Laboratories), X, Y, 15, and 17 (Cellay Inc.) following the manufacturer's instructions.
Pregnancy determinations
The biochemical pregnancy was assessed 14 days after the embryo transfer by measuring the human chorionic gonadotropin beta subunit in blood. The clinical pregnancy was determined by transvaginal ultrasonography to detect gestational sacs and fetal heartbeats at approximately 21 and 28 days after transfer, respectively.
Statistical analysis
Statistical analysis was carried out using the statistical package Stata 12 (StataCorp). Data are represented as mean ± standard deviation. Group comparisons were made using the χ2-test and Student's t-test. P values <0.05 were considered for showing statistically significant differences.
The normal fertilization rate was calculated from the number of zygotes with two pronuclei of IMSI/ICSI and divided by the number of injected oocytes by 100. The cleavage rate was calculated from the number of embryos with six or more cells at day 3 and divided by the total number of zygotes by 100. The implantation rate was calculated from the number of gestational sacs observed by ultrasound at day 21 post transfer and divided by the total number of transferred embryos by 100. The rate of clinical pregnancy was calculated from the number of patients with at least one gestational sac divided by the total number of transferred embryos by 100. The miscarriage rate was defined as the number of pregnancies with a total loss of gestational sacs before 20 weeks of gestation divided by the numbers of pregnancies by 100. The aneuploidy rate was defined as the number of genetically abnormal embryos, analyzed by the 12 chromosomes, divided by the total number of biopsied embryos by 100.
Results
During a period of three years and seven months, 31 couples with an indication of a 12-chromosome PGD were involved in 11 IMSI cycles (IMSI group) and 20 ICSI cycles (ICSI group). There was no difference in terms of age of females and males in both evaluated groups (Table 1). The mean number of days of stimulation were significantly higher in the ICSI group compared to that in the IMSI group (8.35 ± 1.39 vs 7.18 ± 0.75 days, respectively; P < 0.01); the same situation was observed for the mean rFSH treatment dose when comparing the IMSI and ICSI groups (1636 ± 322 vs 1207 ± 339 IU/L, respectively; P < 0.01) (Table 1). The parameters of morphology, volume, concentration, and progressive motility were similar between the IMSI and ICSI cycles (Table 2). There was no difference in the DNA fragmentation percentages between the IMSI and ICSI groups (20% vs 18%, respectively; P: not significant [NS]).
Characteristics of study patients.

Statistically significant difference between IMSI and ICSI groups (P < 0.001).
Seminal parameters in the IMSI and ICSI groups.

Laboratory outcomes obtained from both groups are shown in Table 3. A total of 67 and 139 oocytes were retrieved from women of the IMSI and ICSI groups, of which 60 and 127 oocytes were injected, respectively. The normal fertilization rate (2PN) was similar in both evaluated groups (IMSI group: 70% vs ICSI group: 73%). There was no difference in the cleavage rate, but the percentage of good quality embryos at day 3 was higher in the IMSI group (95% vs 73%; P = 0.02). Blastocyst development rate was higher in the IMSI group than in the ICSI group (33% and 19%, respectively; P < 0.01). There was a statistically significant tendency toward a higher number of cycles with at least one blastocyst in the IMSI group (54% and 35.1%, P < 0.01) and a lower number of cycles with all embryos arrested at the morula stage or earlier (0% and 45%, P < 0.01). When blastocysts developed, they did not differ in their quality according to the procedure of sperm microinjection, except for hatching blastocysts (43% and 28% for IMSI and ICSI, respectively; P < 0.01) (Fig. 2). There were approximately the same proportions of full (57% and 67%) and expanded (0% and 5%) blastocysts in the current IMSI and ICSI group cycles, respectively. The IMSI and ICSI groups presented approximately the same proportions of blastocysts according to the morphology of the ICM, but a tendency toward a better TE morphology was observed in the IMSI group (Fig. 3). Euploidy and aneuploidy rates were similar in both study groups (23% vs 29% and 77% vs 71%, respectively; P > 0.05) (Table 5).
Comparison of laboratory results between IMSI and ICSI groups.

Statistically significant difference between IMSI and ICSI groups (P < 0.001).
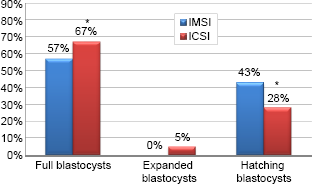
Blastocyst quality in the IMSI and ICSI cycles.

Comparison of ICM and TE from blastocysts between IMSI and ICSI groups.
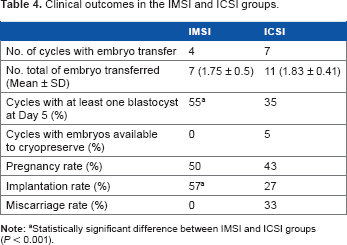
Clinical outcomes obtained from both groups are shown in Table 4. Cancelation rates, mainly because of abnormal PGD or bad quality embryos, were similar in both groups (64% and 65%). The mean number of embryos transferred per patient was similar in both groups (1.75 ± 0.5 and 1.83 ± 0.41; P: NS). The clinical pregnancy rate per transfer in the IMSI group tended to be higher than that in the ICSI group (80% vs 50%, P = 0.559), although not statistically significant. All pregnancies achieved by the blastocyst transfer were singleton, and one pregnancy in the ICSI group terminated with an abortion (0% vs 33%, P = 0.439). The implantation rate was higher in the IMSI group than in the ICSI group (57% and 27%; P < 0.01); miscarriage rates were similar in both groups (Table 4).
Clinical outcomes in the IMSI and ICSI groups.
Discussion
Cleavage rate, multinucleation, and embryo morphology at the cleavage stage have been shown to be important markers of embryo quality and viability during in vitro culture.42,43 After the introduction of extended culture to blastocyst stage, improved clinical outcomes have been reported because of better embryo selection and better synchronization between the embryo developmental stage and uterine environment. However, with a better sperm selection, we can be confident that embryos will have better chances of achieving the blastocyst stage, thereby increasing their chances of implantation.
Classical parameters of sperm quality are not always related to a good prognosis and fertility, since sperm DNA fragmentation and head vacuoles might be present. This problem can be dealt with by the use of sperm selection under high magnification or IMSI. The results from this study indicate that patients undergoing ICSI would benefit from sperm selection under high magnification. The benefits of IMSI to improve outcomes in cases of male infertility patients have already been published.4,6,32,44,45
In the present study, couples included in both study groups showed similar baseline characteristics (Tables 1 and 2), and thus there were no confounding factors regarding sperm parameters, oocyte characteristics, and infertility etiology. The IMSI and ICSI differ only in terms of treatment characteristics, the total doses of rFSH, and the number of days of stimulation (1636 ± 322 vs 1207 ± 339 and 8.35 ± 1.39 vs 7.18 ± 0.75, respectively, P < 0.01). This difference does not qualify as a confounding factor since several recent studies reported that gonadotropin doses and duration of the protocol of controlled ovarian stimulation are independent of laboratory and clinical outcomes.46,47 Increasing the starting dose of FSH stimulation does not yield a significant improvement in oocyte or embryo yield or pregnancy rates observed following such an upward FSH dose adjustment. Furthermore, a shorter protocol allows obtaining IVF results similar to those of a classical long protocol with a high dose of exogenous gonadotropins, as shown by recent studies and meta-analysis.48,49 In the present study, there were no differences in embryo quality until day 3 of development; however, after the eight-cell stage, improvements occurred in the development rate and in the blastocyst quality for IMSI patients, in accordance with previous studies that suggested that the beneficial effect of MSOME can be observed at the later stages of embryo development.4,31,44,50 Therefore, the IMSI procedure appears to positively affect embryo development closely to and after the embryo genome activation, representing an early and late paternal effect. Our hypothesis is that the presence of vacuoles and their size influence the outcome of an ART cycle by an early and late embryo development action. Early paternal effect is a consequence of sperm cytoplasmic content, which can influence fertilization and early stages of embryo development probably until the embryo genome activation at the four- to eight-cell stage with a subsequent effect on cellular division and embryo fragmentation, thereby affecting embryo quality. 13 The increased top quality embryo rate observed in the IMSI group is due to the early paternal effect of the sperm selected by the MSOME method, which, in the absence of nuclear vacuoles, can be observed at the early stages of embryo development. 51 These effects may be genetic or epigenetic, since a weak transcriptional activity has been detected in human male pronuclei52,53 that is crucial for nucleolar development. 54 More research is needed in order to ensure if this phenomenon that causes a lag in male pronuclear development is the origin of the observed early paternal effect on embryo development.
The elevated blastocyst development rate, high proportion of good quality blastocysts, and high proportion of cycles with at least one blastocyst in the IMSI group are an indication that a late paternal effect is present. Various studies have proved a positive association between sperm nuclear vacuoles and sperm DNA damage9,10 and vacuoles and chromatin remodeling during the sperm maturation process.55,56 An error in the integrity of sperm chromatin can lead to abnormal embryo development or failure to progress to the blastocyst stage57,58 because the sperm chromatin is highly condensed and its insoluble nature plays a protective role during the transfer of paternal genetic information through the male and female reproductive tracts.59–61 Sperm DNA fragmentation effects can be present at different stages of the ART procedure, starting from preimplantation development of the embryo to the birth of healthy offspring. Several studies proved the effect of sperm DNA damage and its correlation with crucial laboratory and clinical outcomes, including fertilization rates, embryonic development, implantation, pregnancy, and miscarriage rates.62–64 If sperm selection had been done on the same population who underwent IMSI, but using the classic ICSI parameters, the odds of selecting spermatozoa with large nuclear vacuoles (LNV) or multiple ones would have been very high, thereby severely affecting posterior embryo development. Moreover, some studies observed a significant improvement in the implantation rate after IMSI in the presence of male factor-related infertility.65,66 The higher blastocyst development rate in the IMSI procedure is related to the fact that in this group approximately 60% of the cycles have at least one blastocyst and few embryos were arrested at the morula stage or earlier; with a higher chance of developing a blastocyst, there will be a lower chance of an embryo to be arrested through development. The blastocysts from the IMSI group were mostly high quality ones, presenting better quality than those from the ICSI procedure that had more low quality blastocysts (67% vs 57% of full blastocysts). Furthermore, the blastocysts from the IMSI group showed a tendency for better ICM quality and a statistically significant difference for a better TE quality by a 15% increment on grade A when compared with those from the conventional ICSI. The initial contact between the blastocyst and maternal tissues is by adhesion of the trophoblast to the uterine epithelium and the cell-to-cell interaction. This interaction is believed to be critical for implantation, so the trophoblast quality highly influences the chances of embryo implantation.67–69 Therefore, by generating a better TE quality blastocyst, the IMSI method has more likelihood of implantation and pregnancy with a lower miscarriage rate, which is in agreement with our findings. The role of the TE is better understood during and after implantation as it plays a key role in apposition, adhesion, and invasion of the endometrium, thus allowing the blastocyst to embed in the uterus. The ICM quality should not be disregarded since it would become the primordial structure of the fetus,70,71 and thus, a low or bad quality ICM could result in an unembryonated gestational sac. Despite the fact that the implantation rate of the IMSI group is higher, we observe no difference in the miscarriage rate. This can be due to multiple factors unrelated to embryo quality such as genetic, immunologic, and anatomic characteristics of the patient. We should not forget that this study has very few patients, so further studies should be addressed in order to confirm the benefits in clinical outcomes observed (pregnancy, implantation, and miscarriage rates).
Several studies indicate that the injection of DNA-damaged or vacuolated spermatozoa is related to a blockage of embryo development before and after implantation, reflecting a late paternal effect,9,51,72 providing more evidence to support the fact that the ICSI method may yield lower embryo quality and clinical outcomes than IMSI because of the chance of injecting highly vacuolated and DNA-damaged sperm into the oocytes, 73 since sperm DNA integrity, chromosomal constitution, and nuclear morphology cannot be assessed in the sperm cell used for ICSI. An adverse late paternal effect can be characterized by poor embryo development to the blastocyst stage, implantation failure, and pregnancy loss as observed in the ICSI group. Furthermore, a high percentage of abnormal spermatozoa with LNV, according to MSOME criteria, was observed in male patients older than 40 years, 74 and up to 65% of spermatozoa that were deemed suitable for ICSI by conventional methods were subsequently deselected after high magnification analysis (MSOME). 75 Since the mean male age in both the study groups was approximately 42 years and because male factor infertility is mostly related to advanced paternal age, it is safe to assume that in the cases of ICSI, the reduced potential of success can be caused by the injection of abnormal sperm with LNV. In addition, LNV are closely associated with chromatin condensation failure and a potential increase in susceptibility to DNA damage during the IMSI/ICSI procedure,76,77 thereby explaining the reduced potential of success of the ICSI procedures.
The IMSI sperm selection method requires more time and the spermatozoa are present for a prolonged time in the PVP microdroplet. This may be damaging to the sperm cells since PVP can cause significant damage to sperm membranes and induce the acrosome reaction, thereby reducing the fertilization rate by causing injury to the ultrastructure of the mitochondria and sperm tail.78,79 Some studies report that there are risks of using the PVP microdroplet and that the prolonged exposure of sperm cells can lead to DNA damage.80,81 Consequently, the IMSI method damages the sperm DNA, since the average time to select a vacuole-free or less than 4% of nuclear space is twice as that for conventional ICSI.10,74
On the other hand, a study on embryo aneuploidy reported that the proportion of abnormal embryos is increased with the severity of male factor condition, but the type of defect depends on the sperm characteristics. 82 In addition, less information is available regarding the comparison of aneuploidy rate between ICSI and IMSI, with one study reporting that autosomal aneuploidy was not affected by sperm selection method, but there was a higher sex chromosomal aneuploidy in IMSI. 33 The aneuploidy rates obtained in this study are in agreement with previous studies that report embryo aneuploidy in cleavage stage and blastocysts utilizing FISH 83 and comparative genomic hybridization (CGH). 84
However, our findings show that the aneuploidy rate remains the same between the IMSI and ICSI groups, implying that the time used for selecting the suitable MSOME grade spermatozoon on a PVP microdroplet is not detrimental to DNA integrity and does not lead to lower embryo quality or lower blastocyst development rates and that IMSI presents no apparent adverse effects. Furthermore, since no damage to DNA is done, the IMSI procedure is adequate for patients with elevated DNA fragmentation in order to select the sperm with better morphology, but without increasing the underlying DNA fragmentation since it is related to poor semen parameters85,86 and clinical outcomes. 87 Sperm aneuploidy was not evaluated since the sperm DNA fragmentation test is simpler, easier, and less expensive and its results have been proven to be correlated to sperm parameters and clinical outcomes.85–87
It is important to point out that aneuploidy is independent of embryo quality since a good quality blastocyst can be aneuploid and implanted, but it will result in a miscarriage. 88 This phenomenon was also observed in our PGD reports (Table 5), where a significant number of embryos achieving the blastocyst stage were aneuploid and unsuitable for transfer (16% and 22% for IMSI and ICSI, respectively; P: NS). However, it is important to indicate that the PGD diagnosis by FISH provides information regarding only the 12 chromosomes, in contrast to a method such as CGH that has a 24-chromosome resolution, which is associated with better clinical outcome and more accurate PGD results. 84 We suggest that further studies should be conducted with CGH in order to obtain precise information about the effect of IMSI on chromosome aneuploidy.
Aneuploidy rate in patients from IMSI and ICSI cycles with PGD.

Conclusion
In conclusion, the IMSI procedure not only significantly improves embryo morphology and implantation and pregnancy rates but also presents a tendency to increase day 3 embryo quality, reduce miscarriage rates, and maintain an equal aneuploidy rate as compared with ICSI. The IMSI procedure provides more viable blastocysts without chromosomal abnormalities as a consequence of a greater blastocyst development rate that will increase the chances of having genetically normal blastocysts. Despite the reduced number of cycles, these data justify the clinical application of IMSI. Therefore, the IMSI technique can be considered an alternative to bypass the gap created by male factor infertility in couples trying to achieve a successful ART cycle outcome. Well-designed prospective studies, in which variables are controlled and include a greater number of cycles, are needed in order to corroborate the tendencies observed in this retrospective study and to reveal more advantages regarding the application of IMSI in ART.
Author Contributions
Reported initial concept and designed the study: DL. Analyzed data: DL and JGF. Responsible for the original and revised drafts of the manuscript: DL, RH, JD-C, RR, PZ, LV, JG-F. All authors revised and approved the final manuscript.
