Abstract
Objective
Ketoconazole (KCZ) is an anti-fungal agent extensively used for clinical applications related to its inhibitory effects on adrenal and testicular steroidogenesis. Much less information is available on the effects of KCZ on synthesis of steroid hormones in the ovary. The present study aimed to characterize the in situ effects of KCZ on steroidogenic enzymes in primary rat ovary cells.
Methods
Following the induction of folliculogenesis in gonadotropin treated rats, freshly prepared ovarian cells were incubated in suspension for up to four hours while radiolabeled steroid substrates were added and time dependent generation of their metabolic products was analyzed by thin layer chromatography (TLC).
Results
KCZ inhibits the P450 steroidogenic enzymes in a selective and dose dependent manner, including cholesterol side-chain cleavage cytochrome P450 (CYP11A1/P450scc), the 17α-hydroxylase activity of CYP17A1/P450c17, and CYP19A1/P450arom, with IC50 values of 0.3, 1.8, and 0.3 μg/mL (0.56, 3.36, and 0.56 μM), respectively. Unaffected by KCZ, at 10 μg/mL, were the 17,20 lyase activity of CYP17A1, as well as five non-cytochrome steroidogenic enzymes including 3β-hydroxysteroid dehydrogenase-δ5-4 isomerase type 1 (3βHSD1), 5α-reductase, 20α-hydroxysteroid dehydrogenase (20α-HSD), 3α-hydroxysteroid dehydrogenase (3α-HSD), and 17β-hydroxysteroid dehydrogenase type 1 (17HSD1).
Conclusion
These findings map the effects of KCZ on the ovarian pathways of progestin, androgen, and estrogen synthesis. Hence, the drug may have a potential use as an acute and reversible modulator of ovarian steroidogenesis in pathological circumstances.
Introduction
Ketoconazole, (KCZ, Nizoral®) is an anti-mycotic imidazole drug known to inhibit the activity of several steroidogenic enzymes. As such, the drug has been clinically exploited in the last decades to suppress steroidogenesis in conditions like Cushing's syndrome, prostate cancer, precocious puberty, and hirsutism.1–4 Therefore, it is not surprising that the drug's effects on the adrenal and testis were thoroughly investigated.5–7 In contrast, there are fewer studies on the effects of KCZ on ovarian steroidogenesis and its potential effects related to fertility treatments. For example, we and others have previously found that administration of a low dose of KCZ to polycystic ovarian syndrome patients during superovulation attenuated ovarian folliculogenesis 8,9 and better controlled the hyperstimulated response to gonadotropins. 8 In addition, KCZ was also shown to improve clomiphene responsiveness in PCOS patients. 10 In view of the potentially beneficial use of KCZ in reproduction, several studies attempted to analyze the biochemical basis for the drug effect on the ovary.8,11–18 However, the use of multiple experimental models yielded incomplete and occasionally contradictory information that led us to seek a model providing a complete view of the drug effects on the entire ovarian steroidogenic enzymatic cascade starting from cholesterol down to estradiol synthesis. To this aim, we used freshly prepared ovarian cells from either immature rats or females induced to ovulate by gonadotropin administration. Freshly isolated cells were then incubated with different steroid substrates and the effect of KCZ was assessed on each of the enzymatic activities.
Materials and Methods
Hormones, Radiochemicals, and Reagents
Pregnant mare serum gonadotropins (Equine Chorionic Gonadotropin; eCG; Gestyl) and human chorionic gonadotropin (hCG; Pregnyl) were purchased from Organon (Oss, Holland). Steroid hormones were purchased from Sigma (St. Louis, MO, USA); [1,2,6,7-3H]-progesterone (109 Ci/mmol), [7-3H]-pregnenolone 22 Ci/mmol), [1,2-3H]-17α-hydroxyprogesterone (50 Ci/mmol), [2,4,6,7-3H]-estrone (98 Ci/mmol), and [1β,2β-3H]-testosterone (53 Ci/mmol) were purchased from New England Nuclear (Boston, MA, USA); [1,2-3H]-25-hydroxycholesterol (47 Ci/mmol) was custom-prepared by Amersham International (Little Chalfont, United Kingdom). [3H]-pregnenolone was purified by thin layer chromatography (TLC) after extraction of culture media from [3H]-progesterone-fed cells. 19 KCZ (R41,400, Janssen Pharmaceutica, Inc.) base powder, was a gift from Abic Ltd. Pharmaceutical and Chemical Industries (Netanya, Israel). Insulin (I-5500), transferrin (T-4515), hydrocortisone (H-4001), collagenase type V, and DNase type I, were obtained from Sigma Chemical Co. (St. Louis, MO). Cyanoketone (R-103-UH) was obtained from Sterling Drug Inc. (Rensselaer, NY).
Animals
Intact immature female rats (22-25 days old) from the Hebrew University Wistar-derived colony were provided with water and rat chow ad libitum and housed in air-conditioned rooms illuminated for 16 hours per day. The animals were sacrificed by cervical dislocation. All protocols had the approval of the institutional committee on animal care and use (IACUC), The Alexander Silberman Institute of Life Sciences, The Hebrew University of Jerusalem.
Cell Preparation and [3H]-Steroid Metabolism Assays
The different protocols for preparation of granulosa cells following administration of gonadotropins to immature rats used needle pricked ovaries as previously described.20,21
CYP11A1/P450scc and 20α-HSD activity assays. Granulosa-lutein cells (4 x 10 4 ) were prepared from eCG-pretreated rat (s.c. 15 i.u.), 22 hours after hCG administration (i.p., 4 i.u.). For the P450scc assay, cells were incubated with KCZ (0.1-1 μg/mL) and 10 μM cyanoketone, 30 minutes prior to the addition of 1 μM [3H]-25-hydroxycholesterol for a further four-hour incubation. For assay of 20α-HSD activity, cells were similarly preincubated with 10 μg/mL KCZ prior to the addition of 1 μM [3H]-progesterone, and further incubated for 45 minutes.
3βHSD1, 5α-reductase, 3α-HSD, and 17HSD1 activity assays. Granulosa cells were prepared from prepubertal rat, 44 hours after administration of eCG. KCZ (10 μg/mL) was added 30 minutes prior to adding 1 μM [3H]-pregnenolone (3βHSD1 assay) or 1 μM [3H]-estrone (17HSD1 assay), and further incubated for two and six minutes, respectively. To assay 17HSD1, we preferred to use estrone as androstenedione is rapidly converted to 5α-androstanedione (data not shown) by the high prepubertal ovarian 5α-reductase. 22
CYP17A1/P450c17 (17α-hydroxylase and 17,20 lyase) activity assays. Whole ovarian cell suspension was prepared from prepubertal rat examined 24 hours after eCG (ie, FSH + LH) administration. Owing to the rapid metabolism of [3H]-progesterone to pregnanolone, we assessed the effect of KCZ on the dual activities of CYP17A1/P450c17, using [3H]-pregnanolone as substrate added following a preincubation (30 minutes) with KCZ (0.1-10 μg/mL, 2.5 x 10 5 cells). For assay of the 17,20 lyase cellular activity, cells as above were incubated for 10 minutes with [3H]-17α-hydroxyprogesterone substrate; excessive androstenedione (50 μM) was added five minutes prior to the addition of the radioactive substrate. Cells treated with KCZ (10 μg/mL) underwent preincubation as above.
CYP19A1/P450arom activity assay. Granulosa cells were prepared from prepubertal rat, 44 hours after administration of eCG. CYP19A1 activity was determined by measuring the stereospecific release of tritium to produce 3 H2O when [1β,2β- 3 H]-testosterone (0.25 μM, 1.5 x 10 5 cpm in 300 μL medium and 2.5 x 10 5 cells) is aromatized to 17β-estradiol.23,24 After four hours, the cell suspension was transferred into plastic RIA tubes for quantification of aromatization dependent release of 3 H2O as previously described.23,24
Steroid metabolism. Assay of [3H]-steroid metabolism was assessed using 5-8 x 10 5 cells incubated in 500 μL of DMEM/F-12 (1:1) medium containing KCZ added 30 minutes prior to the addition of the steroid tracer. At the indicated time points, 100-200 μL aliquots of the cell suspension were withdrawn into 500 μL diethylether for extraction and TLC analysis of the [3H]-steroids metabolites. 22
Results
Figure 1 depicts the steroidogenic pathways previously characterized by us and others in cells of the immature rat ovary.19,25,26 Following the key step catalysis of cholesterol conversion to pregnenolone by CYP11A1, we used the 3βHSD1 product, progesterone, for further analysis of three potential pathways: first, progesterone can be substrate for the activity of 5α-reductase (Fig. 1c) yielding pregnanolone; this pathway prevails in the immature rat ovarian cells.19,22 Second, the 20α-HSD activity (Fig. 1j) transforms progesterone to the much less active 20α-OHP in granulosa-lutein cells.
27
Third, in this ovarian cell model, the slow activity rate of CYP17A1 does not allow the demonstration of progesterone processing to androgens hence, this enzyme activity can be measured once pregnanolone is fed as substrate (Fig. 1e-g).
22
Aromatization of androgens carried by CYP19A1/P450arom in gonadotropin-primed granulosa cells can be assessed upon addition of testosterone substrate (Fig. 1m). Finally, we also assessed the activity of 17HSD1 normally required for conversion of estrone to 17β-estradiol (Fig. 1n).
Ovarian steroidogenic pathways and effects of KCZ. This figure describes the steroidogenic pathways in the ovary and summarizes the results of KCZ effects on enzymatic activities. Each enzymatic reaction is marked by a letter in the order of its evaluation and description in the Results section. Thick horizontal lines indicate inhibitory loci of KCZ. The authentic steroid names denote the generic equivalent as following: 20α-hydroxy-4-pregnen-3-one (20α-hydroxyprogesterone; 20α-OHP); 20α-hydroxy-5α-pregnan-3-one (20α-OH-pregnanone); 5α-pregnane-3α,20α-diol (pregnanediol); 5α-pregnane-3,20-dione (pregnanedione); 3α-hydroxy-5α-pregnan-20-one (pregnanolone); 17α-hydroxyprogesterone (17α-OHP); 17α-hydroxy-5α-pregnane-3,20-dione (17α-OH-pregnanedione); 3α,17α-dihydroxy-5α-pregnan-20-one (dihydroxypregnanone); 5α-androstane-3,17-dione (androstanedione); androst-4-ene-3,17-dione (androstenedione); dihydrotestosterone (DHT); 5α-androstane-3α,17β-diol (androstanediol). Enzymes in order of their evaluation are denoted as following: CYP11A1 (side-chain cleavage of P450; P450scc), 3βHSD1 (3β-hydroxysteroid dehydrogenase-δ isomerase type 1), 20α-steroid dehydrogenase (20α-HSD); 3α-steroid dehydrogenase (3α-HSD); CYP17A1 (17α-hydroxylase/17,20 lyase; P450c17), CYP19A1 (aromatase; P450arom), and 17HSD1 (17β-hydroxysteroid dehydrogenase type 1).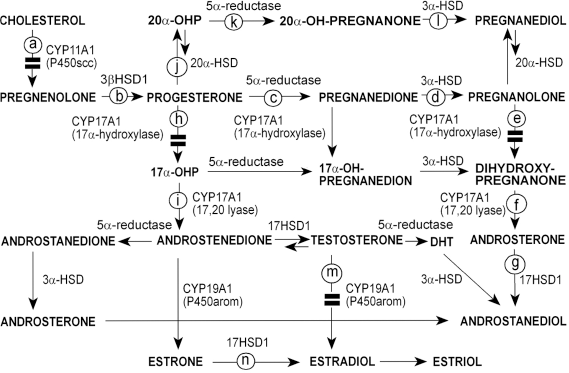
CYP11A1
The effect of KCZ on CYP11A1 activity was assessed in granulosa-lutein cells showing a dose dependent inhibition of hydroxycholesterol conversion to pregnenolone (KCZ IC50 = 0.3 μg/mL) (Fig. 2). In the absence of KCZ, pregnenolone accumulation was evident because of the presence of cyanoketone mediated inhibition of 3βHSD1.
Inhibitory effect of KCZ on CYP11A1 activity. Conversion of [3H]-hydroxycholesterol (1 μM) to pregnenolone was measured in granulosa-lutein cells incubated in the presence of cyanoketone (10 μM, a 3β-HSD inhibitor) without KCZ (NO ADD) or KCZ doses. Following four-hour incubation, the steroids were extracted and analyzed by TLC.
3βHSD1, 5α-Reductase, and 3α-HSD
To determine the conversion of pregnenolone to other progestins initiated by 3βHSD1 (Figs. 3b-d), we examined granulosa cells expressed in eCG primed prepubertal animals, which are highly steroidogenic in follicles approaching their first ovulatory LH surge. Figure 3 shows that KCZ did not affect the 3βHSD1 activity consuming 95% of the
3
H-pregnenolone in 6 minute incubation. As expected, the terminal steroid metabolite, pregnanolone, accumulates because of the high 5α-reductase and 3α-HSD activities in the prepubertal rat ovary.
22
KCZ did not inhibit the latter two enzymes.
KCZ does not affect activities of 3βHSD1, 5α-reductase, or 3α-HSD. Granulosa cells were retrieved 44 hours after eCG administration and incubated with [3H]-pregnenolone (1 μM) for the indicated times in the absence (NO ADD) or presence of KCZ and the steroid metabolites were analyzed by TLC.
20α-HSD
In order to investigate the conversion of [3H]-progesterone by 20α-HSD (Fig. 4j) we used luteal cells from eCG-treated rats 22 hours after hCG administration, which are inherently devoid of CYP17A1/P450c17. Moreover, subsequent to the first ovulation, the 5α-reductase activity is substantially attenuated, while the activity of 20α-HSD is markedly increased.
28
Figure 4 demonstrates that KCZ does not affect the 20α-HSD activity. Residual conversion of 20α-OHP to pregnanediol, via a low reductase and 3α-HSD activities, corroborate that KCZ does not affect those non-cytochrome enzymes.
Lack of KCZ effect on 20α-HSD activity. Post ovulatory granulosa-lutein cells were incubated with [3H]-progesterone (1 μM, 45 minutes) in the absence (NO ADD) or presence of KCZ. Steroid metabolites were analyzed by TLC.
CYP17A1
Incubation of whole ovarian cell preparation with progesterone as CYP17A1 substrate and KCZ inhibited the cytochrome activity. However, the accumulation of pregnanolone corroborated the predominance of the 5α-reductase pathway over that of 17α-hydroxylase (Figs. 5c-i). The absolute KCZ inhibition of androsterone and androstanediol production (Figs. 5f,g) provided further support in favor of this pathway.
Inhibitory effect of KCZ on the androgen pathway. Metabolism of [3H]-progesterone (1 μM, 60 minutes) by whole ovary cell suspension was assessed in the absence (NO ADD) or presence of KCZ and the steroid metabolites were analyzed by TLC.
To directly determine the IC50 value for KCZ inhibition of CYP17A1, [3H]-pregnanolone was purified from ovarian cell cultures and used as substrate. Figure 6A shows that KCZ inhibited the metabolism of pregnanolone with an apparent IC50 of 1.8 μg/mL. However, since it was not clear which of the two inherent CYP17A1 activities was affected by KCZ, we added [3H]-17α-OHP as immediate substrate for the 17,20 lyase activity (Fig. 6B). To prevent loss of the [3H]-17α-OHP toward the 5α-reductase pathway, we added excessive unlabeled androstenedione (50 μM). In the absence of KCZ, as well as in presence of the drug, close to 60% of the [3H]-17α-OHP substrate was converted to androstenedione (Fig. 6B) suggesting no inhibition of 17,20 lyase by KCZ. Collectively, these results suggest that KCZ inhibits only the first of the dual catalytic reactions of CYP17A1.
Effect of KCZ on CYP17A1 activities. (
CYP19A1
KCZ inhibited (IC50 = 0.3 μg/mL) the conversion of testosterone to 17β-estradiol in granulosa cells retrieved from eCG-treated rat ovary (Fig. 7m).
Inhibition of CYP19A1 activity by KCZ. Granulosa cells were prepared as described in Figure 3 and aromatase assay was performed in the presence of KCZ doses.
17HSD1
Estrone was preferred as the substrate for the assessment of the effect of KCZ on 17HSD1 (Materials and Methods section) showing that KCZ has no effect on this enzyme activity (Fig. 8).
Lack of KCZ effect on 17HSD1 activity. Metabolism of [3H]-estrone (1 μM, 90 minutes) by granulosa cells was analyzed in the presence or absence (NO ADD) of KCZ. Steroid metabolites were analyzed by TLC.
Reversibility of KCZ Effect
To examine whether KCZ inhibition is reversible, CYP17A1 was the enzyme of choice because of its fast activity rate among the P450s. To this aim, [3H]-pregnanolone was fed to a whole ovarian cell suspension. Figure 9A shows that within 60 minutes, over 60% of substrate was converted to the enzyme products (Figs. 9e-g). A complete inhibition of the latter was observed in the presence of KCZ (Fig. 9B), which could be fully reversed 30 minutes after the cells were washed free of the drug (Fig. 9B”).
Reversibility of KCZ inhibition of CYP17A1/P450c17. After 60 minutes of KCZ-mediated inhibition of [3H]-pregnanolone metabolism to androgens (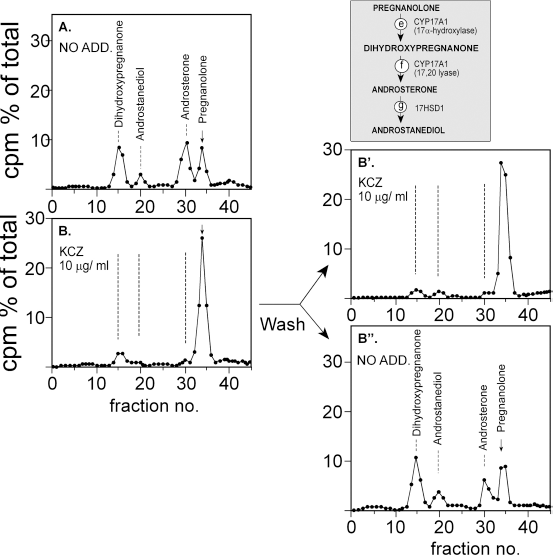
Discussion
To obtain a comprehensive dissection of KCZ effects on the steroid biosynthetic pathways in the ovary, we followed the metabolic fate of radiolabeled steroid precursors in intact cells. This was our method of choice because freshly prepared cells better reflect the physiological characteristics of the ovarian response to KCZ, including the uptake of the drug into the cells, its intracellular metabolism, and the subsequent inhibition of the steroidogenic enzymes. Using this experimental model, the results clearly show that KCZ inhibits dose-dependently and selectively the rat ovarian CYP11A1, 17α-hydroxylase activity of CYP17A1, and CYP19A1. In contrast, the five non-P450 enzymes and the 17,20 lyase activity of CYP17A1 were not inhibited by as much as 10 μg/mL of KCZ.
This is the first report showing a direct inhibitory effect of KCZ on CYP11A1 catalysis in the ovary. The observed IC50 value (0.3 μg/mL; 0.56 μM) for KCZ action on CYP11A1 of rat ovarian cells is in good agreement with previously reported values obtained in isolated cells of rat adrenal (0.3 μg/mL), 29 cell-free preparations of rat adrenal and testis,7,30 and mitochondrial enriched fractions of human placenta (0.3 μM). 31
It is well known that similar to the other steroidogenic P450s, CYP17A1 performs a two-step catalysis consisting of substrate hydroxylation followed by cleavage of a carbon–carbon bond. 32 Clearly, KCZ inhibited the 17α-hydroxylase activity (IC50 ~ 1.8 μg/mL) and thus corroborates earlier findings in rat testicular tissue, 33 Leydig cells preparations from human, stallion, and pig testes, 34 as well as in rat and human testicular homogenates.35,36 Moreover, in the present ovarian cell model KCZ did not inhibit the 17,20 lyase activity of CYP17A1. Thus, the selective KCZ-mediated inhibition of 17α-hydroxylase is valuable in light of the apparent inconsistencies observed in other studies using different models of testicular origin, including minced tissue pieces,33,37 isolated Leydig cells, 34 cell-free assays,31,35,36 or even different use of CYP17A1 substrates. 38
In ovarian models, the effect of KCZ on the dual activities of CYP17A1 was never assessed before in a defined cell model. For example, KCZ inhibition of microsomal 17α-hydroxylase prepared from whole ovary homogenate was reported using very high concentrations (50-5000 μM) 16 previously found toxic in our hands. 8 Others concluded that KCZ inhibits the 17,20 lyase activity of CYP17A1 by incubating pieces of human ovary, 18 or preparing ovarian microsomes from rats orally pretreated with KCZ (4 mg/day). 13
As expected, KCZ also inhibited the activity of the third steroidogenic cytochrome P450, CYP19A1 (IC50 = 0.3 μg/mL; 0.56 μM) in the rat granulosa cells. Thus, except for one study, 16 our results corroborate previously reported findings by others.12–15,17,18,39,40
While reviewing previous studies on the effect of KCZ on the non-P450 steroidogenic enzymes, one can note inconsistent findings depending on the experimental model. Similar to our results, studies using intact cell models observed lack of KCZ effect on 3β-HSD and 17β-HSD7,8,33,36 while the use of cell-free preparations yielded inhibitory effects of the drug at exceedingly high concentrations. 16 Similarly, we could not detect any effect of KCZ on 5α-reductase, 3α-HSD, and 20α-HSD.
Several studies have previously argued for the preferential inhibitory effect of KCZ on the gonadal CYP17A1. For example, KCZ decreased intratesticular testosterone production by inhibiting both 17α-hydroxylase and 17,20 lyase activities, following oral pretreatment with the drug in men with prostate cancer who underwent orchiectomy. 36 Similarly, a relatively stronger inhibitory effect of KCZ was shown for the CYP17A1 activity in Leydig cells from human, stallion, and pig testes. 34 In contrast, this report using a defined cell model did not reveal IC50 differences in KCZ effects on the rat ovarian cytochromes P450s.
In conclusion, the results of the present study support the notion that KCZ is a selective and effective inhibitor of the ovarian P450 enzymes. The comprehensive analysis of the KCZ effect on the ovarian steroidogenic pathways may assist in the development of selective drugs applied in clinical situations requiring acute, yet reversible suppression of ovarian steroidogenesis related to rapidly growing ovarian cysts, induction of ovulation, and ovarian hyperstimulation syndrome.
Author Contributions
MG planned and executed the experiments and was responsible for data interpretation and writing of the manuscript; JO helped in planning, supervised the work, and participated in data analysis and interpretation. All authors reviewed and approved of the final manuscript.
Dsiclosures and Ethics
As a requirement of publication the authors have provided signed confirmation of their compliance with ethical and legal obligations including but not limited to compliance with ICMJE authorship and competing interests guidelines, that the article is neither under consideration for publication nor published elsewhere, of their compliance with legal and ethical guidelines concerning human and animal research participants (if applicable), and that permission has been obtained for reproduction of any copyrighted material. This article was subject to blind, independent, expert peer review. The reviewers reported no competing interests.
