Abstract
Chronic fatigue syndrome (CFS) is characterized by disabling fatigue of unknown etiology. The thalamus is a key subcortical structure in sleep disorders and certain cognitive functions previously shown to be impaired in CFS patients. We investigated the association between subjective sleep quality and thalamic size in CFS. Twelve right-handed CFS patients and 12 age-, gender-, and handedness-matched healthy controls completed the Jenkins Sleep Questionnaire in order to assess subjective sleep problems. Thalamic size was determined by MR-based volumetry. More sleep problems correlated with greater total thalamic volume in patients (rP = 0.62, 95% CI 0.07–0.88, p = 0.032) but not in controls (rP = −0.034, p = 0.30). In post hoc analysis, more sleep problems correlated with right thalamic size in patients (rP = 0.70, 95% CI 0.21–0.91, p = 0.012) but not in controls (rP = −0.080, p = 0.81). Our preliminary results provide a basis for further studies on a possible role of the thalamus in sleep complaints and fatigue of patients with CFS.
Introduction
Chronic Fatigue Syndrome (CFS) is a symptom complex characterized by disabling fatigue of unknown etiology, self-reported impairments in concentration and short-term memory, sleep disturbances, and musculoskeletal pain. People tend to report “fatigue” as a perceived difficulty, or even inability, to initiate and sustain voluntary activities (Chaudhuri and Behan, 2004). This feeling is commonly communicated as a lack of drive or action initiation (van der Linden et al. 2003). Nonetheless, fatigue mainly represents a subjective state of mind. Therefore, it remains difficult to disentangle complaints of tiredness, lack of energy related to depression, and fatigue as a consequence of chronic pain or as a symptom of a physical disease. Particularly, it has to be mentioned that depression co-occurs at a relatively high prevalence with CFS (Fukuda et al. 1994). Accordingly, any definition of fatigue must account for subjective perception and overlaps of the various clinical entities associated with fatigue (Moncrieff and Fletcher, 2007). The definition of CFS requires that the fatigue state must last for 6 or more consecutive months without any active medical condition to explain chronic fatigue (Fukuda et al. 1994).
The subjective feeling of “fatigue” is a common symptom in neurology and occurs in diseases of the central and peripheral nervous system. Fatigue can be distinguished as symptoms of peripheral neuromuscular fatigue on the one hand and symptoms of physical and mental fatigue on the other. Central fatigue may occur because of an integration failure of limbic input and motor functions within the basal ganglia affecting the striatal-thalamic-frontal cortical system (Chaudhuri and Behan, 2004). Whether these pathophysiologic mechanisms contribute to central fatigue in CFS is unknown. However, for a better understanding of fatigue in many diseases and in order to develop new treatment options, it seems important to learn more about these mechanisms.
The thalamus has a strategic position in the central autonomic network, running from the limbic cortical regions to the lower brain stem such as to regulate the body's homeostasis in an integrated fashion. In addition, the thalamus is a key subcortical structure for certain cognitive functions such as attention, speed of information processing, and working memory, all of which may show functional disturbance in CFS patients (Busichio et al. 2004). Besides fatigue, unrefreshing sleep is the most commonly reported case-defining symptom of CFS. A substantial proportion of CFS patients indicate sleep complaints starting after the onset of illness (Reeves et al. 2006). In healthy subjects the thalamus is deeply involved in the pathophysiology of sleep and has been shown to play a primary role in the organization of the wake-sleep rhythm (Reeves et al. 2006; Nofzinger, 2006). Altered thalamic function was also observed in sleep disorders showing both increase and decrease of thalamic activity. In stroke patients, paramedian thalamic neuronal loss is accompanied by deficient arousal during the day, leading to hypersomnia and insufficient spindling and slow-wave sleep production at night (Lugaresi, 1992). SPECT revealed a decreased blood flow in the left thalamus during hypersomnolent periods of recurrent hypersomnia (Nose et al. 2002). An fMRI study revealed increased bilateral thalamic activation in healthy patients performing cognitive tasks after 24h of sleep deprivation (Bassetti et al. 1996). PET showed pronounced thalamic hypometabolism in familial fatal insomnia in which severe neuronal depletion in the mediodorsal (MD) and anteroventral nuclei of the thalamus were demonstrated (Nose et al. 2002). These nuclei constitute the limbic part of the thalamus, interconnecting limbic and paralimbic regions of the cortex and other subcortical structures in the limbic system including the hypothalamus.
To the best of our knowledge investigations of the association between thalamic size and sleep have not been performed in healthy persons. Volume measurement of brain structures has become an increasingly useful tool to better understand neuropsychiatric disorders (Chee and Choo, 2004; Montagna et al. 2003). Variability in the relationship between thalamic volume and sleep function may be based on differences in the number of thalamic neurons, neuronal or axonal density, size of myelin sheaths, glia, and fluid content. Such mechanisms might be more prominent in subjects with relatively more sleep problems. A parallel line of research on neuropsychiatric diseases other than CFS, such as schizophrenia (Geuze et al. 2005; Danos et al. 2003) and parkinsonism (Byne et al. 2002), demonstrated an association between reduction of number of neurons and a decrease in thalamic volume with disturbances in vigilance and attention. In this preliminary study we therefore investigated whether subjective sleep quality could be related to thalamic size in patients suffering from CFS. Supported by the above literature, we assumed that the smaller the thalamic size, the more the vigilance and attention would be perturbed, and, as a consequence, the higher the sleep pressure would be. Therefore, we hypothesized vice versa that the more subjectively impaired the sleep, the greater the thalamic size in CFS patients, with no such relation seen in non-CFS controls.
Methods
The local ethical committee approved the study protocol and all participants gave written informed consent. Patients were recruited from our Psychosomatic Division in the Department of General Internal Medicine, University Hospital Bern, Switzerland. Eligible patients were approached by the physician responsible for the hospital ward and asked whether they would volunteer to participate in a study on brain imaging in CFS. All measurements were made in the outpatient psychosomatic clinic. The study was conducted on 12 right-handed patients (9 women and 3 men) suffering from CFS as diagnosed by previously defined research criteria (Fukuda et al. 1994). Specifically, patients had no somatic condition which could possibly explain fatigue as per a thorough medical history, physical examination, and laboratory work-up following the recommendations by the Centers for Disease Control and Prevention (Danos, 2004; Henderson et al. 2000). Four patients had received psychotropic medication within the last 4 months; one had had paroxetine and methylphenidate, one amitriptyline, one doxepine, and one continuous release morphine, respectively.
Most psychiatric disorders are not exclusion criteria for a diagnosis of CFS (Danos, 2004; Henderson et al. 2000). Therefore, only the following life-time and current psychiatric disorders which are specified exclusion criteria for CFS were identified in a clinical interview performed by an experienced physician: bipolar affective disorder, severe depressive disorder with melancholic features, any psychotic disorder, any eating disorder, and alcohol or illicit drug abuse. Specifically, patients were asked whether a physician (incl. a psychiatrist) had ever mentioned their having any of these disorders.
The sample of healthy controls was recruited by word-of-mouth, and comprised mainly employees of the university hospital. The 12 controls who volunteered to participate were all carefully case-matched in terms of handedness, sex and age (± 1 year) with CFS patients. None of the controls took any psychopharmacological drug. Moreover, controls had been required to indicate no significant mood disturbance and a negative history for any life-time and current psychiatric disorders as verified by an experienced psychiatrist in a clinical interview.
Clinical Measurements
CFS Diagnosis and Severity
The symptomatology of CFS was assessed using the semi-structured clinical interview asking for duration and number of symptoms as defined by the International CFS Study Group (Fukuda et al. 1994). The average intensity of the nine CFS defining symptoms was interviewer-rated on a Likert scale (0–4) to yield a maximum CFS severity score of 36 points. At the time of the interview all patients suffered from CFS for longer than 6 months as per definition. Patients were specifically asked when they first perceived a symptom they retrospectively would relate to CFS. This provided a proxy measure of duration the condition had possibly last.
Sleep Quality
Subjective sleep quality of patients and controls was assessed by the Jenkins Sleep Questionnaire (JSQ) (Jenkins et al. 1988). The JSQ has been designed to track common sleep problems in clinical populations and has been successfully applied in a German speaking population. The JSQ comprises 4 items asking into a) difficulty in initiating sleep, b) awakening during the night, c) awakening during sleep with difficulty maintaining sleep, and d) awakening exhausted in the morning despite having slept as usual. All items are rated on a Likert scale (0–5) yielding a maximum score of 20 points. Higher scores indicate more sleep problems and lower sleep quality, respectively.
Depression
Symptoms of depression were assessed using the German version (Herrmann et al. 1995) of the 7-item depression subscale of the Hospital Anxiety and Depression Scale (HADS) (Kudielka et al. 2004) rendering a severity score between 0 and 21. Depressive symptom levels do not allow for diagnosing a clinical depression, but are suggestive of mild depression (8–10 points), moderate depression (11–14 points), and severe depression (≥ 15 points), respectively. In studies comparing the HADS depression subscale score with gold standard clinical assessment of depression in medically ill patients, sensitivity estimates ranged from 56% to 100% and specificity estimates were between 73% and 94% (Zigmond and Snaith, 1983; Goldberg, 1985). In patients with CFS, a cutoff >8 points on the HADS depression subscale has been proved valid and efficient for the screening of a depressive disorder as diagnosed in a structured clinical interview (Silverstone et al. 1994; Henderson, 2005). Because controls had been required to indicate no significant mood disturbance they did not complete the depression scale.
Importantly, the questionnaires applied to measure sleep quality (Jenkins et al. 1988) and depressive symptoms (Zigmond and Snaith, 1983) have been widely used. In spite of their development in the 1980s, they are seen as state-of-the art to assess the domains of subjective sleep complaints (Steptoe et al. 2008) and depressed mood (Henderson et al. 2005), respectively.
MRI Recording and Analysis
All MRI examinations were performed with a Sonata 1.5T scanner (Siemens Erlangen, Germany) with a 40 mT/m (200 mT/m-ms) gradient system and a CP standard head coil, using the scanner software Syngo MR 2002B (VA21B). For anatomical imaging, a T1-weighted, sagittal oriented 3D-MPRAGE sequence (TR/TE/TI 2000/3.93/590 ms, matrix 256 × 256, FOV 256 × 256 mm, flip angle 15°, slab 160 mm) with a 1 mm3 isovoxel resolution was obtained. Volumetric evaluations were performed by manually tracing the thalamus in three planes on high-resolution T1-weighted scans using the commercially available BrainVoyager software (Version 4.9.6). The 3-dimensional on-line ROI tracing simultaneously in 3 planes allows the exact delineation of the thalamic anatomical borders, which were defined in accordance with the T1-signal changes at the grey/white matter interface at the boundaries of the thalamus. The anterior boundary of the thalamus was defined as the posterior point of the interventricular foramen and the posterior boundary coincided with the section in which pulvinar thalami were seen. The lateral boundary of the thalamus was defined at the plane, where the posterior limb of the internal capsule was seen. The superior and inferior boundaries of thalamus was defined at the level of body of the fornix and hypothalamic sulcus respectively. The manual ROI tracing was performed by a rater (TL) with experience in neuroanatomy and blind to diagnosis. A spheric water filled dummy model was used for calibration of the realistic thalamic volumes.
Statistical Analyses
Statistical analyses were performed using SPSS 12.0 software package (SPSS Inc. Chicago, IL). The level of significance was set at p < 0.05 and all tests were two-tailed. All data showed a normal distribution as per the Kolmogorov-Smirnov test. Pearson's correlation test (rP) was used to investigate significant associations between two continuous variables. Because this was a case-control design, conditional logistic regression was performed to test whether continuous measures (e.g. sleep score, thalamic volume) predict CFS status. Our small sample size precluded, however, reliable computation of logistic regression analysis with the use of covariates and their interaction terms (Greenland et al. 2000). Alternatively, we tested whether there would be a significant bivariate correlation between sleep problem score and the total thalamic volume in CFS patients and controls. Post hoc comparisons applied the conservative Bonferroni correction for multiple testings. That is, in case of a significant correlation between sleep problems and total thalamic volume in CFS patients, a p-value of < 0.025 would be required to state statistical significance of a correlation between the sleep problem score and thalamic size of either side.
Results
Table (1) demonstrates that matching in terms of age, gender, and handedness between patients and controls was achieved. Thalamic size of either hemisphere and aggregate volume of thalamic nuclei did not significantly predict CFS status.
Subjects' characteristics.
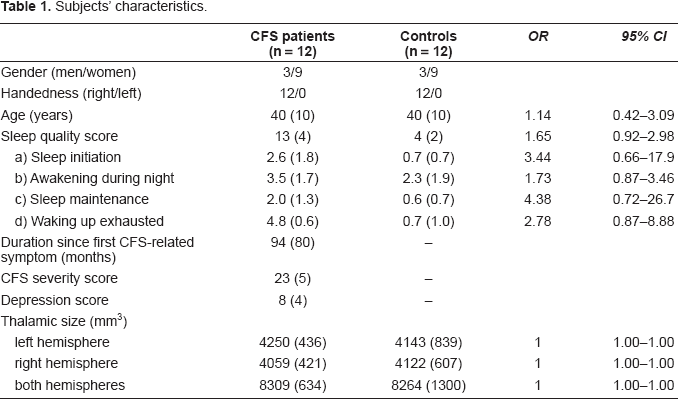
In CFS patients, there was a positive correlation between the sleep score and total thalamic volume (i.e. volumes of the right and left thalamus combined) (rP = 0.62, 95% CI 0.07–0.88, p = 0.032; Fig. 1a), which was not seen in non-CFS controls (rP = −0.34, p = 0.30). Even though the total sleep score (p = 0.84) and total thalamic volume (p = 0.95) showed a normal distribution (Kolmogorov-Smirnov test), the orientation of the regression line between the sleep problem score and total thalamic volume in patients with CFS was suggested to be influenced by one subject with comparably good sleep (i.e. a score of 5 on the JSQ) and small thalamic volume (cf Fig. 1a). Therefore, we analyzed our data computing standardized residuals and Cook's distance D to detect potential outliers and influential cases in our regression model. We found that all values were within acceptable limits (statistical analyses not shown in detail).
Sleep quality and total thalamic size in CFS patients.
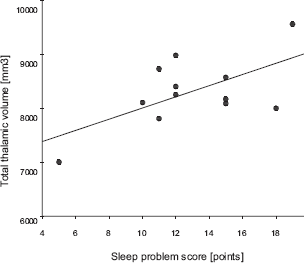
In a post hoc analysis we explored which thalamic size would possibly make a greater contribution to the relationship between more sleep problems and a larger total thalamic volume. There was a significant correlation between the sleep score and right thalamic size in patients (rP = 0.70, 95% CI 0.21–0.91, p = 0.012; Fig. 1b) but not in controls (rP = −0.08, p = 0.81). In contrast, the correlation coefficients between the sleep score and left thalamic size was not significant in patients (rP = 0.23, p = 0.48) and in controls (rP = −0.41, p = 0.19). In CFS patients the number of cases required to discover a statistically significant correlation between the sleep score and left thalamic size (p = 0.05, two-sided) with a power of 80% would have been n = 116. In contrast, a sample size of n = 13 would have been required to detect such a significant correlation between the sleep score and right thalamic size suggesting our sample size of n = 12 CFS patients sufficed to deem this relationship “truly” significant.
Total thalamic volume and thalamic size of either side did not significantly correlate with CFS severity and duration since the first CFS-related symptom occurred.
In addition, we found six patients who scored above the cut-off level suggestive of clinical depression on the HADS depression subscale. More precisely, one patient was severely depressed, three patients were moderately depressed, and two patients were mildly depressed. However, compared to patients scoring in the normal range of depressive symptom levels, depressed patients showed no significant difference in right thalamic size, left thalamic size or total volume of thalamic nuclei, respectively. Consistently, depressive symptom scores were not significantly associated with any of the thalamic volume measures across all patients. A stratified analysis in terms of gender revealed no significant correlation between sleep problem scores and any thalamic volume measure in patients and controls.
Discussion
We used MR-based volumetry to investigate the relationship between thalamic size and sleep quality in 12 patients with CFS and 12 age-, and gender-matched healthy, non-CFS controls. Clearly, our study must be viewed as preliminary and as such may primarily serve as a basis for future, more extensive investigations on possibly perturbed neurofunctional networks in CFS patients. We therefore first acknowledge three limitations of our study which imply that following interpretation and discussion of findings must apply caution pending replication in larger samples of main findings with more sophisticated statistical analyses.
First, our sample size did not allow us to apply complicated statistical regression modelling because this can be unstable when controlling for too many covariates (Greenland et al. 2000; Papoulis, 1990). As a consequence we could not control our analyses for depressed mood and CFS duration. Although depression and the time since the first CFS-related symptoms occurred were not individually related to thalamic size, these variables could, when aggregated and applied to a larger sample, account for some of the relationship between sleep quality and thalamic size observed in CFS patients. Some symptom domains of depression and CFS may overlap and relate to cognitive, limbic, and autonomic functions in all of which the thalamus plays a role. However, the lack of association between depressive symptoms and thalamic volumes in our patients is in accordance with some of previous post-mortem studies showing that depressed patients (without CFS) and non-depressed subjects had similar volumes of subcortical nuclei and of the thalamus in particular (Vasic et al. 2008; Bielau et al. 2005; Caetano et al. 2001). In contrast, a recent study found that patients with a major depressive disorder, and thus considerably higher levels of depressed mood than observed in our CFS patients, had enlarged total thalamus volume. Moreover, neuroendocrine function is altered with longer duration of CFS (Gaab et al. 2004) such that it could be theorized that alteration in cortical structures might also occur with longer duration of CFS symptoms. However, there was great variability in the duration since first CFS-related symptoms occurred making it difficult to detect such an association.
Second, our study does not allow the distinction of whether thalamic size correlated with sleep problems per se, total sleep time, or some other complaints that are unrelated to sleep problems but similarly prevalent in CFS patients (e.g. anxiety). More elaborated study designs, for instance, to compare patient groups with sleep complaints with relatively longer and shorter total sleep times or to assess objective measures of sleep quality (e.g. by actigraphy) are needed to resolve this. Nevertheless, as yet, there is not much evidence for a major role of abnormalities in sleep architecture and objective measures of sleepiness in CFS (Ball et al. 2004; Watson et al. 2004). Third, our psychiatric and psychometric assessment was limited by the fact that we did not assess life-time and current psychiatric disorders in a structured clinical interview. In line with other studies (Wessely et al. 1999), however, half of our CFS patients had depressive symptom scores above a cut-off level previously shown to perform well in identifying CFS patients with clinically diagnosed depression (Henderson and Tannock, 2005).
Owing to the preliminary nature of our study, a series of further limitations also deserve consideration. Fellow researchers may want to address these drawbacks in future study designs. Although the orientation of the regression line between the sleep problem score and total thalamic volume in patients with CFS was not evidently influenced by outliers in statistical terms, more data is needed to confirm the validity of the findings. Notably, our data do not preclude the possibility that there was also a clinically relevant direct association between subjective sleep problems and left thalamic size since power analysis showed that the sample was clearly too low to detect a statistically significant relationship in our patients. The relationship between sleep and thalamic size in CFS patients is difficult to interpret as there was no difference in thalamic size between patients and controls which observation might also reflect a power issue. Moreover, confidence intervals are rather wide in relation to the small sample size and therefore difficult to interpret. The questionnaire to assess sleep problems is widely applied; however, to the best of our knowledge, its validity and reliability has not been tested in patients with CFS. Our control subjects were mainly recruited among hospital employees, which are a selected population, thereby potentially limiting the generalizability of our data. We did not relate thalamic volume to total brain volume. However, gender may be viewed to some extent as a proxy measure of total brain volume that was indirectly considered by the nature of our case-control design.
We found a positive correlation between more sleep complaints and total and right thalamic size in CFS patients but not in controls. The observation suggests that the more sleep disturbance CFS patients perceived, the greater was their right thalamic size. We may interpret that the size of the thalamus could be associated with the pathology of sleep processing in CFS. In contrast, the CFS symptom score was not associated with thalamic size. However, besides the core symptom fatigue, the CFS symptom scale considers eight additional symptoms of which only one relates to subjectively perturbed sleep (i.e. “unrefreshing sleep”). Therefore, we may speculate that subjectively perturbed sleep is perhaps the core dimension of CFS relating to thalamic size, whereas thalamus volume might play less of a role in the relationship with the other CFS symptom dimensions. Alternatively, the lack of a statistical association between the CFS severity score and thalamic size might also indicate that other central structures than the thalamus underlie total CFS symptom severity.
Consistent with previous findings, we found no hemispheric asymmetry in the thalamic size of either side in healthy subjects (Szabo, 2003). In this first volumetry study of the thalamus in CFS, we also found that right and left thalamic size were similar in CFS patients and that thalamic size was not different in patients compared to controls. Besides a power issue as mentioned above, we offer three possible explanations for this observation. First, we hypothesize that there could be antecedent microstructural thalamic differences in subjects with sleep disturbances related to CFS which are not present in healthy people. Second, even though not detected by our macroscopic volumetric issue there could be some differences on a microstructural level influencing sleep pattern (Begré et al. 2006; Yamada et al. 2006). Further structural evaluation by diffusion tensor imaging could enlighten this suggestion. Third, augmented thalamic perfusion was previously shown in patients with CFS compared to healthy matched controls (Tomoda et al. 2000; MacHale et al. 2000). Similarly, functional MRI in fatigued people with multiple sclerosis showed an inverse correlation between fatigue severity and activation of ipsilateral thalamus during simple motor tasks (Filippi et al. 2002). We may thus hypothesize that, as a result of chronic sleep impairment, a compensatory increase of thalamic perfusion could have led to thalamic hypertrophy in our CFS patients.
At least 90% of thalamic synapses arise from brain regions making up the many non-primary afferent thalamic inputs implicated in arousal and attention (Sherman, 2005) suggesting the thalamus could be a major region in the processing of fatigue. For instance neurodegeneration in thalamic gray matter, which can also be found in neuropsychiatric diseases other than CFS, might contribute to the genesis of “fatigue”. Neuropathological findings in patients with fatal insomnia (Nose et al. 2002) and Creutzfeldt-Jakob disease (Taratuto et al. 2002) showed thalamic neuronal loss, spongiform changes and prominent gliosis, and altered thalamic function in sleep regulation, all of which can also change thalamic size. Aside from any focal lesion or inflammation, MRI, MRS and postmortem histopathology studies in multiple sclerosis revealed reduced neuronal thalamic density by N-acetylaspartate (NAA) concentration and reduced thalamic volume (Cifelli et al. 2002; Wylezinska al. 2003). In poliomyelitis, postmortem histopathology demonstrated consistent presence of poliovirus lesions also in thalamic nuclei (Bruno et al. 1998). Given thalamic size in both hemispheres did not differ between our CFS patients and controls, one preliminary interpretation could be that neuronal loss had not occurred. Four of our patients had been on psychotropic medication (i.e. paroxetine and methylphenidate, amitriptyline, doxepine, and morphine within the last 4 months, all of which could possibly affect sleep subjectively and objectively. An influence of antidepressants (Mayers and Baldwin, 2005), methylphenidate (Banerjee et al. 2004) and morphine (Villablanca, 2004) on sleeping patterns and arousal is well documented. However, retrospectively, our patients reported no definite effect on sleep quality between before and after they were prescribed these medications. We therefore feel confident that psychotropics did not inflate a spurious result in terms of the relationship between perceived sleep problems and right thalamic size in our CFS patients.
Moreover, a possible influence of psychotropic medication on thalamic volume was documented solely for paroxetine in obsessive-compulsive patients who experienced reduction in symptom severity after psychotropic treatment (Rosenberg et al. 2000; Gilbert et al. 2000), however, none of our patients reported obsessive-compulsive symptoms. Also, metabolic thalamic function was normalized with paroxetine treatment in a PET-study on patients with major depression. A SPECT study showed deactivation in the right thalamus with citalopram (Carey et al. 2004), but it is unknown whether paroxetine could have a similar effect in our CFS patients. Increased regional cerebral blood flow in the right thalamus after methylphenidate prescription was shown using PET (Schweitzer et al. 2004). Altogether, we acknowledge that our sample size did not allow us to perform meaningful statistics on unmedicated patients alone.
Although our results are preliminary they may contribute to the growing body of research suggesting that the thalamus does not only accumulate sensory information before sending it to the cortex, but rather plays a major role as a work station dynamically regulating information processing of a complicated arrangement of neuronal connections organized in feedforward, feedback, and loop circuits (Basso et al. 2005). Therefore, the thalamus could critically be involved in explaining the pathogenesis of the heterogenous symptom complex of CFS.
Conclusions
Using MR-based volumetry, we found a positive association between subjective sleep problems and thalamic volume in patients with CFS suggesting an association between impaired sleep quality and thalamic functioning in CFS. This notion corresponds to the literature on the importance of thalamus in the regulation of sleep and to the distinctive features of sleep problems frequently reported by patients with CFS. Because this was not a mechanistic study, more elaborated study designs are needed to investigate to what extent the thalamus could be part of the neurofunctional network pertinent to CFS. Our findings must be understood as preliminary and more research in larger samples is clearly required to increase knowledge of the interplay of brain structures, their functioning, sleep, and the subjective state of fatigue in CFS patients. Such studies might advance our currently limited understanding of central processing of fatigue not only in CFS but also in a variety of other neuropsychiatric diseases and disorders in order to develop target interventions in the future for the often disabled patient suffering chronic fatigue.
