Abstract
AIM
To study the pattern of various adverse drug reactions (ADRs) occurring in children attending the Department of Pediatrics, SMGS Hospital, Jammu over 1 year.
Subjects and Methods
This was a prospective study, with study population of patients attending Department of Pediatrics over a period of 1 year. A structured format was used to enroll the participants. A pilot study was conducted to test the suitability of the format and feasibility of the study. The study was carried out to review various pattern of ADRs by using the Naranjo probability scale, and severity was assessed by using the Hartwig severity scale. ADRs were classified according to the classification used by the Adverse Drug Reaction Monitoring Center, Central Drugs Standard Control Organization, New Delhi, India.
Results
In the present study, 104 patients were found to have developed acute drug reactions. Among these, 83.6% were type B, 14.42% type A, and 1.92% were type U. Furthermore, 25.96% ADRs were due to anticonvulsants, followed by antibiotics (22.11%), antipyretics (11.53%), vaccination (8.65%), steroids (6.73%), decongestants (5.67%), snake antivenom and antiemetics (3.84%), and fluids, insulin, and antacids (1.92%). The patients’ dermatological system was involved in 67.30%, followed by the central nervous system (CNS) in 11.53% patients. Renal system was involved in 6.73% patients. Cardiac, musculoskeletal, metabolic, and other systems were involved in 4.80%, 3.84%, 2.88%, and 0.96%, respectively. According to the Hartwig severity scale of ADRs, 64.4% patients had moderate ADRs, 29.8% patients had severe ADRs, and 5.76% had mild ADRs. In the present study, 64.4% patients expressed moderate severity, whereas 29.8% expressed high severity and 5.76% expressed mild ADRs.
Conclusion
ADRs were seen in 71% of the patients between 1 and 5 years of age, 26% in the age group of 5–10 years, and 3% were more than 10 years old. Anticonvulsants (25.96%) and antibiotics (22.11%) were responsible for majority of ADRs. Rash (55.76%) was the most common presentation of ADR. Owing to the high number of ADRs, the present study points to the need for rigid adverse drug monitoring among pediatric patients to ensure the safety of drug therapy.
Introduction
“Primum Non Nocere” (First, do no harm.) Hippocrates (460–370 BC)
The above principle is enshrined in the Hippocratic Oath. Essentially, children cannot be regarded as small adults; they are afflicted with many conditions and disease processes that are different from those in adults, particularly neonates and infants. Drugs may behave differently in children (different pharmacokinetics) compared to adults and also may cause different effects (different pharmacodynamics) in children.1–3 Pediatric patients require specific formulations of drugs and may experience specific adverse effects not suffered by adults; thus pediatric patients are a susceptible population to adverse drug reactions (ADRs). On one hand, insufficient information on the efficacy of drugs can lead to suboptimal treatment. On the other, without safety data gained from clinical trials, children may be exposed to serious unintended harm arising from drug toxicity. 4 ADRs are one of the major causes of iatrogenic disease, and they are as old as medicine itself. Historically, there have been many examples of patients causing harm to themselves through the use of prescribed medicines: for example, 107 deaths were directly associated with diethylene glycol poisoning after ingesting the elixir of sulfanilamide. 5 ADRs not only may result in hospital admission or prolonged hospitalization but also may lead to permanent disability or even death. Lazarou et al. 6 estimated that ADRs were the cause of 5% hospital admissions and that approximately 11% of hospital inpatients experience them. In the United Kingdom, a large prospective study found that 6.3% of admissions were related to ADRs and that the admissions accounted for 4% of hospital bed capacity, although only adults above 16 years of age were included in the study. 7 The concept of ADR reporting is still new. Hardly any detailed ADR surveys have been done in India. In one of study, 3.7% of hospital patients experienced ADR, 0.7% of the admission were due to ADRs, and 1.8% had fatal ADRs. 8 Another study done in India, on the pattern of ADR, revealed incidence of ADR as 0.15%. 9 Yet another study done during 2009 found 30 ADRs over 6 months, and 60% of ADRs were in children below 1 year of age. 10 ADRs due to antibiotics are the most common in this country because these are the most commonly used drugs in therapy. Considering the impact of ADRs on the morbidity, mortality, economic burden, and the potential vulnerability of children, the present study was conducted over a period of 1 year among the children attending our hospital. The present study may help in rationalizing drug use in pediatric practice in our region. Furthermore, it will provide a rough estimate of the ADR burden and, if communicated effectively, this information will allow evidence-based use of medicines and thus have the potential of preventing many adverse reactions.
Subjects and Methods
The present study was a prospective observational study conducted in Department of Pediatrics, SMGS hospital, Jammu, India, over a period of 1 year from November 1, 2011 to October 31, 2012. The institution's Human Ethics Committee's approval for waiver of consent was obtained prior to initiation of the study. The study was conducted in accordance with the Declaration of Helsinki. Patients in the wards and outpatient department of pediatrics during the study period were monitored actively for occurrences of any ADRs till their discharge from the hospital. All patients in the age group from 1 month to 19 years and of either sex were included in the study. Children with a history of poisoning due to any drug were excluded from the study. Parents of all the eligible candidates were explained the purpose of the study for their participation. For patients visiting the outpatient department, complete address and contact telephone numbers were taken. Patients and parents were interviewed in detail as per the format (see appendix 1). All patients were subjected to detailed history and clinical examination. A detailed history of the symptoms, type of drug, date of starting the drug, duration of drug, dose of drug, age, sex, severity of reactions, and any previous history of drug reaction were noted. Detailed clinical examination including the central nervous system (CNS) and dermatological, cardiovascular, respiratory, and gastrointestinal systems was performed. Follow-up of the inpatients for outcome was made till their discharge in the case of inpatients, and follow-up of the outpatients was monitored telephonically and through visits. Data as per the format was collected from all the patients and carefully analyzed. An ADR is defined by the World Health Organization as “a noxious and unintended response to a drug that occurs at a dose normally used in man for prophylaxis, diagnosis, or therapy”. 11 The classical pharmacological classification of ADRs separates them into two major subtypes: type a (augmented) and type b (bizarre). 12 This classification was further extended to include other subtypes in order to facilitate the inclusion of reactions that did not find their place in the subtypes a or b.13–15 Onset of the event is defined as acute (within 60 minutes), sub-acute (1 to 24 hours), and latent (>2 days). 16 Severity of the ADRs according to the Hartwig severity scale 17 are mild, moderate, and severe. The causal relationship between the ADR and the suspected drug therapy was assessed by using the Naranjo probability scale, 18 and severity was assessed by using the Hartwig severity scale appendix 2. 19 ADR was classified according to classification used by the ADR Monitoring Center, Central Drugs Standard Control Organization, New Delhi, India. But due to the short duration of study and short follow-up, our study patients were classified as A, B, or U. The presence or occurrence of ADR was confirmed by two investigators specially chosen for this purpose. On confirmation by both investigators, the case was taken up for the study.
Results
In the present study, 104 patients were found to have developed ADRs out of a total of 28,864 patients (0.3%). Among them, 66.34% were male and 33.65% were female.
Causality
In all, 83.6% of ADRs were type B, 14.42% were type A, and 1.92% were type U. Anticonvulsants were the most frequent cause (25.96%), followed by antibiotics (22.11%); antipyretics (11.53%); vaccination (8.65%), steroids (6.73%); decongestants (5.67%); snake anti-venom and antiemetic together (3.84%); fluids, insulin, antacid, and anti-motility drugs (1.92%); and bronchodilators, multivitamins, and osmotic diuretics (0.96% each).
Clinical Manifestation
Dermatological system was the most commonly affected (67.30%), followed by CNS (11.53%) and the renal system (6.73%). Cardiovascular, musculoskeletal, metabolic, and other systems were involved in 4.80%, 3.84%, 2.88%, and 0.96%, respectively.
Severity
According to the Hartwig severity scale of ADRs, 64.4% patients had moderate ADRs, 29.8% had severe ADRs, and 5.76% had mild ADRs. In the present study, 64.4% patients expressed moderate severity, whereas 29.8% expressed high severity and 5.76% expressed mild ADRs. Out of the total number of patients, 65.38% had no risk factors, while 15.38%, 5.7%, and 13.46% patients had inappropriate monitoring, inappropriate use, and inappropriate prescription, respectively.

Pie chart showing age-wise distribution of patients.
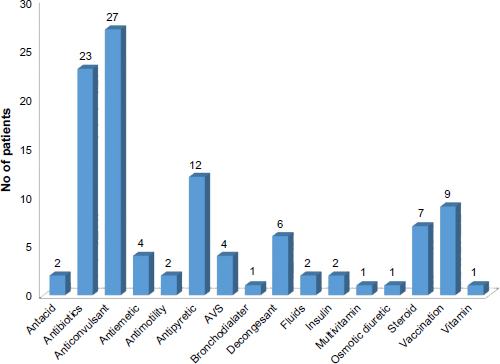
Bar chart showing the distribution of ADRs according to class of drug involved.
Discussion
ADRs are an important public health problem especially in the pediatric population. Despite the efforts being made to reduce the incidence of medication-related adverse events, the morbidity and mortality due to drug-induced reactions continue to be unacceptably high. ADRs are one of the major causes of iatrogenic diseases. They are often not recognized and, even if they are recognized, they are underreported. Many health professionals are unaware of their importance and possible consequences.
Incidence
This study illustrates the magnitude of the problem of ADRs in selected pediatric population with the finding that ADRs were reported in 104 patients (0.3%). It included 68.26% patients admitted for ADRs and 31.73% patients who developed ADRs during the hospital stay. Symth et al. 20 in their systemic review reported the incidence of ADRs ranging between 0.4% and 10.3% of all children and that from 0.6% to 16.8% of all children developed ADRs during hospital stay. The higher incidence of ADRS in our admitted patients can be explained on the basis of the active search for such reactions in our population and vigorous monitoring.
Gender Predisposition
In the present study, 66.34% male and 33.65% female children had ADRs. These are comparable to the percentages reported by Oshikoya et al. 21 who reported 59% males and 31% females had ADRs and by Rohit et al. 22 who reported 56.7% males and 43.3% females with ADRs. This pattern of gender predisposition of ADRS is comparable with that in the present study.
Rural/Urban
In the present study, 57% patients belonged to urban areas, while 43% belonged to rural areas. The higher incidence in urban population may be explained as due to the easy availability of drugs in urban areas, easy accessibility of heath institutions, and the life style of the patients living in urban areas.
Age Group
In the present study, 71% patients were below 5 years, 26% were in 5–10 year age group, and 3% were more than 10 years old. Priyadharsini et al. 12 reported that 60% patients were below 1 year, 20% were between 1 and 3 years, and 20% were between 4 and 6 years of age (Figure 1).
Causative Agents
In the present study, anticonvulsants were responsible for 25.96% of the ADRs, whereas antibiotics contributed 22.11%. These observations are consistent with a study done by Easton et al. 23 , who reported that anticonvulsants were responsible for 30% of ADRs, whereas Priyadharsini et al. 12 reported antibiotics as the most common drug implicated in 67% patients (Fig. 2)
Organ of Affection
In the present study, the dermatological system was found to be involved in 67.30% patients. Similar observations were made by Srivastava et al. 24 and Priyadharsini et al. 12 , who reported rashes as the presentation of ADR in 37.24% and 37% patients, respectively. The majority (83.6%) of ADRs in the present study were constituted by type b reaction. This shows that most of the ADRs were inevitable and unavoidable. Uday Kumar et al. 25 observed that 68% ADRs were type b, which is in concurrence with the present study. In contrast, a meta-analysis 6 showed that 76.2% ADRs were of type a. This discrepancy may be due to the fact that the large magnitude of ADRs was due to antibiotics and anticonvulsants, which are indispensable for patient management. Out of all the patients, 90% ADRs were probable while 10% were possible in the present study. Priyadharsini et al. 12 reported that 80% ADRs were probable, 17% were possible, and 3% were definite. Arulmani et al. 26 reported that 62.2% had probable association. The observations made in the present study were consistent with those of the above studies (Fig. 3)

Pie chart showing the distribution of patients according to the system involved.
Severity
In the present study, 64% ADRs were moderate, 30% were severe, and 6% were mild. Priyadharsini et al. 12 observed that 77% patients had moderate ADRs and 23% had severe ADRs. The observations made in the present study are consistent with the above study (Fig. 4)

Bar chart showing the distribution of patients according to type of ADR.
Conclusion
In this study, ADRs occurred more among infants, and anticonvulsants were more commonly implicated, followed by antibiotics. Most of the reactions were of moderate severity. This indicates the need for rigid ADR monitoring among pediatric patients to ensure the safety of drug therapy. Various pharmacovigilance awareness programs should be conducted to increase the spontaneous reporting of ADRs.
Author Contributions
Conceived and designed the experiments: KKD. Analyzed the data: AP. Wrote the first draft of the manuscript: KKD, RB. Contributed to the writing of the manuscript: AP, GS. Agree with manuscript results and conclusions: KKD, AP, GS, RB. Jointly developed the structure and arguments for the paper: AP, RB, GS. Made critical revisions and approved final version: AP, GS. All authors reviewed and approved of the final manuscript.
Footnotes
Appendix 1
Questions and responses regarding ADRs reported in this study.

| QUESTION | YES | NO | DON'T KNOW |
|---|---|---|---|
| Are there previous conclusion reports on this reaction? | +1 | 0 | 0 |
| Did the adverse event appear after the suspect drug was administered? | +2 | -1 | 0 |
| Did the ADR improve when the drug was discontinued or a specific antagonist was administered? | +1 | 0 | 0 |
| Did the ADR reappear when drug was readministered? | +2 | -1 | 0 |
| Are there alternate causes [other than the drug] that could solely have caused the reaction? | -1 | +2 | 0 |
| Did the reaction reappear when a placebo was given? | -1 | +1 | 0 |
| Was the drug detected in the blood [or other fluids] in a concentration known to be toxic? | +1 | 0 | 0 |
| Was the reaction more severe when the dose was increased or less severe when the dose was decreased? | +1 | 0 | 0 |
| Did the patient have a similar reaction to the same or similar drugs in any previous exposure? | +1 | 0 | 0 |
| Was the adverse event confirmed by objective evidence? | +1 | 0 | 0 |
