Abstract
Recurrence of testicular tumors in young males generally prompts rapid investigation and treatment. We report 3 patients with past histories of treated testicular cancer referred with radiographic evidence suggestive of intrathoracic metastases. In each case chest imaging demonstrated mediastinal/hilar lymphadenopathy. In one case pulmonary nodules were also identified. In all three patients further work-up revealed non-caseating granulomas consistent with sarcoidosis. All patients have since been followed and remain free of testicular cancer. No patient has required therapy for sarcoidosis. Although rare, new intra-thoracic lymphadenopathy in previously treated testicular cancer patients can represent sarcoidosis. Tissue diagnosis is essential when the clinical picture remains unclear.
Introduction
Testicular cancer is the most common form of cancer in young men (Sternberg, 1993). There will be an estimated 830 new cases of testicular cancer and 30 deaths from testicular cancer in Canada during 2007 (Canadian Cancer Society, 2007). Because treated testicular cancer patients are at risk for disease recurrence, frequent radiographic evaluation and follow-up are essential. Recurrent testicular cancer frequently presents with enlarged lymph nodes although not all enlarged nodes represent recurrent cancer. We present three cases of patients that were referred with presumed recurrent testicular cancer who, after diagnostic work-up, were found to have sarcoidosis.
Case 1
A 37 year old male developed stage I testicular cancer ten years earlier. At that time, pre-orchiectomy blood work had shown: alpha-fetoprotein (α-FP) = 335 μg/L, and beta-human chorionic gonadotropin (β-HCG) =11 IU/L. Pathology, after orchiectomy, revealed embryonal carcinoma, seminoma and teratoma. Scattered non-caseating granulomas were noted in the spermatic cord. A staging retroperitoneal lymphadenectomy was normal. His post-operative tumor markers normalized. He was closely monitored. Eight months after his original diagnosis he developed recurrent disease. CT scans discovered new retroperitoneal and left hilar lymphadenopathy. Blood work showed: lactate dehydrogenase (LDH) = 1.66 × ULN, α-FP = 740 μg /L, β-HCG = 290 IU/L. He received four cycles of bleomycin, etoposide, cisplatin (BEP). His tumor markers and staging investigations normalized. He was subsequently discharged from medical oncology and received yearly follow-up CT scans through his family doctor. A routine follow-up chest CT scan discovered extensive mediastinal and hilar lymphadenopathy. The largest nodes were 1.5 cm in diameter (Fig. 1). The patient was referred back to medical oncology with a presumed second recurrence.
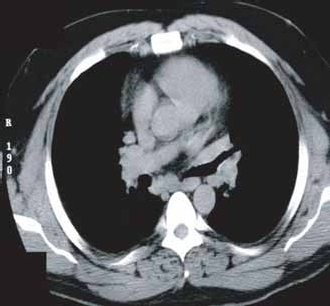
37 year old male with new mediastinal and hilar lymphadenopathy.
Case 2
A 45 year old male developed stage I testicular cancer 10 years earlier. Pathologic evaluation, after orchiectomy, revealed embryonal carcinoma with foci of yolk sac tumor, immature teratoma and seminoma. Pre-orchiectomy tumor markers were not available. Post-orchiectomy tumor markers were normal. A staging retroperitoneal lymph node dissection was unremarkable. The patient was closely followed and subsequently discharged from medical oncology. Prior to his presentation he developed dyspnea on exertion. A CT scan showed new hilar/mediastinal lymphadenopathy (Fig. 2) and pulmonary infiltrates. The patient was referred back to medical oncology with presumed recurrent testicular cancer.
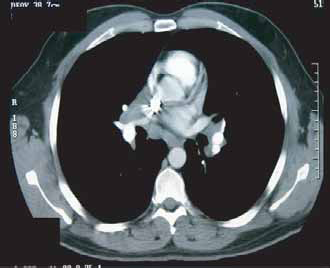
45 year old male with new mediastinal and hilar lymphadenopathy.
Case 3
A 29 year old male developed stage I testicular cancer 13 months earlier. Pathology, after orchiectomy, revealed embryonal carcinoma and teratoma. Pre-operative tumor markers were not available. Post-orchiectomy tumor markers and staging investigations were normal. The patient was subsequently followed. A routine surveillance CXR identified marked bilateral hilar lymphadenopathy. A follow-up CT scan showed symmetric hilar/mediastinal lymphadenopathy (Fig. 3) and a right upper lobe 1.8 × 1 cm lung nodule suggestive of testicular cancer recurrence.

29 year old male with new mediastinal and hilar lymphadenopathy.
At the time of presentation, for presumed testicular cancer recurrence, blood work in all three patients revealed normal β-HCG, and α-FP levels. Two of the three patients had mildly elevated LDH values (1.1–1.2 × ULN). All patients underwent bronchoscopy and mediastinoscopy. In all cases pathology showed non-caseating granulomas on hematoxylin and eosin (H+E) staining as demonstrated by case 1 (Fig. 4). No carcinoma cells were identified. Cultures for bacteria, mycobacterium, and fungi were negative. The patients were evaluated by respirology and subsequently diagnosed with sarcoidosis. No patient has had further evidence of testicular cancer recurrence or required therapy for sarcoidosis.

Low and high power H+E stain showing non-caseating granulomas.
Discussion
Recently we have encountered three patients with a prior history of testicular cancer who subsequently presented with enlarged mediastinal lymph nodes. In each case work-up revealed sarcoidosis without evidence of tumor metastasis.
Sarcoidosis is a disease of unknown etiology without a specific diagnostic test. Sarcoidosis is diagnosed when other causes of non-caseating granulomatous disease have been excluded (Rybicki et al. 2001). Although sarcoidosis can be associated with many signs and symptoms up to half of patients present asymptomatically, often with enlarged thoracic lymph nodes (Muller et al. 1989). The treatment of sarcoidosis is controversial. Treatment is generally not indicated in patients with early stage disease, and minimal or no symptoms, since spontaneous remissions are common.
The association between testicular cancer and sarcoidosis has been seen in a variety of situations. Sarcoidosis has been found prior to a diagnosis of testicular cancer,(Rayson et al. 1998) as a synchronous diagnosis,(Heffner et al. 1990) and as a primary testicular mass (Emberton et al. 1992).
The most common sarcoidosis and testicular cancer presentation is as metachronous diagnoses (the diagnosis of sarcoidosis occurring after the diagnosis of testicular cancer) (Rayson et al. 1998). In fact, chemotherapy has been given for enlarged thoracic lymph nodes, that were thought to represent recurrent testicular cancer, which later were found to represent sarcoidosis (Waterston et al. 2006). Sarcoidosis has been estimated to occur 100 times more frequently in treated testicular cancer patients than in the general population, with a median time between diagnoses of about 5 years. The reason for this potential association is unknown, although surveillance bias may explain it (Rayson et al. 1998).
Isolated metastasis of testicular cancer in the mediastinum is uncommon. Non-seminomatous isolated thoracic metastasis has been estimated to occur less than 5% of the time while the rate of isolated thoracic metastasis in seminomatous disease may be higher (Rayson et al. 1998). Testicular cancer is most likely to recur within the first two years after diagnosis (Daugaard and Roerth, 2000). Recurrence would be uncommon 10 years after the initial diagnosis as was seen in two of our patients.
No imaging test can definitively establish the diagnosis of metastatic testicular cancer or sarcoidosis. Recent reports have shown that newer imaging modalities such as positron emission tomography (PET) scanning have mistaken sarcoidosis for recurrent testicular cancer (Cremerius et al. 1999). Tumor markers can aid in distinguishing between recurrence and sarcoidosis although these markers may be normal. Tissue diagnosis is therefore essential when the clinical picture remains unclear.
Sarcoid-like reactions have been described and can occur when malignant cells induce immune mediated granulomatous changes. This process has been observed in other malignancies, most commonly in Hodgkin's disease (Brincker, 1992). Sarcoid-like reactions may be difficult to distinguish from sarcoidosis when only a few tumor cells (micro-metastasis) induce granulomatous reactions. Some have suggested that immunohistochemical analysis may aid in identifying these patients. Placental alkaline phosphatase (PLAP) positivity in the absence of epithelial membrane antigen (EMA) has been thought to be a unique staining pattern for germ cell tumors, although this has been questioned (Karapetis et al. 2001; Niehans et al. 1988). Care must be taken before diagnosing a patient with recurrent testicular cancer based on immunohistochemical analysis results alone.
Conclusions
In conclusion, not all new lymphadenopathy in patients with a history of testicular cancer represents tumor recurrence. Sarcoidosis may present in a similar fashion to recurrent testicular cancer. Tissue diagnosis is essential when the clinical picture remains unclear.
