Abstract
Background
Despite the revolutionary success of introducing tyrosine kinase inhibitors (TKIs), such as imatinib mesylate (IM), for treating chronic myeloid leukemia (CML), a substantial proportion of patients’ treatments fail.
Aim
This study investigates the correlation between patient adherence and failure of TKIs’ treatment in a follow-up study.
Methods
This is a follow-up study of a new cohort of CML patients. Adherence to IM is assessed using the Medication Event Monitoring System (MEMS 6 TrackCap, AARDEX Ltd). The 9-item Morisky Medication Adherence Scale, medication possession ratio (MPR) calculation, and the electronic medical records are used for identifying potential factors that influence adherence. Clinical outcomes are assessed according to the European LeukemiaNet 2013 guidelines via reverse transcriptase quantitative polymerase chain reaction measurement of the level of BCR-ABL1 transcripts in peripheral blood. Response is classified at the hematological, cytogenetic, and molecular levels into optimal, suboptimal, or failure.
Results
A total of 36 CML patients (5 citizens and 31 noncitizen residents) consented to participate in the study. The overall mean MEMS score was 89. Of the 36 patients, 22 (61%) were classified as adherent (mean: 95) and 14 (39%) were classified as nonadherent (mean: 80.2). Adherent patients were significantly more likely to obtain optimal response (95%) compared to the nonadherent group (14.3%; P < 0.0001). The rate of poor adherence was as high as 39% using MEMS, which correlates with 37% treatment failure rate. The survey results show that 97% of patients increased the IM dose by themselves when they felt unwell and 31% of them took the missing IM dose when they remembered. Other factors known to influence adherence show that half of patients developed one or more side effects, 65% of patients experienced lack of funds, 13% of patients declared unavailability of the drug in the NCCCR pharmacy, and 72% of patients believed that IM would cure the disease. The MPR results reveal that 16% of patients had poor access to treatment through the hospital pharmacy.
Discussion and Conclusion
This is the first prospective study to evaluate CML patients’ adherence and response to IM in Qatar. The high rate of treatment failure observed in Qatar is explained by poor adherence. An economic factor (unaffordable drug prices) is one of the main causes of nonadherence and efforts should be made locally to improve access to medication for cancer diseases. Other risk factors associated with poor adherence could be improved by close monitoring and dose adjustment. Monitoring risk factors for poor adherence and patient education that include direct communication between the health-care teams, doctors, nurses, pharmacists, and patients are essential components for maximizing the benefits of TKI therapy and could rectify this problem. The preliminary results show that patients’ response to treatment may be directly linked to patients’ adherence to treatment. However, further in-depth and specific analysis may be necessary in a larger cohort.
Keywords
Background
Despite the revolutionary success of introducing tyrosine kinase inhibitors (TKIs), such as imatinib mesylate (IM), in treating chronic myeloid leukemia (CML), a substantial proportion of CML patients fail treatment with interruption and discontinuation of the drug, rapidly leading to reemergence from minimal residual disease into full-blown disease.1–11
Adherence to therapy and compliance with professional instructions are critical in the management of CML. Compliance is defined by the World Health Organization (WHO) report as “the extent to which a patients’ behavior taking medication corresponds with agreed recommendations from a health-care provider”. 12
Several studies suggest that poor adherence to IM is frequent and leads to worse clinical outcomes. Noens et al. 13 reported that patients with suboptimal response are significantly more likely to miss doses of their IM (23%) than those with optimal response (7%). Similarly, Marin et al. 14 showed that patients with a complete cytogenetic response (CCyR) are also less likely to neglect their IM (9%) compared to those with an incomplete cytogenetic response (23%). They show that the six-year probability of achieving major molecular response rates is significantly higher for patients who score more than 90% levels of adherence, followed by no complete molecular response (adherence ≤90%) and then no molecular responses (MRs; adherence ≤80%). 14
Furthermore, Ibrahim et al. 15 showed that the adherence rates in patients who eventually fail IM therapy is significantly lower (78.1%) than those of patients who respond to therapy (97.8%). In their study, patients with an adherence rate ≤85% had significantly lower event-free survival (EFS; 54.4%) than adherent patients (EFS rate: 91.4%). In addition, Jonsson et al. 16 showed that 97% of their patients achieved optimal response in correlation with optimal adherence. On the other hand, the results of a multicenter STop IMatinib Trial by Mahon et al. 17 showed that 61% of patients relapsed after discontinuation of IM, and Yhim et al showed that 71% of patients relapsed within 9.5 months after discontinuation of IM.14–19
In the first study of our series, we uncovered the mechanisms of resistance in 78% of patients, where two patients had BCR-ABL1 kinase domain mutations, one patient had E459K (rs1064156), one patient had a unique insertion of three nucleotides, six patients had additional chromosomal abnormalities as an underlying mechanism of resistance, four patients had no identifiable cause of resistance, and two patients were intolerant to treatment. However, 22% of the resistant cases displayed no explanatory underlying mechanisms.20,21
Thus, the main aim of this follow-up study is to investigate the relationship between adherence and failure of IM treatment in Qatar. Several methods including Medication Event Monitoring System (MEMS 6), 9-item Morisky Medication Adherence Scale (9-MMAS), medication possession ratio (MPR), and electronic medical records (eMR) are used. This study gives an insight into whether adherence affects patients’ responses to treatment and thus helps deign suitable interventions to increase patients’ awareness. As far as the author is aware, this is the first study to be conducted among patients in the state of Qatar.
Design and Method
Patient Recruitment
Patients aged 16–65 years (mean age: 42 years) who attended the NCCCR between January 2010 and December 2012 with Philadelphia chromosome positive (Ph+) CML for at least 12 months and received only IM treatment are included in this study. A total of 36 CML patients met the inclusion criteria and have consented and been recruited into the study.
Treatment Regimens
Patients in chronic phase (CP) received 400 mg orally once a day as first-line treatment IM, while accelerated phase (AP) patients received 600 mg once a day.
Adherence Measures
A holistic approach is taken to assess adherence of CML patients to treatment and correlate the result with response to treatment (Fig. 1).
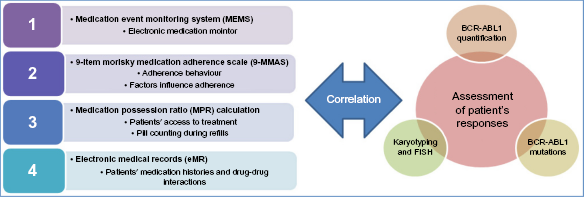
Assessment of patients’ adherence and response to IM and correlating with treatment response.
Adherence to IM is assessed using the Medication Event Monitoring System (MEMS 6 TrackCap, AARDEX Ltd).
In addition, the 9-MMAS, MPR calculation, and eMR are used to identify potential factors that influence adherence. The presence or absence of nonadherent behaviors is measured by the percentage of time a patient has access to medication and by reviewing patients’ medication histories. There are four methods employed as follows.
Method 1: Medication Event Monitoring System (MEMS)
Patient adherence over a period of 6–12 months is monitored in real time. The MEMS medication bottles contain a microelectronic chip fitted into the cap of a normal-looking medication bottle that automatically records and registers the date and time of every bottle opening. Each bottle opening is assumed to represent an event of medication intake. The system does not monitor the actual dose received; however, MEMS is considered the gold standard for measuring adherence.22,23 A cutoff of less than or equal to 90% is considered nonadherence.14,22,23
Patients are not notified about the monitoring system in the bottle caps but are told that their adherence is going to be monitored by counting the number of IM tablets returned.
Method 2: adherence and factors that affect therapy
The 9-item MMAS to measure and evaluate adherence to IM.

Questions 1–8 are answered by yes or no (Yy/Nn). Iin questions 1–4, 7, and 8, the answer “yes” gives 0 point and “no” gives 1 point. questions 5–6, the answer “yes” gives 1 point and “no” gives 0 point.
Question 9 is answered by “never/rarely” (5 points), “once in a while” (4 points), “sometimes” (3 points), “usually” (2 points), or “all the time” (1 point).
Potential factors that influence adherence to therapy.

Method 3: MPR calculation.
MPR is employed to gain insight into patients’ access to treatment. The MPR is calculated by dividing the sum of supply days of medication by the number of days between the first fill and the last refill. An MPR ≥ 80% is considered high adherence and is the benchmark most commonly reported in the literature.29,30
Method 4: eMR
The eMR is reviewed to gain insight into the patients’ medication histories and drug-drug interactions using MICROMEDEX® 1.0 (Healthcare Series). Patient adherence is correlated with patient's response treatment and response is assessed according to the 2013 European LeukemiaNet (ELN) guidelines.
Method 1: absolute quantification of BCR-ABL1
Peripheral blood samples are collected and the level of BCR-ABL1 transcripts measured locally via reverse transcriptase quantitative polymerase chain reaction (RT-QPCR). Patients’ responses to treatment are assessed via serial RT-QPCR for absolute quantification of BCR-ABL1 to monitor the ratio of BCR-ABL1 to normal ABL1 transcripts. The measurement of BCR-ABL1 is optimized locally according to the international guidelines of the Europe against Cancer (EAC). The absolute quantification of BCR-ABL1 is done using two different kits and methods [(1) Ipsogen BCR-ABL1 Mbcr IS-MMR DX and (2) Xpert BCR-ABL Monitor™, Cepheid].
The Ipsogen BCR-ABL1 Mbcr IS-MMR DX used in this study includes an IS-MMR calibrator, allowing conversion of results to the international scale (IS). The BCR-ABL1 IS is calculated using this formula.31–34
International Scale - Normalized Copy Number (IS-NCN)sample = (NCNsample x International Scale - Calibration IS-Cal value)/NCNcal
A fully automated cartridge-based assay (Xpert BCR-ABL Monitor™, Cepheid) combines sample preparation with real-time PCR. The results generated are converted to the IS using an assay-specific conversion factor determined by comparison to an IS reference assay.35–41
The MR is determined every third month by analysis of the BCR-ABL1 transcript level in peripheral blood (RT-QPCR) with ABL1 as a housekeeping gene. The BCR-ABL1 is reported in the IS 37 as well as described elsewhere.20,21
Method 2:assessment of patients’ responses
The ELN 2013 recommendations for the management of CML is adopted and employed in this study to assess the response/resistance of patients to treatment. Responses are defined at the hematological, cytogenetic, and molecular levels. Patients’ responses are classified as optimal, suboptimal, or failure. 42
Method 3:BCR-ABL1 mutations
Sequencing of tyrosine kinase domain of BCR-ABL1 is carried out to identify mutations as an underlying mechanism of resistance.
Sequencing reactions are performed in the forward and reverse directions separately using Big Dye chain terminator reagents on an ABI PRISM 3130 Genetic Analyzer, as described elsewhere. 21
Method 4: Karyotype Analysis and Fluorescence in Situ Hybridization (FISH)
Conventional karyotyping is carried out to identify ACAs as an underlying mechanism of resistance.
Chromosomes are identified and arranged according to the International System for Human Cytogenetic Nomenclature 2009. The number of cells investigated for each patient at each analysis ranges from 20 to 30 metaphases, and the cytogenetic response is assessed according to the ELN criteria. Cytogenetic analysis of metaphases of bone marrow samples as well as FISH for BCR-ABL on interphases of peripheral 100–200 white blood cells is done routinely as part of the clinical protocol, and the results are reported on the hospital's health information system.
Ethics
Patients gave their written informed consent to participate in the study that was conducted in accordance with the principles of the Declaration of Helsinki and was approved by Hamad Medical Corporation Research Committee [HMC (GC-1013)]. All data and patients’ identities were stored anonymously, and only the principal investigator has access to the code key. Thus, individual results were not seen by the treating physician.
Statistical Analysis
Descriptive statistics (means, ranges, frequencies, and percentages) were shown for each measure accordingly. Pearson correlation was used to investigate the relationship between the three adherence techniques (MMAS, MPR, and MEMS). In addition, Fisher's exact test was performed to study patients’ adherence and responses and the factors that affect them. A significance level of P ≤ 0.05 (two tailed) was considered. GraphPad Prism 5 statistical package was used for analysis.
Results
Demographic features results
Of the 36 patients, 28 (78%) were male and 8 (22%) were female, with more than half (N: 21; 59%) having an educational level of secondary school or less. A total of 27 patients (75%) were married while 9 patients (25%) were single. Patients were from 12 different countries with only 5 Qatari patients (13%; Table 3).
Demographic features of the 36 patients (survey questionnaire).

The clinical presentation of the 36 CML patients at diagnosis was as follows: 89% (32 patients) CP, 9% (3 patients) AP, and 3% (1) blastic crisis phase (BCP). A total of 35 patients were alive and one patient had died (Table 4).
Clinical features/characteristics of the 36 patients.

A total of 32 patients were treated with standard dose IM 400 mg, while 4 patients were treated with 600 mg due to a history of either loss of complete cytogenetic response (CCyR; n = 1) or suboptimal MR (n = 3).
Method 1: MEMS Results
The overall mean MEMS score was 89.0% (range: 40–100). Of the 36 patients, 22 (61%) were classified as adherent patients (mean: 95%; range: 91%–100%) and 14 (39%) were classified as nonadherent patients (mean: 80.2%; range: 40%–90%).
Adherent patients were significantly more likely to achieve optimal response [95% (21/22)] compared to the nonadherent group [14.3% (2/14); P < 0.0001; odds ratio (OR): 126, 95% confidence interval (CI): 10.3–1541; Table 5 and Fig. 2].
Patients’ response according to adherence status using the MEMS technique.


Patients’ adherence to IM using MEMS.
Method 2: adherence and factors that affect therapy
Patients’ response according to adherence status using the 9-MMAS technique.

The 9-MMAS revealed that 14 patients (38%) missed medication sometimes, 5 patients (14%) stopped taking their medication when they felt that the disease was under control or found sticking to treatment plan difficult. All patients (100%) had a special routine or reminder system to help them take medication on time, and 13 patients (36%) sometimes had difficulty remembering to take all their medicines.
Adherent patients were more likely to achieve optimal response (72%; 18/25) compared with the nonadherent group (45.5%; 5/11). However, this difference was not significant.
Patients behavior about IM intake and knowledge-based questions.

A total of 18 patients (50%) developed one or more side effects such as fatigue, nausea, vomiting, headache, muscle pain, abdominal pain, skin rash/itching, orbital edema, leg edema, memory change, sunburn, weight gain, or infection (muscle pain and skin rashes accounted for 35% and 29% of the side effects, respectively).
Side effects due to intake of IM.

Excluding five Qatari patients as the IM is free of charge for citizens while non-citizen residents pay the remaining 10% of IM, which is uncovered by the hospital.
Quality of educational information provided to the patients.

Comparison of categorical variables with adherence using the 9-item MMAS.

Method 3: MPR results
Of the 36 patients, 4 managed to obtain the IM at home. The mean of the overall MPR results for the 32 patients was 94% (range: 56.6–100).
Of the 32 patients, 27 (84%) were classified as adherent (MPR mean: 96%; range: 84.9%–100%) and 5 (16%) were classified as nonadherent (MPR mean: 57.8%; range: 56.6%–59.0%; Table 11).
Patients’ response according to adherence status using the MPR technique.

Tthe total number of patients were assessed via MPR is 32 as 4 patients were excluded in the MPR analysis because their medications were obtained in their own countries.
The adherent patient group was significantly more likely to have optimal response (74%; 20/27) than the nonadherent patient group (20%; 1/5; P = 0.037; OR: 11.43, 95% CI: 1.1–120.4; Table 11).
Method 4: eMR results
No drug-drug interactions were identified using MICROMEDEX® 1.0 (Healthcare Series).
The relationship between the three techniques: Pearson correlation was performed for the three techniques (MMAS, MPR, and MEMS). There was a significant high positive correlation between MPR and MEMS (r = 0.74; P = 1.053373e−006), MMAS and MEMS (r = 0.66; P = 3.547150e−005), and MPR and MMAS (r = 0.5; P = 0.0036).
Patients’ Response
Of the 36 patients, 23 (63%) responded optimally and 13 (37%) failed the treatment.
Those who responded optimally had complete hematological response, CCyR, and deep MR.
Of the 23 patients who responded optimally, 22 were from CP and 1 was from AP according to their initial diagnosis, and thus, patients’ initial disease phase (CP, AP, and BCP) could be attributed to the treatment outcomes.
It is important to note that achieving early-stage treatment targets is more likely to have better long-term outcomes. This study found that of the 23 patients who responded optimally, 22 achieved early-stage treatment and these patients continued to have optimal response.
On the other hand, in total, 13 patients failed treatment, 12 were resistant, and 1 (P25) showed intolerance. Of the 12 resistant cases, 1 patient (P14) showed both E459K mutation and ACAs, 3 patients (P6, P20, and P24) had ACAs, and all IM failed patients switched to the second line of treatment, either dasatinib or nioltinib.
Long-term disease progression or survival rates upon IM treatment showed that of the 36 patients, only 1 (P25) (2.8%) was deceased, and this patient was known to have primary resistance to all TKIs. None of the 36 patients progressed, and 35 (97.2%) survived (Supplementary Table 1).
Discussion and Conclusion
This is the first prospective study to evaluate CML patients’ adherence and response to IM in Qatar. The rate of treatment failure of CML patients treated with IM in Qatar has been previously reported to be high (54%).20,21 Due to this high rate of IM failure in Qatar, patient's adherence to treatment was studied by observing a new cohort of 36 CML patients. One-third of the patients (N: 14, 39%) were classified as nonadherent and 13 patients (37%) failed treatment. Thus, poor adherence explains the high rate of treatment failure observed. Nonadherence to the treatment was one of the most common causes of IM failure in the patient cohort, documented in 39% of cases using the gold standard method (MEMS), which seems to be consistent with international data, indicating nonadherence rates between 25% and 50%. 43 Economic factors (eg, unaffordable drug prices) were one of the main causes of nonadherence. Efforts should be made locally to improve access to medication for cancer diseases or through participation in patient assistance programmes such as Glivec International Patient Assistance Programme (GIPAP), which provides Glivec free of cost to eligible patients in developing countries who meet specific medical and socioeconomic guidelines. Briefly, GIPAP helps patients who are not insured, not reimbursed, cannot pay for treatment privately, and are in developing countries that have minimal reimbursement capabilities, 44 or using generic IM that has been available in the market since February 2016 which could reduce the cost burden of treatment. Other risk factors associated with poor adherence can be improved by close monitoring and dose adjustment. Monitoring risk factors for poor adherence in combination with patient education that includes direct communication between the health-care teams (ie, doctors, nurses, and pharmacists) and patients are essential components for maximizing the benefits of TKIs and could rectify this problem. The preliminary results show that patients’ responses to treatment may be directly linked to patients’ adherence to the treatment. However, further in-depth and specific analysis may be necessary in a larger cohort.
In several studies, such as a Belgian study that included 169 patients, adherence to IM treatment was assessed via questionnaires, interviews, and pill counts. A total of 30% of the patients were shown to be nonadherent, and only 14% of the patients took the prescribed IM dose. 13 In a study from the United Kingdom, 26% of the patients were shown to be nonadherent. 14 In a follow-up study, the most common reason for intentional nonadherence was side effects, and the most common reason for unintentional nonadherence was forgetfulness. 45 In a Taiwanese study, Chen et al. 19 showed that 26.9% of patients showed poor adherence to IM. In these three studies, in Belgium, Britain, and Taiwan, a relationship between nonadherence and treatment response was observed, which is consistent across the studies.14,19,45 Chen et al. 46 also showed in another study that Taiwanese CML patients were adherent to IM and reported that adverse drug effects and associated polypharmacy were the main and key reasons and concerns influencing their adherence to long-term use of IM.
Interestingly, in a Swedish study, Jonsson et al. 16 showed that 97% (37) of CML patients were classified as adherent and all patients optimally responded to treatment.
Nonadherence and treatment interruptions both lead to undesired clinical outcomes and appear to be more prevalent than previously believed or expected.
There are some limitations to the study. MMAS is a subjective method and is the least reliable of the methods. However, a questionnaire survey is able to identify individual patient's concerns and subsequently tailor appropriate intervention.
Certainly, the disadvantages of such approaches should not be underestimated. Relatively poor sensitivity and specificity can occur due to false data input by patients, accidently or purposefully, or imperfect communication skills and questions constructed by interviewers or in the design of the survey.
Negativity in questions, suggesting blaming patients for not fulfilling their prescribed regime, may lead to bias. Patients’ psychological state can impact their answers to the questionnaire.
Physicians, nurses, and pharmacists need to educate patients and closely monitor their adherence to treatment. Improving adherence and limiting treatment interruptions would optimize clinical outcomes and reduce the burden of disease, and therefore should be assessed routinely as they are correlated with poor response to treatment.
Author Contributions
Designed the study and wrote the manuscript: NIA-D. Recruited patients into the study as well as provided clinical data: MAY. Dispensed IM in the MEMS bottles: RSG. Interviewed patients and collected survey questionnaires’ results: CCG, SKB, AJN. Carried out the molecular studies and assisted in data collection: MAI, AAA, MMA, RMA-J. Assisted in writing the initial draft of the manuscript and in statistical analysis: HMM, TIB-O, MMS. Assisted in designing the survey questionnaires and MPR calculation: RBK. In addition, all the authors provided critical revisions to the various manuscript drafts. All the authors read and approved the final manuscript.
Supplementary Material
Supplementary Table 1
Showing the 36 patients responses’ to IM treatment.
