Abstract
Background
Patients with recurrent or metastatic head and neck cancer (HNC) have a poor response and survival with currently available chemotherapy agents. Thus new agents are needed. We report the results of a phase II trial of irinotecan in patients with metastatic or recurrent HNC.
Patients and Methods
Chemonaive patients were treated with irinotecan 125 mg/m2 on a weekly schedule for four weeks, followed by a two week rest. However, due to excessive toxicity, the dose and schedule of irinotecan was changed to 75 mg/m2 on days 1 and 8 of a 21 day cycle. All previously treated patients received 75 mg/m2 on days 1 and 8 of a 21 day cycle.
Results
Forty-nine patients were enrolled on study. The response rate was 20% in the chemonaive patients treated at the 125 mg/m2 dose. The response rate decreased to 12.5% at the 75 mg/m2 dose. In the previously treated cohort, the response rate was 20%. Median survival for the chemonaive and previously treated cohorts were 6.7 months and 5.0 months, respectively.
Conclusions
Irinotecan has modest activity in chemonaive and previously treated HNC, thus it merits further study in this advanced disease population.
Introduction
Despite progress in the treatment of locally advanced head and neck cancer, a significant percentage of patients develop local recurrence or distant metastases. The majority of recurrent and metastatic patients are not candidates for curative treatment with either surgery or radiation therapy. Unfortunately, chemotherapy in this setting remains palliative, not curative. Median survival for patients with recurrent disease using contemporary regimens is 6–9 months with a 1-year survival of approximately 30% (1). Multiple single agents have shown modest activities with response rates varying between 15% and 40%. Unfortunately, randomized trials have failed to demonstrate a survival advantage for the use of combination chemotherapy over single agents alone in the metastatic/recurrent setting (1–3). Even taxanes and taxane based combinations have failed to demonstrate a significant improvement in survival in the metastatic or recurrent population (4–7). For patients who respond to chemotherapy, tumor regression is brief, responses are usually partial, and the impact on survival has been minimal. Therefore, studies to identify new active agents are warranted.
Irinotecan, a camptothecin analogue, inhibits topoisomerase I, an enzyme which is critical for normal DNA replication. Topoisomerase I creates single-strand breaks in the DNA backbone, allowing relaxation and religation of the DNA during replication. Inhibition of this process leads to replication arrest, double strand-breaks and ultimately apoptosis. The activity of irinotecan stems from its active metabolite, SN-38, which is 1000 times more potent than the parent compound. Irinotecan demonstrates significant activity in multiple malignancies and is FDA approved for the treatment of colorectal carcinoma (8–11).
In vitro and animal studies demonstrate that irinotecan has substantial anti-tumor activity against a number of common solid tumor cell lines (8–11). We decided to investigate the efficacy and toxicity of irinotecan in patients with chemonaive or previously treated recurrent or metastatic squamous cell carcinoma of the head and neck.
Materials and Methods
Patient selection
Forty-nine patients were enrolled on study between 1997 and 2000. Patients had histologically confirmed local-regionally recurrent or metastatic squamous carcinoma of the head and neck. Patients were deemed incurable by surgery or radiation therapy. Inclusion criteria included ECOG performance status 0–1, adequate hematologic, hepatic and renal parameters, absence of clinically apparent brain metastases, bidimensionally measurable disease, and lack of significant comorbidities. Exclusion criteria included: nasopharyngeal primaries, prior topotecan or irinotecan. Patients were stratified based on prior therapy. Cohort 1 included chemonaive patients with either previously untreated metastatic disease or local-regional recurrence after primary therapy. Cohort 2 included patients who had received at least 1 chemotherapy regimen aimed at treating metastatic or recurrent disease. Patients who had received chemotherapy as a part of combined modality primary treatment a minimum of 6 months prior to recurrence were deemed chemonaive and included in cohort 1. If recurrence developed sooner than 6 months, patients were considered chemo failures and included in cohort 2.
Methods
The protocol was approved by the Institutional Review Board at the Vanderbilt University School of Medicine and all patients signed informed consent. The first 22 patients on Cohort 1 received irinotecan at 125 mg/m2 over a 90-minute intravenous infusion on days 1, 8, 15, 22, followed by a two week rest period. Dose reductions in increments of 25 mg/m2 were made for ≥Grade 2 hematological or nonhematological toxicities that occurred during a course of therapy and for ≥Grade 3 hematological or nonhematological toxicities that occurred at the start of subsequent cycle. Due to excessive toxicity, the dose and schedule were subsequently amended to irinotecan at 75 mg/m2 on days 1, 8 of a 21-day cycle for Cohort 1. All patients in Cohort 2 received irinotecan 75 mg/m2 on days 1, 8 of a 21-day cycle. Sequential dose-escalation to 100 mg/m2 and 125 mg/m2 occurred if no significant toxicity was noted after 6 weeks. Dose-decrease to 50 mg/m2 occurred for clinically significant grade 2 or greater toxicity. Patients were assessed for response every 6 weeks using standard World Health Organization (WHO) criteria. Patients were only deemed eligible for response evaluation if they remained on study for six consecutive weeks. Any patient who received drug was eligible for toxicity evaluation. Patients with stable disease or better were allowed to continue on therapy to a maximum of nine treatment cycles. Patients with progressive disease, unacceptable toxicity or desire to withdraw from study were removed from this therapy. Toxicities were graded according to the NCI Common Toxicity Criteria (CTC) version 2.0.
Statistical methods
The primary objective of this phase II clinical trial was to determine the response rate of irinotecan in patients with chemonaive recurrent/refractory or metastatic head and neck cancer (HNC) (Cohort 1). The study was designed to detect a true overall response rate of at least 20%. A two-stage accrual design described by Simon was employed (12). Initially, 15 eligible patients would be entered onto the study. If there was no response in the first 15 patients, the trial would be terminated with the conclusion that there was little evidence to suggest that the overall response rate would reach 20%. If one or more responses in the first fifteen patients were seen, the trial would accrue to a total of 37 patients. Assuming that the overall response for patients without treatment was 5%, the design provided a statistical power to detect differences of 85% with a significance level of 0.05. Given the early responses noted in Cohort 1, the study was amended to add a second cohort to explore the efficacy of irinotecan in previously treated patients with recurrent or refractory metastatic HNC.
Results
Forty-nine patients were enrolled on study: 34 in cohort 1 and 15 in cohort 2. The accrual goal was not met as the study was ultimately terminated due to a slow accrual and the need to open a high priority, competing protocol at this institution. Patients were considered evaluable for toxicity if they received any therapy, but they were evaluable for response only if they remained on study for at least six consecutive weeks. In cohort 1, twelve patients were not evaluable for response: one due to missing data and eleven patients were removed from study due to significant toxicity prior to the six week response evaluation. Thus, 22 patients are evaluable for response. In cohort 2, one patient was not evaluable for toxicity as they did not receive any therapy. Six patients were removed from study due to toxicity prior to the six week response evaluation and thus were not evaluable for response. Thus 15 patients were evaluable for toxicity and 8 patients were evaluable for response. Patient characteristics are shown in Tables 1 and 2.
Patient Characteristics Cohort 1.

Patient Characteristics Cohort 2.

Toxicity
In cohort 1, the first 22 patients were treated with irinotecan at 125 mg/m2 IV weekly for four weeks, followed by a 2 week-rest. However, toxicity was excessive. See Table 3. Five patients required hospitalization due to gastrointestinal toxicity or febrile neutropenia. Twelve patients required dose reductions. The majority of dose reductions were due to gastrointestinal toxicity. This included four patients with grade 2 diarrhea. With the amended dose and schedule of irinotecan, toxicity was improved. In fact, six patients underwent intrapatient dose escalation. Four patients required dose reduction to the 50 mg/m2 dose.
Number of Patients with Grade 3 and 4 Toxicities in Cohort 1.
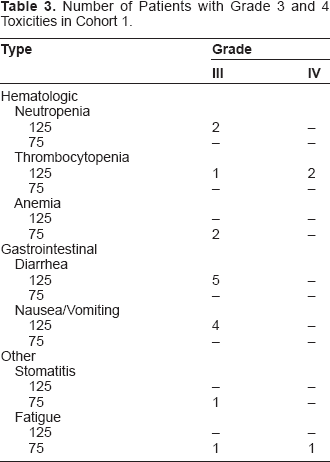
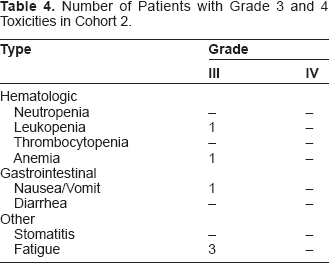
In cohort 2, six patients were removed from study due to excessive toxicity. See Table 4. Five of the eight remaining patients underwent dose escalation to 100 mg/m2 with one patient subsequently undergoing a second dose escalation to 125 mg/m2. Three patients, including one patient who was dose escalated to 100 mg/m2, required dose reduction due to toxicity.
Number of Patients with Grade 3 and 4 Toxicities in Cohort 2.
Response
The overall response rate for evaluable patients in cohort 1 was 20% for patients treated at the 125 mg/m2 dose and schedule and 12.5% at the 75 mg/m2 dose level. Sixty-one percent of patients demonstrated stable disease, which lasted an average of 4.5 months. Mean overall survival for cohort 1 was 9.3 months. The median overall survival was 6.7 months. In cohort 2, two of nine evaluable patients demonstrated a PR for a response rate of 22%. Fifty-four percent of patients demonstrated stable disease, which lasted an average of 4.3 months. Importantly, three of the six patients who had a best response of stable disease remained on study for 8 to 9 cycles of therapy. The mean overall survival rate for this cohort was 6.6 months with a median overall survival of 5.0 months.
Discussion
We now report the results of a phase II trial of irinotecan in chemonaive and previously treated patients with recurrent/refractory HNC. Irinotecan demonstrated activity in chemonaive patients and is comparable to other commonly used chemotherapy agents. In the chemo naïve cohort, the response rate decreased from 20% to 12.5% with dose reduction, suggesting a dose-response relationship. However, the toxicity of irinotecan 125 mg/m2 was prohibitive. In fact, 12 patients (35%) were removed from study prior to response assessment due to excessive toxicity. Excessive toxicity was manifested early in the treatment course, with a significant number of patients receiving only 1 or 2 doses. Irinotencan was better tolerated at the 75 mg/m2 dose level.
Importantly, irinotecan demonstrated activity in the previously treated population, a group targeted by very few clinical trials due to poor prognosis and performance status. The response rate was 20% with a median survival 6.7 months. Similar to the chemonaive patient population, 6 patients (40%) were removed from study due to excessive toxicity prior to response evaluation. Despite the excessive toxicity, a number of patients remained on study for extended periods of time (8 to 9 cycles in three patients). The unexpectedly high response rate and survival in the previously treated population may be a result of an inherent selection bias. That is, cohort 2 represented a group of patients who maintained a good performance status despite previous therapy. Alternatively, these patients may have a favorable disease biology.
The results of this study are limited in several ways. The study failed to meet its accrual goal (34 enrolled versus 37 planned for the chemonaive population). Secondly, a significant number of patients were removed from study due to toxicity prior to the six week response evaluation. Because of the specifications in the protocol, patients who did not complete a full cycle of chemotherapy were nor evaluable for response. The excess toxicity and high percentage of patients who were removed early from study were not anticipated. Of note, however, all patients were included in the survival analysis.
While the patient numbers in both the chemo-naïve and the previously treated patients are too small to make firm conclusions, the study does demonstrate several very important issues. Firstly, irinotecan has activity in the treatment of recurrent, metastatic HNC in both a chemo-naïve and previously treated population. Secondly, irinotecan at 125 mg/m2 administered on a weekly schedule for four weeks, followed by a two week rest, is a toxic regimen in this patient population. At the time of this study, patients were not tested for the UGT11 polymorphism. The recognition of the potential role of the UGT1A1 polymorphism in irinotecan handling may ultimately allow clinicians to prospectively determine which patients will have the best risk/benefit ratio with the use of this drug (13–14).
Based on our results, we designed and executed a prospective phase II evaluation of the combination of irinotecan and cisplatin in patients with recurrent or metastatic SCCHN at the Vanderbilt University Ingram Cancer Center and VICCAN (Vanderbilt Ingram Cancer Center Affiliated Network). The results are being analyzed at this time. The Eastern Cooperative Oncology Group (ECOG) also recently presented the results of E3301: A phase II trial of weekly irinotecan and docetaxel in recurrent or metastatic head and neck cancer. An interim analysis of this study revealed a response rate of 24% in the chemonaive cohort with a median progression free survival of 3.2 months and an overall survival of 9.8 months. Accrual is ongoing (15).
Interest in irinotecan for the treatment of HNC patients has increased with the introduction of targeted agents. Bortezomib is a novel anticancer agent that targets cells by inhibition of the ubiquitin-proteasome pathway (critical for cellular protein homeostasis). Dysregulation of the NF-kB pathway in cancer cells promotes cell survival and plays a significant role in tumor cell resistance to irinotecan (16–18). This combination of agents is presently being studied in both chemo-naïve and chemotherapy failure patients with recurrent/metastatic HNC through an Eastern Cooperative Oncology Group (ECOG) trial.
In summary, irinotecan demonstrated evidence of clinical response in a poor prognosis group of patients. The therapeutic benefit may be significantly increased by overcoming irinotecan resistance through the addition of novel targeted agents. Clinical trials of these novel combinations should be investigated further in clinical trials.
Note
No potential conflict of interest relevant to this article are reported.
