Abstract
Nanobacteria (NB) contribute to pathological calcification in the human and animal body. It has been isolated from salivary stones and suggested that it may act as a nucleus for the initiation of these stones. In the present study, we examined its role in the recurrent salivary gland stones using immunodetection with NB-specific monoclonal antibodies and scanning electron microscopy (SEM) hoping to provide a method for preventing the recurrence of these stones in the patient that has suffered from salivary stones. Our study comprised 30 patients with recurrent salivary gland stones (group I) and 30 patients with salivary gland stones for the first time (group II), in addition to 30 normal controls (group III). We could detect 100–500 nm nanoparticles in 24/30 (80%) cases in group I with significant difference <0.05 and <0.01 when compared with group II and group III in which they were detected in 19/30 (63.3%) and 6/30 (20%) respectively. Also there was a significant difference <0.05 between group II and group III. We proposed that salivary stone formation is a nanobacterial disease initiated by bacterial infection. This bacteria may play an important role in the recurrence of salivary stone. So the use of calcium chelator, ethylene diamine tetra acetic acid (EDTA), before or in combination with the suitable antibiotic that is given in an amount effective to inhibit or prevent the growth and development of nanobacteria may eradicate these stones and prevent their recurrence.
Keywords
Introduction
Salivary gland swelling can occur when one of the ducts that carry saliva from the salivary gland to the mouth is blocked. The most common cause of blockage is the stones. Salivary gland stones are most common in adults; 25% of those with stones have more than one. A stone can form from salts contained in the saliva. Blockage makes saliva back up inside the duct, causing the salivary gland to swell. A typical symptom of a blocked salivary duct is swelling that worsens just before mealtime or particularly when a person eats a pickle, a sour pickle's taste stimulates saliva flow. 1 Mumps, certain bacterial infections, and other diseases (such as AIDS, Sjögren's syndrome, diabetes mellitus, and sarcoidosis) may be accompanied by swelling of the major salivary glands. Swelling also can result from cancerous or noncancerous tumors in the salivary glands. Swelling resulting from a tumor is usually firmer than that caused by an infection. If the tumor is cancerous, the gland may feel stone-hard and may be fixed firmly to surrounding tissues. Most noncancerous tumors are moveable. 2
The reason why a stone forms is not known. A salivary gland stone is sometimes called a sialolith or a salivary calculus. Most salivary stones are mainly made of calcium however, there is no abnormality of the blood calcium level or any other problem with calcium in the body. Salivary gland stones are not usually associated with any other disease. 3 Apparently idiopathic extra skeletal calcifications containing calcium mineral salts are a common medical problem. 4 This calcification is a complicated, actively regulated process of mineralization that similar to bone formation and remodeling. 5 Mineralogists found that for crystal formation/biomineralization to start, nidi (nucleus) and an environment of available dissolved components at or near saturation concentrations, along with the absence of inhibitors for crystal formation are needed. Bacteria or other agents producing such nidi, if present in body fluids, are very likely candidates to launch and accelerate pathologic calcification in vivo. 6
Nanobacteria is a perfect model for studying biogenic mineralization/calcification because NB is a self-replicating particle and has less complicated metabolic pathway, accumulates calcium and phosphate under physiologic conditions, produces a calcium phosphate mineral similar to bone and exists in physical conditions (e.g. pH, gravity, temperature) that is easy to manipulate and that can be replicated for the physiologic model. NB is also called Calcifying Nano Particles (CNP) and has unique properties, including an extremely small size (100–500 nm). 7 Nanobacteria contributes to pathological calcification in the human and animal body, including diseases such as kidney stones, salivary gland stones, dental pulp stones and atherosclerosis. 8
This study aimed to find the relation between nanobacteria in saliva and recurrent salivary gland stones using immunodetection with NB-specific monoclonal antibodies and scanning electron microscopy (SEM) hoping to provide a method for preventing the recurrence of salivary stones in a patient that has suffered from salivary stones, comprising administration of calcium chelator, ethylene diamine tetra acetic acid (EDTA), before or in combination with the suitable antibiotic that is given in an amount effective to inhibit or prevent the growth and development of nanobacteria.
Patients and Methods
Our study comprised 30 patients with recurrent salivary gland stones (group I) which were previously removed by either gentle probing into the duct from inside the mouth with a thin blunt instrument to free the stone which then falls into the mouth or therapeutic sialendoscopy using a very thin endoscope (tube) with a camera and light at the tip, and 30 patients with salivary gland stones for the first time (group II). They attended the Kasr El-Aini ENT Clinic during the period 2007–2009, in addition to 30 normal controls (group III). The patients complained of pain and swelling of the affected gland at meal times, this occurs if the stone completely blocks a duct. The pain was sudden and intense just after starting a meal. Swelling soon followed. The pain and swelling eased over about 1–2 hours after a meal. However, in most cases stones did not block the duct completely and only partially block saliva flow, or not block the flow at all if it was embedded in the body of the gland. In these situations the symptoms were one or more of the following: Dull pain from time to time over the affected gland, swelling of the gland which was persistent, or vary in size from time to time. Infection of the gland occurred in some cases causing redness and pain and developed into an abscess (ball of infection and pus). Some people with salivary gland stones had no symptoms at all. A stone was found by chance on an x-ray picture taken for another reason. However, in about 6 of the 60 cases, the stone did not appear on the x-ray and other tests were done:
A scan such as a CT scan, ultrasound scan or MRI scan.
Sialography (silogram). This is a special x-ray test. It helps to show up the gland, the duct and any abnormalities of the duct such as a stone. For this test a small plastic tube is passed into the affected duct. A dye is then injected into the salivary gland. The tube is removed and x-ray pictures are taken. The x-ray pictures show up the dye within the gland and duct. This gives a good outline of the structures, and shows up any abnormalities.
Sialendoscopy. In this test a very thin tube (endoscope) with a tiny light and camera at the tip is pushed into the salivary duct. The doctor can then see directly into the duct and gland to see if a stone is present.
Saliva was collected from all subjects and filtered with 0.22-µm membrane. 9 Part of saliva was spread on slides pretreated with 3-APTES. A standard avidin-biotin immunoperoxidase technique was used. 10 The slides were treated with 0.3% hydrogen peroxide to inhibit the activity of endogenous peroxidase. Slides were incubated overnight at 4 °C with the primary anti-NB-specific monoclonal antibodies. 11 (Santa Cruz Biotechnology, Santa Cruz, California). The antibody were diluted 1:50 in phosphate buffer saline (PBS) and incubated for 24 hours at 4 °C. The specificity of this antibody was demonstrated previously. 12 The following day, the slides were washed 3 times in PBS, then slides were incubated for 15 minutes with biotinylated secondary antibody and then with avidin-biotin complex horseradish peroxidase solution (Vector, Burlingame, California). After incubation for 10 minutes, the peroxidase activity was revealed by the addition of freshly prepared diaminobenzidine (0.03%) for 20 minutes at 37 °C in dark then washed 3 times in PBS or tries-HCL buffer pH 7.6. Slides were counter stained with May-Grunwal-Giemsa stain and Meyer's hematoxylin, respectively. Negative controls were processed in an identical manner by substitution of primary antibody with a normal rabbit IgG.
Other part of saliva was diluted 1:10 with ultra pure water. A drop from each of them (10 µl) was put in closed polystyrene Petri dish sealed with a permeable tape and placed together with two other dishes containing water into a larger Petri dish which was closed to extend the time of evaporation to be examined by SEM after an evaporation time of 72 hours at 20 °C or 48 hours at 30 °C. 13
Results
Both immunodetection technique using NB-specific monoclonal antibodies and SEM, revealed 100–500 nm nanoparticles in 24/30 (80%) cases in group I with significant difference <0.05 and <0.01 when compared with group II and group III in which we detected 19/30 (63.3%) and 6/30 (20%) respectively (Figs. 1–3) (Table 1). Also there was a significant difference <0.05 between group II and group III.
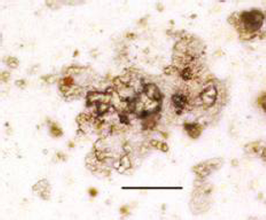
Light micrograph of saliva of a patient with recurrent salivary gland stones showing positive immunoperoxidase reaction for NB using NB-specific monoclonal antibodies (contrasted section with May-Grunwal-Giemsa stain; x 400). (Bar = 1 µm).
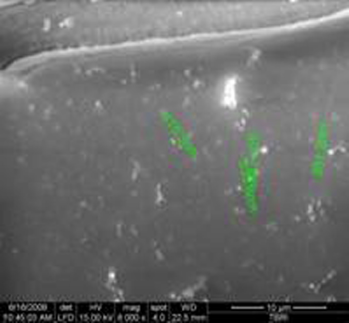
SEM showing nanoparticles (100–500 nm) in saliva of a patient with recurrent salivary gland stones appear either single or in groups (arrow).

Nanoparticles (100–500 nm) could be detected by SEM in saliva of a patient with salivary gland stones either single or in groups (arrow).
The relation between the detection of nanoparticles in saliva and salivary stones.

Significant difference <0.05 when compared to group I;
Significant difference <0.01 when compared to group I;
Significant difference <0.05 when compared to group II.
Discussion
Nanobacteria is a cytotoxic, sterile-filterable, gram-negative, atypical bacteria that could be detected in bovine and human fluids. It can produce carbonate apatite on its cell wall. Also, this extremely small bacteria is capable of precipitating calcium salts implicated in the pathogenesis of calculi. The pathogenesis of this calcinosis cutis is incompletely understood. 8
In this study, immunodetection technique using NB-specific monoclonal antibodies and scanning electron microscopy (SEM) highlighted the resemblance in size and morphology of nanobacteria in 43/60 (68.8%) patients with salivary gland stones (group I & II) with significant difference <0.01 when compared with normal controls (group III) in which we could detect these particles in 6/30 (20%).
Our results exist that nanobacteria behaves as a microbe that appears to show a correlation with calcification-related salivary gland stones. This agent has unique properties, including an extremely small size (0.1–0.5 mm), as seen in Figures 2, 3. The biologic characterization of NB is yet to be fully understood, Kajander and Ciftcioglu found that the precipitation and growth of calcium phosphate readily occur in systems containing trace amounts of NB but not in identical control systems lacking NB. 14 The exact mechanism(s) by which apatite is nucleated and formed around NB is unknown. Ciftcioglu et al found that when the serum concentration in the medium is reduced (# 5%) in the NB culture conditions, NB starts to mineralize and grows larger in size owing to calcium and phosphate deposition on its surface. 7 Kajander et al 15 also could detect by the electron and light microscopy nanobacteria-like particles scraped from DMEM-containing subcultures of 0.45-µm membrane-filtered saliva.
Also, scanning electron microscopy (SEM) highlighted the resemblance in size and morphology of nanobacteria in 24/30 (80%) patients with recurrent salivary gland stones (group I) with significant difference <0.05 and <0.01 when compared with patients with salivary gland stones for the first time (group II) and to the normal controls (group III) in which it was detected in 19/30 (63.3%) and 6/30 (20%) respectively. Also there was a significant difference <0.05 between group II and group III.
Our results are in agreement with Kajander et al 16 who found that nanobacteria shelters itself from the immune system and the antibodies (calcific semi-dormant defense) and it can live where other bacteria cannot (extremophilic defense) as it has a calcific” defense. They also found that the biofilm elaborated by individual nanobacteria renders it “sticky”, so that it can bind to mammalian cells, trick the cell into endocytosing them, and then causes the invaded cell to commit apoptosis.
Kajander 17 and Shoskes et al 18 found that Nanobacteria susceptibility to tetracycline or the high doses of aminoglycoside antibiotics can be increased with, calcium chelator such as ethylenediaminetetraacetic acid (EDTA), cytosine arabinoside, 5-fluorouracil, or gamma irradiation.
Conclusions
We proposed that salivary stone formation is a nanobacterial disease initiated by bacterial infection and subsequently endogenous and dietary factors may influence their progression. This bacteria may play an important role in the recurrence of salivary stone. So the use of calcium chelator such as ethylenediaminetetraacetic acid (EDTA) before or in combination with the suitable antibiotic may eradicate these stones and prevent their recurrence.
Disclosures
This manuscript has been read and approved by all authors. This paper is unique and is not under consideration by any other publication and has not been published elsewhere. The authors and peer reviewers of this paper report no conflicts of interest. The authors confirm that they have permission to reproduce any copyrighted material.
