Abstract
Head and neck cancer is a broad term used to describe malignancies that arise in the nasal and oral cavities, pharynx and larynx, as well as the paranasal sinuses. Head and neck squamous cell carcinoma (HNSCC) affects the squamous epithelium of the oral cavity, tongue and oropharynx, excluding the nasopharynx. Recent advances in molecular technology, including gene expression and proteomic profiling appear to offer the potential for the development of specific biomarkers including diagnostic tools which may act as an aid to guide therapy for this malignancy. The other human head and neck cancer included in this review, nasopharyngeal carcinoma (NPC) is a malignancy derived from the undifferentiated epithelium of the nasopharyngeal cavity, and is considered here as a separate entity because its strong association with Epstein-Barr virus (EBV) presents the opportunity for the development of virus related and unrelated biomarkers. In particular, IgA antibodies to EBV and high levels of EBV DNA in serum samples of NPC patients have been recorded. This review aims to summarize some current and also potential new biomarkers that could be used for screening, diagnosis, monitoring and prognostic prediction for cancers of the head and neck, including NPC and HNSCC.
Introduction
Cancers of the head and neck
Head and neck cancer is a broad term used to describe malignancies that arise in the nasal and oral cavities, pharynx and larynx, as well as the paranasal sinuses. Most of these epithelial cancers are squamous cell carcinomas. Here, we review the epidemiology and treatment of these cancers, as well as biomarkers in use and in development, for diagnosis, prognosis and therapy.
Head and Neck Squamous cell Carcinoma (HNSCC)
Head and neck squamous cell carcinoma (HNSCC) affects the squamous epithelium of the oral cavity, tongue and oropharynx, excluding the nasopharynx (see below). It is the sixth most common type of cancer, representing approximately 6% of all new cases (1). HNSCC is thought to account for an estimated 650,000 new cancer cases and 350,000 cancer deaths worldwide per year (1). High risk regions for the disease are known, including south-central Asia for cancers of the oral cavity, and South America and western Asia for laryngeal cancers (1). Two-thirds of patients present with advanced disease, usually involving regional lymph nodes, with 80% of cases eventually metastasising to involve the lymph nodes of the neck. However, distant metastasis at presentation is comparatively rare. It is well established that the major risk factors for HNSCC are tobacco and alcohol consumption. There is increasing evidence however, for human papillomavirus having a role in the cause of at least a subset of HNSCC.
Alcohol consumption and smoking tobacco have been implicated in approximately 75% of HNSCC (2,3). Among consumers of both products, risks of oropharyngeal cancer tended to combine more in a multiplicative than additive fashion and were increased more than 35-fold among those who consumed two or more packs of cigarettes and more than four alcoholic drinks per day (2). Meta-analysis indicated that tobacco had a stronger effect on the larynx than on any other site, while alcohol's effect was strongest on the pharynx (3). Significant alcohol consumption or smoking alone have also been associated with HNSCC. Chewing tobacco or betel quid are known risk factors for HNSCC (4).
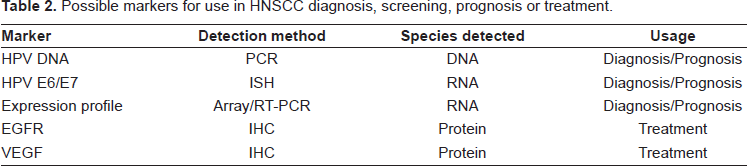
HPV, in particular HPV 16, is a causal factor for HNSCC. Approximately 15 to 25% of HNSCC contain genomic DNA from HPV (5), and the association varies based on the site of the tumor. HPV DNA is found most often in cancers of the tonsil (45 to 67%), less often in the hypopharynx (13%–25%), and least often in the oral cavity (l2%–18%) and larynx (3%–7%) (6). There is emerging evidence suggesting that HNSCC with HPV is a distinct clinicopathological entity, and presence of HPV is a favorable prognostic marker (7) see Table 2. The high risk HPVs, such as HPV 16 and HPV 18, encode two oncoproteins (E6 and E7), which induce cellular transformation and alter cell cycle control. E7 binds to pRb and induces its proteolytic degradation, whereas E6 inactivates p53 by accelerating its ubiquitin-mediated proteolysis (8).
HNSCC is thought to arise from a multistep process with histologically distinct precursor lesions that harbour specific genetic alterations. Normal appearing epithelium first shows signs of epithelial hyperplasia, which progresses to mild dysplasia then severe dysplasia or carcinoma in situ and finally to truly invasive carcinoma. Loss of heterozygosity (LOH) of 9p21 appears to be an early event in squamous neoplasia of the head and neck and has been found in preneoplastic lesions, including 30% of cases of squamous hyperplasia (9). Loss of chromosomal region 9p2l is found in 70%–80% of cases, thus representing the most common genetic alteration seen in squamous dysplasia and HNSCC (9, 10). The CDKN2A gene locus found at chromosome 9p21 encodes two different transcripts, p16 and p14, which are responsible for cell cycle regulation and degradation of p53 respectively. p16 is often inactivated in HNSCC through homozygous deletion or by promoter methylation (11). Loss of the 3p chromosomal region is also a common early event in squamous dysplasia and HNSCC, although uncertainty remains about the exact target of the deletion (12). Amplification of 11q13 and over-expression of cyclin D1 have been found in up to 30% of cases of squamous dysplasia and HNSCC, and may correlate with more aggressive tumour behaviour (13, 14). LOH of 17p and specific point mutations of the tumour suppressor gene TP53 are observed in approximately 50% of HNSCC, and seem to occur later in the progression from severe dysplasia to invasive carcinoma (15). Over 90% of cases of HNSCC over-express the epidermal growth factor receptor (EGFR) (16), which correlates with a poorer outcome for patients with HNSCC (17) (see below).
Nasopharyngeal carcinoma (NPC)
Nasopharyngeal carcinoma (NPC) is a human malignancy derived from the epithelium of the nasopharyngeal cavity. It is one of the most striking examples of a human malignancy that is consistently associated with a virus. The Epstein-Barr virus (EBV) genome is contained in all malignant NPC cells and it encodes viral proteins that contribute to the malignant phenotype (18, 19). NPC is a disease with a remarkable geographical and racial distribution. In the year 2000, over 80% of new cases registered worldwide were reported from Southeast Asia, with the highest incident rate found in Southern China (25–30 cases per 100,000 persons per year), South-East Asia, and North Africa. NPC occurs relatively infrequently in Western areas. In these endemic areas implicated factors in the pathogenesis of NPC include a diet from a young age of preserved meats and Cantonese-style salted fish known to contain carcinogenic nitrosamines. Other environmental factors associated with the developed of NPC include smoking, alcohol and Chinese herbal medication.
NPC is an epithelial neoplasm arising from the fossa of Rosenmuller of the postnasal space. It can then extend within or out of the nasopharynx to the other lateral wall and/or posterosuperiorly to the base of the skull or the palate, nasal cavity or oropharynx; it then typically metastases to cervical lymph nodes. Distant metastases may occur in bone, lung, mediastinum and, more rarely, the liver. Approximately 30% to 40% of the patients with advanced stage NPC develop local recurrences and metastatic disease. The prognosis for patients with metastatic disease is poor, with a median survival of less than 12 months.
Cervical lymphadenopathy is the initial presentation in many patients, and the diagnosis of NPC is often made by lymph node biopsy. Symptoms related to the primary tumour include trismus, pain, otitis media, nasal regurgitation due to paresis of the soft palate, hearing loss and cranial nerve palsies. Metastatic spread may result in bone pain or organ dysfunction.
The significance of differentiation
NPC presents with varying degrees of differentiation and has been classified by the World Health Organization (WHO) into three histological types: squamous cell carcinomas (type 1), are highly differentiated with characteristic epithelial growth patterns and keratin filaments, non-keratinising carcinomas (type 2) which retain epithelial cell shape and growth patterns, and undifferentiated carcinomas (type 3) which do not produce keratin and lack a distinctive growth pattern (20). Types 2 and 3 are clearly associated with Epstein-Barr virus (EBV), whereas virus involvement in type 1 is more controversial.
Current treatments and unmet needs
The prognosis of the patient depends greatly on the staging of the disease. Early diagnosis is crucial to improve patient survival. However NPC often presents with minimal or non-specific local symptoms and the nasopharynx is relatively inaccessible to routine examination. These factors represent challenges in making an early diagnosis. In view of the large population at risk of developing NPC in the Asian region, a cost-beneficial screening program is needed. Given the strong association of NPC with EBV, a serological test for EBV-associated antibodies was advocated to be a screening test (21).
Standard treatments for patients with nasopharyngeal cancer include radiation therapy, chemotherapy and surgery. High-dose radiation therapy with chemotherapy is the primary treatment of nasopharyngeal cancer, both for the primary tumour site and the neck (22). When feasible, surgery is usually reserved for nodes that fail to regress after radiation therapy or for nodes that reappear following clinical complete response. Radiation therapy dose and field margins are individually tailored to the location and size of the primary tumour and lymph nodes (23, 24). Although most tumours are treated with external-beam radiation therapy (EBRT) exclusively, in some tumours radiation therapy may be boosted with intracavitary or interstitial implants or by the use of stereotactic radiosurgery when clinical expertise is available, and the anatomy is suitable (25, 26). Although NPC is highly sensitive to radiotherapy and chemotherapy, relapse often occurs.
At present treatment of HNSCC involves surgical excision or radiotherapy (or a combination of both) with or without chemotherapy but as expected most therapeutic options in this region of the body carry significant morbidity and often affect speech, chewing, swallowing and breathing. Despite advances in surgical techniques and adjuvant therapies (which have improved outcomes and increased organ preservation for some sub-sites) the overall average five year survival from HNSCC has remained stubbornly fixed at approximately 50% for the past 40 years. Several recent reports have indicated that a new optical technique called narrow-band imaging (NBI) endoscopy with magnification is able to improve the detection of some types of HNSCC, in particular superficial pharyngeal cancer (27, 28). NBI is based on limiting the depth of light penetration into the mucosa. The recognition of the HNSCC lesions can be dramatically improved. NBI enables a more accurate diagnosis and may increase the detection rate of HNSCC.
There is an urgent need for clinicians to be able to more accurately predict how various tumours will behave so that treatment can be tailored for each patient. Recent advances in molecular technology, including gene expression and proteomic profiling, may elucidate novel tumour markers or signatures that will enable this goal. Further, identification of novel biomarkers may lead to development of specific treatments for these cancers, as evidenced in other cancers with targeted therapies.
Markers for Diagnosis of Cancers of the Head and Neck
The development of microarray technology has allowed gene expression profiling at the RNA level to be conducted for nearly all expressed genes simultaneously by hybridising an array of known sequences with labelled cRNA reverse transcribed from the sample RNA. Expression profiling using microarrays was originally used for cancer classification (29–31), and progressed to identification of distinct clinical outcomes based on profile (32). The eventual aim is to develop personalised treatments based on the patient's specific tumour profile. Other DNA microarrays designed to examine regions of chromosomal amplification or deletion, or chromosomal methylation are also widely used. Whole genome expression profiling using microarray platforms is now well established and has been used to investigate a range of different tumours for biomarkers that may have prognostic or therapeutic significance.
Discovering novel markers in HNSCC
A large effort by many groups has been made to identify novel tumour markers in HNSCC using gene expression profiling. Many of the initial studies described global changes in gene transcription that distinguished normal head and neck squamous epithelia from carcinoma (see Table 2). We previously studied the common alterations in the transition from mucosa to primary tumour using matched autologous tissues from 7 patients across over thirteen thousand genes (33). We found over 1,200 gene products showing statistical significant differences in expression in the transition from normal mucosa to the primary tumour. A number of other groups have performed similar experiments (34–36), with most studies identifying similarly up- or down-regulated genes in their data sets (37).
Expression profiling studies have demonstrated grouping of transcriptional profiles that distinguished preneoplastic versus cancerous epithelium (36). Patients with verrucous leukoplakia and erythroplakia, both premalignant conditions, were found to share a higher degree of relatedness to oral SCC samples than to normal controls (36). Other studies have also shown that most of the transcriptional alterations in HNSCC occur during the transition from normal mucosa to premalignant lesions rather than in the transition from premalignant to invasive carcinoma (38). These changes in gene expression occurring before the development of malignancy may represent good candidates for development as biomarkers to detect very early-stage lesions.
HNSCC salivary test development
Clinical examination and histopathological analysis of biopsy material are the currently used diagnostic methods for determining if a lesion is cancerous. However, these methods are not ideal for a number of reasons. Firstly, the taking of biopsies can be an invasive procedure, possibly involving surgery depending on the site of the lesion in question. Small biopsies may not provide sufficient material for an accurate diagnosis, while on the other hand biopsy of large lesions may not give an accurate histopathological picture due to tumour heterogeneity. For these reasons, development of additional diagnostic methodologies is important.
Significant interest has been generated by the promise of “lab-on-a-chip” type technologies in the diagnosis of cancers, including those of the head and neck (39, 40). This technology refers to the miniaturization and automation of laboratory procedures into a card or “chip”-like format (39, 40). Systems have been already devised to perform cell lysis, PCR and sizing of the resulting product (41), and even microarray analysis (42). While debate continues as to the method or species to be detected in such a device, it is envisaged that the patient would merely provide a sample of saliva that would be put into the chip. The device would then detect either specific cancerous cells present in the saliva based on known biomarkers, a series of proteins present in the sample, or process the cells and profile those present using microarray analysis. While an exciting approach, the success and further development of the “lab-on-a-chip” technology for HNSCC diagnosis will depend on the identification of a series of robust biomarkers.
Markers for diagnosis of NPC
NPC patients sustain higher levels of serum EBV-specific antibodies, such as early antigen (EA) IgA, viral capsid antigen (VCA) IgA, nuclear antigen 1 (EBNA 1) IgA and transactivator ZEB virus replication activator protein (EB1or Zta) IgA, than healthy carriers and patients with other head and neck cancers (43). First described by Werner Henle, this characteristic feature had been widely applied since in serology diagnosis of suspected NPC (see Table 1). In this approach, an increase serum level of VCA IgA antibody supports clinical suspicion of the disease and provides an additional indication for further investigation by endoscopy and, if so indicated, histopathology to confirm diagnosis of the cancer. Especially for patients with early disease, when symptoms are often non-specific and innocuous, this approach can facilitate early treatment and thereby better ensure survival (44). Previous population studies also associate a high level of EBV-specific antibody in an otherwise healthy subject with an increased incidence of NPC. EBV serology is therefore included as part of screening programs in some areas with high incidence of the cancer (45, 46).
Possible markers for use in NPC diagnostic, screening and prognosis.
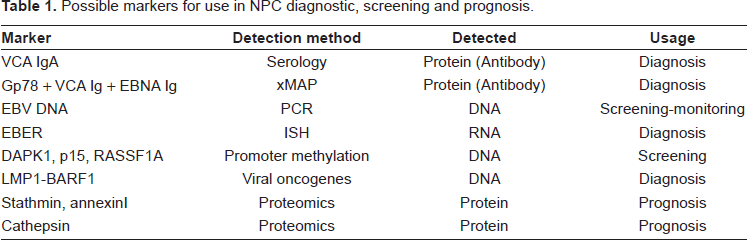
EBV-specific antibodies such as the anti-viral capsid antigen IgA(IgA-VCA) are traditionally determined by immunofluorescence (47) or immunoenzymatic methods (48) against EBV-transformed lymphoblastoid cell lines. Objective enzyme immunoassays (ELISA) specific for individual EBV-specific peptides are available. Most of these assays afford comparable specificity and sensitivity for serologic diagnosis of NPC as do the traditional methods. Moreover, increasing availability of these assays with distinct antigenic specificity makes it possible to use the assays in combination in an approach to effect diagnosis of NPC based on EBV antibody spectrum rather than the level of individual antibodies. Reports by Gu et al. showed that IgA and IgG antibodies against gp78, a membrane protein, were statistically higher in NPC populations than in healthy controls (49). They suggested the use of Luminex multi-analyte profiling (xMAP) technology to detect multiple parameters in NPC, such as antibodies against gp78, VCA, EA-D and EBNA1 simultaneously (see Table 1). Meanwhile, Ji et al. found a sustained elevation of EBV antibody levels preceding clinical onset of NPC and suggest that effective control of NPC can be achieved by repeating serologic screening every two years to identify cases as they enter the serologic window (50).
Serological EBV antibody screening of EBV has been practiced for many years. However, it has the problem of either low specificity or low sensitivity. Furthermore, the antibody titer remains persistently high in most patients in remission after treatment (51). There is no reliable cutoff value in the differential diagnosis of recurrence or remission.
Cell-free circulating DNA is an emerging source of biomarkers for cancer screening and treatment response monitoring. Elevated plasma DNA concentration has been reported in patients with epithelial malignancies including head and neck carcinomas (52, 53). A few years ago, quantitative plasma EBV DNA analysis was shown to be a sensitive molecular marker for NPC as it directly measures tumour-derived EBV genomic material rather than an antibody response to genomic or peptidic components of the EBV (54). It was associated with disease staging and shown to be useful for monitoring of disease progression. Fan et al. reported elevated serum EBV levels in progressive disease (late stage and relapse) while undetected in remission, suggesting that viral load is related to tumour burden and/or to the presence of metastases (51). The quantification of EBV DNA in plasma or serum can be of great clinical value and can assess the efficacy of new treatments. The detection of EBV encoded RNAs (EBERs) by in situ hybridization (ISH) in tumour cells can also be used as a diagnostic marker (see Table 1).
EBV DNA is also present in most EBV-associated benign or malignant diseases apart from NPC, including infectious mononucleosis, EBV-associated hematological malignancies, chronic active EBV infection, EBV-associated hemophagocytic syndrome and transplantation-related lymphoproliferative disease. Since the serological EBV antibody screening has low specificity, in the absence of a reliable EBV antibody or DNA biomarker with both high sensitivity and high specificity, a second biomarker or combination of several markers has to be considered to allow better selection of patients for additional expensive and invasive nasoendoscopy and biopsy procedures or to exclude the presence of primary NPC.
An alternative approach is to detect tumour markers in samples collected directly from the nasopharyngeal (NP) region via a non-invasive procedure, such as NP brushing or swab. Tong et al. propose to use the detection of EBV DNA from NP brushing/swab samples as a useful tumour marker for NPC and combine it with the detection of promoter hypermethylation of tumour-specific genes (55). Whereas, Leung et al. suggested the combined detection of EBV DNA and IgA-VCA to increase the sensitivity and specificity for NPC diagnosis and screening (56).
Promoter methylation of many tumour suppressor genes is found frequently in cancer cells but is normally absent in normal cells (57). This level of specificity is one of the desirable requirements of tumour biomarkers for cancer screening. The plasma DAPK1, p15, and RASSF1A-methylated DNA had high specificity and were found mostly in NPC patients but not in normal controls (58). In comparison with the serological EBV antibody or EBV DNA alone, the use of a combination of serological EBV antibody and the panel of methylation markers has markedly increased the overall diagnostic accuracy in both detection and exclusion of NPC. Recently a study by Houali et al. (59) suggested to use the detection of the viral oncogenes LMP1 and BARF1 proteins in serum as an alternative diagnostic test for NPC in particular for young patients (see Table 1).
Relapse of disease remains one of the biggest problems in clinical management of cancer. In NPC, more than half of the patients with complete response to radiation therapy developed relapse after further follow up. By the time distant relapse is diagnosed clinically, it is usually hard to manage. Therefore, finding biomarkers to diagnose relapse early is an important undertaking in translational cancer research. With this in mind, Cho et al. analysed the serum proteins components from NPC patients by protein chip profiling to identify potential biomarkers and reported that two isoforms of serum amyloid A strongly correlated with the clinical relapse status in NPC patients (60).
Prognostic Markers for the Management of Patients
Discovery, validation and prospects for routine use in HNSCC
More recent research has focused on the elucidation of gene expression profiles in HNSCC distinguishing metastatic from non-metastatic disease. Tumours of the oropharynx, hypopharynx, and larynx were found to group significantly according to metastatic cervical lymph node status (61). A study evaluated the gene expression profiles of 34 hypopharyngeal tumour specimens and identified a subset of 164 genes that were associated with metastatic potential, as indicated by patients with or without clinical evidence of metastasis 3 years after surgery (62). Others have identified a 116 gene signature set that differentiated primary tumour specimens according to metastatic lymph node status, and showed that tumour specimens from lymph node metastases were similar to lymph node-positive primaries (63). These authors went on to use the identified gene signature to “predict” the presence of lymph node metastases in a number of patients that were not included in the original data analysis.
A recent series of studies by Roepman and colleagues has expanded on the metastasis predictor gene expression signature in HNSCC. These authors examined expression profiles from 82 HNSCC tumour specimens (45 metastatic and 37 non-metastatic) of the oral cavity and oropharynx and established a predictor set of 102 genes that was associated with metastasis. The performance of this predictor set was dependent on tumour tissue specimen storage times, exhibiting improving performance with shorter storage times. When the predictor set was assessed among expression profiles of 22 independent tumour samples, all stored for less than 5 years, lymph node status was correctly predicted in 86.4% of the tissue specimens (64). Further analysis has shown that this initial gene set is part of a larger group of 825 genes (65), with the suggestion that larger gene sets lead to more accurate predictions and are less prone to false negative calls. These findings taken together, suggest that there might be a metastatic gene expression signature present in some primary tumours that predisposes them to metastasize. However, not all studies agree, with some not finding any metastasis-specific gene expression signature (66). Clearly, further validation studies are needed in this area.
A great deal of research has also been conducted into attempting to correlate gene expression profiles from HNSCC tumours with patient clinical outcomes. In an excellent study, Chung and co-authors identified genes signatures from tumours that clustered into four groups, which exhibited significantly different rates of disease recurrence-free survival (61). Others have examined over 50 specimens from multiple sites and identified a set of genes with altered expression that grouped patients according to tumour recurrence, and therefore worse outcome (67). Differences in gene expression profiles have been found between HPV-positive and HPV-negative tumours (68–70). As discussed above, patients with HPV-positive or -negative tumours have a different prognosis. Recent studies from our laboratories have shown that elevated protein expression of osteonectin and alpha-B crystallin, the product of the CRYAB gene, were independent predictors for short disease-free interval and poor overall survival in a group of 62 patients (33, 71). These findings are currently being validated in a larger group of patients.
Some of the markers identified in these studies have the potential to become routinely used tumour markers. A simple test for a small number of markers would be technically easier and probably more widely used. Ideally, it would be possible to detect some or all of these changes by a simple biopsy or even a blood test. Determining the pattern of alteration in these biomarkers may aid in diagnosis, help predict prognosis and indicate which treatment modality would be appropriate for patients. However, many of the genes identified by the various studies are not well characterised, and need to be studied functionally. Significant validation work is now required to correlate the changes in expression pattern with clinical outcome.
Genetic polymorphisms and HNSCC
The completion of the sequencing of the human genome has allowed study of inherited factors such as genetic polymorphisms, as potential predictive and prognostic factors in a number of cancers. Alongside clinical and tumour molecular prognostic factors, genetic polymorphisms may play key roles by increasing the accuracy and validity of outcome prediction models. Polymorphisms in Glutathione S-transferase (GST) genes GSTM1, GSTT1, GSTP1, the DNA repair X-ray repair cross- complementing (XRCC)1, and the cytochrome P450 gene CYP1A1 were recently examined by meta-analyses (72). Significant statistical relationships were found between the GSTM1-null genotype and an increased risk for HNSCC (72). In addition, increased risk for HNSCC was associated consistently with the ALDH2*1/*2, p53 (codon 72 Pro/Pro) and epoxide hydrolase EPHX1 (codon 113 Tyr/His and His/His) genotypes. Other meta-analyses have linked polymorphisms in XRCC1, FGFR, and CCND1 with HNSCC survival (73). Further studies in well characterised patient cohorts are warranted.
EBV screening in serum for NPC recurrence
A clinically useful tumour marker should not only have higher sensitivity and specificity in the diagnosis setting but also have a significant impact on the prognosis prediction. The latter is more important than sensitivity and specificity calculations alone in clinical practice. By measuring antibodies to an EBV-induced membrane antigen component, Neel et al. demonstrated that patients with low titers at diagnosis had significantly worse progression-free survival and overall survival for types II and III NPC patients. Conventional antibody (VCA-IgG, EA-IgA and EA-IgG) assays before treatment could not reflect the prognosis (74). After long-term follow- up, they also found that the post treatment sequential measurements did not accurately predict the outcome (75).
A multi-centre follow-up study on 319 NPC patients showed no prognostic values of initial serology, including IgG and IgA antibodies to VCA, EA or EBNA. However, increasing titers of EA-IgA and EA-IgG 1 year after completion of radiotherapy became highly significant for prediction of relapse, regardless of the initial titers (76). The definite role of anti-EBV antibodies on prognosis prediction is still controversial and the precise point of blood sampling has not been well established. On the contrary, circulating EBV DNA loads measured before or after treatment are consistently correlated with the survival and prognosis of NPC patients (53, 54). This may relate to the essential differences between a marker directly released from tumour cells (EBV DNA) and a marker that essentially is a host response to viral tumour antigens. The latter marker inevitably lags behind with regard to kinetics upon treatment. Also, the dynamic range of antibody responses is fairly limited in general, with only weak correlations observed between the extent of exposure and the level (titer) of the response. The major finding is that EBV DNA assay is superior to the anti EBV antibody assay in both sensitivity/specificity calculation (56, 77), and differential diagnosis of active disease or remission state (51). In addition, plasma EBV DNA levels have better correlation with clinical staging (78) and survival curves (79) than VCA-IgA titers.
Use of Biomarkers in the Development of Novel Therapies
EGFR and angiogenesis inhibitors in HNSCC therapy
Studies have shown that over 90% of cases of HNSCC over-express the epidermal growth factor receptor (EGFR) (16), which correlates with a poorer outcome for patients with HNSCC (17), (see Table 2) and a resistance to therapy (80). As activation of EGFR is associated with tumour growth and progression, anti-EGFR agents have been developed for use in cancers including HNSCC. Cetuximab, a monoclonal antibody targeting the extracellular domain of EGFR, is the first molecularly targeted agent introduced into standard practice for treatment for HNSCC (81). Combination of cetuximab with standard radiotherapy or chemotherapy has also yielded very promising results (82). Vascular endothelial growth factor (VEGF), a molecule crucial in angiogenesis, is also associated with patient prognosis in HNSCC (83). Molecules that target angiogenesis, including bevacizumab (a monoclonal antibody to VEGF) and sorafenib or sunitinib (multitarget receptor tyrosine kinase inhibitors), are currently under investigation in phase II studies in HNSCC, with promising initial results (82). These examples of molecular targeted therapies elucidated from biomarker research are already finding a place in the treatment of HNSCC patients. Novel molecular targets will continue to emerge with further studies, as hopefully will specific corresponding therapies.
Possible markers for use in HNSCC diagnosis, screening, prognosis or treatment.
Use of biomarkers in the development of novel therapies for NPC
Although NPC is classified as a subtype of head and neck squamous cell carcinoma, its epidemiology, clinical characteristics, etiology and histopathology are unique. For example, compare with other head and neck squamous cell carcinomas, NPC tends to present at a more advanced stage of disease because of its deep location and vague symptoms, exhibits higher metastatic potential, and its histologic grading related to radiosensitivity is more confusing. Furthermore, the survival rate for patients with NPC has remained unchanged in the past few years despite advances in diagnosis and treatment. Therefore, early diagnosis, exact histologic grading, and accurate prognostication of NPC are critical for guiding the treatment and improving the prognosis of NPC.
The finding of NPC biomarkers may be a convenient way to achieve these objectives. With the advances in molecular biology in the past decades, a lot of NPC-related molecules, such as serum antibodies against various EBV proteins, serum amyloid A, Bmi-1 and Met protein have been reported as potential biomarkers for the diagnosis, prognosis, or treatment of NPC. However they are of limited clinical value because of low sensitivity or specificity. At present the mainstay for the diagnosis and histologic grading of NPC is endoscopic examination and histologic observation of biopsies, and tumour-node metastasis staging is still the main method of predicting the prognosis of NPC. Therefore it is urgent to discover more effective biomarkers and therapeutic targets for NPC.
As the functional molecules in cells are proteins, proteome analysis may have an advantage over cDNA microarray for clinical use. Differential proteome analysis of tumour and normal tissue allows the identification of aberrantly expressed proteins in cancer that might provide key information for finding biomarkers for the diagnosis and treatment of cancer as well as understanding of carcinogenesis.
High-throughput technologies such as microarrays and proteomics offer the potential ability to find alterations previously unidentified in NPC. Proteomics has introduced a new approach to cancer research which aims at identifying differential expression proteins associated with the development and progression of cancer, providing new opportunities to uncover biomarkers and therapeutic targets for NPC as well as reveal the molecular mechanism underlying this disease. Using tissue samples from patients may be the most direct and persuasive way to find biomarkers and therapeutic targets for cancer by a proteomic approach. A major obstacle, however to the analysis of tumour specimens is tissue heterogenicity, which is particularly relevant to NPC as it often includes numerous infiltrating lymphocytes and stroma. Moreover, normal nasopharyngeal epithelial cells, from which the cancer is believe to arise, represent as little as 10% of nasopharyngeal mucosal tissue. Recently Cheng et al. used laser capture microdissection, to isolate pure cell populations from heterogeneous tissue, and proteomic analysis to screen for proteins associated with cancer and identify stathmin, 14-3-3 σ, and annexin I as potential NPC biomarkers (84). These three proteins were related to the differentiation and/or metastatic potential of NPC cell lines and their expression levels significantly correlated with several clinico-pathologic variables and prognosis of NPC. In another recent report, they recommended the use of cathepsin D (lysosomal hydrolase) as a biomarker for metastasis and prognosis of NPC (85) (see Table 1).
Perspectives
One of the major criticisms of studies attempting to identify biomarkers to date, particularly those attempting to correlate or predict patient outcome, has been the lack of overlap of predicting molecules between like studies. It is likely that the variation in tumour specimen characteristics could significantly impact this. With the development of more standardized platforms for sample preparation and data analysis, it is generally considered that these limitations will be overcome. Further, many have criticized the small patient numbers involved in these early studies. Clearly, larger studies of much larger sample sizes comprising tumour specimens of more uniform characteristics need to be undertaken. It is also crucial that any pattern or gene difference from expression profiling analysis be validated in an independent sample series to ensure the robust nature of the finding. Even with these current drawbacks, it remains possible to hope that some of the markers or patterns of markers identified in these studies could, in the future, be used to detect the presence of HNSCC, metastasis of the cancer, or aid in determining the best treatment for the patient. There is good reason for optimism, with the development and efficacy of EGFR and angiogenesis inhibitors in HNSCC, as well as the use of EBV DNA in patient management in NPC.
Disclosure
The authors report no conflicts of interest.
