Abstract
Epidemiological evidence has established a link among hyperlipidemia, visceral obesity, osteoporosis, and cardiovascular diseases. We have recently proposed a hypothesis that the associations of those disorders are based on interactions of the three organs, i.e. the bone, adipose, and vascular tissues, possibly through multiple actions of several humoral factors and/or transcription factors. We call this unified hypothesis “osteo-lipo-vascular interactions”, which may be explained by the common origin of the cells in each organ, such as mesenchymal stem cells or macrophages. Several groups proposed similar hypotheses. On the other hand, there have been accumulating evidences which indicate that there exist hitherto unknown various interactions between many organs, such as hypothalamus-liver, fat-liver, liver-muscle, intestine-pancreas, kidney-heart and so on. Therefore, it seems insufficient to consider only the interactions among several organs, and the standpoint of considering interactions among all organs may be warranted, especially in order to understand the pathogenesis of metabolic syndrome, insulin resistance and type 2 diabetes. We here propose a hypothesis that the abnormal interactions of all organs (“Ominous Orchestra of Organs”) underlies the pathogenesis of type 2 diabetes. It is to be elucidated which of the “players” or the “conductor” may be mainly responsible for disharmony of the orchestra.
‘Osteo-lipo-vascular Interactions’
Epidemiological evidence has established a link among hyperlipidemia, visceral obesity, osteoporosis, and cardiovascular diseases (CVD), suggesting some interactions between bone, adipose tissue and vessels. We have recently proposed the hypothesis that the associations of those disorders are based on interactions of the three organs, i.e. the bone, fat, and vascular tissues [1], [2]. We have called this unified hypothesis ‘osteo-lipo-vascular interactions’ [3], [4] (Fig. 1). There are several possible mechanisms to explain those interactions.
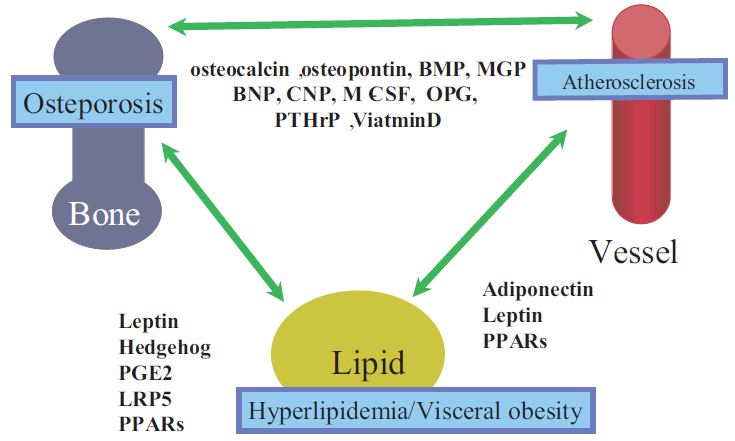
First, these interactions can be attributable to actions of several humoral factors, i.e. hormone in a broad sense. Second, it can also be explained by transcription factors, which are shared by several of the three organs. Third, from the viewpoint of developmental physiology, the fact that there exist interactions between the three organs may be fully reasonable, since the three organs share the cells of common origin, i.e. mesenchymal stem cells (MSCs); the osteoblasts in the bone, the vascular smooth muscle cells (VSMCs) in the vessel, and the adipocytes in the fat (Fig. 2). Fourth, the interactions can also be attributable to the function of macrophages, which evolve into the osteoclasts in the bone, evolve into the foam cells in the vessel, and infiltrate into the fat, causing inflammation of the adipocytes [4], [5], [6] (Fig. 3).


Other researchers have proposed several hypotheses based on similar concept [7], [8], [9]. Burnett and Vasikaran suggested a link between lipids and bone [7]. Hamerman has suggested that osteoporosis and atherosclerosis have biological linkages and that the dual-purpose therapies against osteoporosis and atherosclerosis are hopeful [8]. Pimanda et al. have stated that blood and vascular cells are generated during embryogenesis from a common precursor, and that there are transcriptional linkage between blood and bone [9]. Furthermore, the concept of metabolic syndrome [10] itself is partly based upon the interactions between adipose tissue and vessels, although its clinical usefulness is very controversial [11], [12].
Other Interactions among the Organs
Interactions between the hypothalamus and other organs
Recently, several studies have indicated that there exists some interactions between the peripheral organs and the brain, especially hypothalamus, with regard to glucose metabolism.
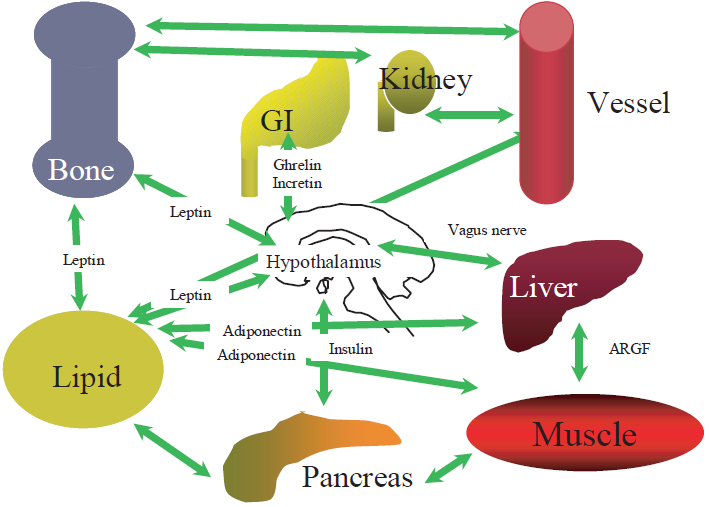
First, it has been revealed that there are several common components to pancreas and liver, such as adenosine triphosphate (ATP)-sensitive potassium channels (KATP channels), malonyl-CoA, and glucokinase [13]–,,[16]. Those molecules are considered to consist the hypothalamic glucose-sensing system, which probably acts as a defense mechanism against hypoglycemia. The activation of KATP channel causes an inhibition of hepatic gluconeogenesis through vagal nerve stimulation, thereby forming a brain-liver circuit [17]. On the other hand, expression of peroxisome proliferator-activated receptor-gamma 2 (PPAR–γ2) in the liver induces a decrease in adiposity, which is transmitted by the pathway from the liver to the brain [18]. Therefore, it is indicated that there are bidirectional pathways between the hypothalamus and the liver.
Second, the hypothalamus is indicated to receive adiposity signals, mainly of insulin and leptin. Recently there have been several experimental evidences indicating that hypothalamic resistance to insulin or to letpin may play a significant role in metabolic syndrome and/or diabetes mellitus [19]. Third, it is plausible that glucocorticoid hyperactivity in the hypothalamo-pituitary-adrenal axis (HPA axis) may play a role in the pathogenesis of metabolic syndrome and/or type 2 diabetes [20]. Finally, very recently, the dysfunction of clock genes including those of hypothalamus has been indicated to be involved in the pathogenesis of metabolic syndrome [21], [22]. Based on those backgrounds, we have proposed a hypothesis that hypothalamic abnormality may be primary pathogenetic mechanism for, at least, some portion of type 2 diabetes [23].
Other interactions
There are several interactions between other organs.
First, there are several evidences indicating interactions between liver and adipose tissues. For example, angiopoietin-related growth factor (ARGF), which is secreted from the liver, affects the function of adipose tissues [24]. Conversely, some “adipo(cyto)kines”, secreted from adipocytes, are involved in hepatic insulin resistance [10]. Furthermore, adipose-specific overexpression of GLUT4 reverses insulin resistance and diabetes in mice lacking GLUT4 selectively in muscle [25]. This result could not be explained by known adipokines, such as adiponectin, or resistin.
Second, the link between brain and gastrointestinal system has been known, which is mainly through peptides, formerly known as ‘brain-gut hormones’ [26]. Ghrelin, a recently discovered hormone secreted from the stomach, is shown to stimulate feeding center of hypothalamus.
Recently, two hormones, gastric inhibitory polypeptide (GIP) and glucagon-like peptide 1 (GLP-1), have been established as ‘incretins’, defined as hormones which are secreted from intestine and stimulate insulin secretion from the pancreas [27], [28].
The concept of chronic kidney disease (CKD) [29] has recently been advocated mainly because of the fact that CKD is frequently associated with CVD based on the interaction of kidney and cardiovascular system. In this context, CKD is alternatively expressed as “cardioneral syndrome” [30]. In addition, another concept, “CKD-mineral-bone disorder (CKD-MBD)”, is based upon the interaction between kidney and bone [31].
Concept of ‘Ominous Orchestra of Organs’
DeFronzo has very recently proposed the term of “the dysharmonious quartet”, which designates a decreased insulin secretion from the pancreas, insulin resistance of the liver, dysfunction of the adipose tissue, and insulin resistance of the muscle [32]. He has further listed several rhetoric concepts such as “quintessential quintet”, “setaceous sextet”, and “septicidal septet”, which designate “the dysharmonious quartet” plus a decreased action of incretin secreted from the intestines, plus an increased secretion of glucagon from the pancreas, and plus the dysfunction of the hypothalamus, respectively [32]. As for the rhetoric expression of the medical concept using the musical term, “the deadly quartet” is well known, i.e. the combination of glucose intolerance, hypertriglyceridemia, hypertension and upper body obesity, proposed by Kalpan [33], which is intended to designate the clustering phenotypes found in high-risk individuals for type 2 diabetes and/or atherosclerosis, contributing to the establishment of the concept of metabolic syndrome.
However, as described above, there have been increasing evidences which indicate that there are many interactions among all of the organs of human body. Therefore, it dose not seem reasonable to limit the components, that is to say, “the players” to several of the organs. Here we propose a new hypothesis of “Ominous Orchestra of the Organs”, which suggests that the dysfunction of any part of these interactions may underlie the pathogenesis of metabolic syndrome, insulin resistance and type 2 diabetes (Fig. 4). Furthermore, there are also several hypotheses, which may highlight the role of intracellular organelles such as mitochondria or endoplasmic reticulum in the pathogenesis of type 2 diabetes or insulin resistance [34], [35]. According to our rhetoric expression of “ominous orchestra”, those hypotheses can be expressed as ones based upon an abnormality of “some part of each musical instrument”.
Future perspective
Our hypothesis of “ominous orchestra of the organs” may provide a new insight into the pathogenesis of metabolic syndrome, insulin resistance and type 2 diabetes. For that purpose, this hypothesis should constitute a novel system taking into the consideration both proximate and ultimate causes, as others have recently proposed as behavioral switch hypothesis [36] or revised thrifty genotype hypothesis [37]. The integrated system of human body may work synchronously through the various interactions as a defense system against the external environmental changes in ancient times, but some dysfuncrtional interaction of the system may result in disorders such as metabolic syndrome, insulin resistance and type 2 diabetes.
Regarding the pathogenesis of type 2 diabetes, it is to be elucidated which of the “player(s)” or alternatively the “conductor” may be mainly responsible for disharmony of the orchestra. Our “hypothalamic hypothesis of type 2 diabetes” [23] can be viewed as a hypothesis indicating the hypothalamus as the “conductor”, who is responsible for abnormality in type 2 diabetes [15, 16, 17, 18, 19].
On the other hand, the concept of metabolic syndrome has been based on the assumption that the “conductor” may be visceral obesity [10], although some researchers argue against that assumption, including the American Diabetes Association and the European Association for the Study of Diabetes [11], [12].
The concept of “ominous orchestra of the organs” may also provide us the basis of new drug development. For example, exenatide, an incretin (GLP-1) analog, which has been already applied for clinical use to treat type 2 diabetes in the United States, is indicated to act on hypothalamus to decrease food intake, indicating its another clinical benefit to decrease body weight in diabetic subjects [28]. Future development of novel drugs may be justified from the standpoint of trying to discover agents which may improve interactions between several organs in order to prevent or treat metabolic syndrome, insulin resistance and type 2 diabetes.
Footnotes
Acknowledgments
We thank Dr. Yasuhiro Murakawa (Kyoto University Faculty of Medicine) for his critical reading this manuscript.
