Abstract
Background
The association between type 2 diabetes mellitus (T2DM) and low total serum testosterone (LST) has been identified in several cross-sectional studies.
Objectives
To assess the prevalence of androgen deficiency and erectile dysfunction (ED) and their relation to glycemic control within a sample of Egyptian men with T2DM.
Research Design and Methods
A cross-sectional study including 70 men having T2DM. Their ages ranged from 30 to 50 years. They were evaluated for symptoms of androgen deficiency and ED, using a validated Arabic-translated Androgen Deficiency in Aging Males questionnaire and five-items version of the International Index of Erectile Function-5, respectively. Total testosterone (TT), glycated hemoglobin (HbA1c), follicle-stimulating hormone (FSH), luteinizing hormone (LH), and prolactin were measured for all study subjects. Penile hemodynamics was assessed using penile duplex study for subjects who gave history of ED.
Results
LST was found in 40% of studied men, and 92.9% of them reported overt symptoms of androgen deficiency. ED was detected in 85.7% of those with LST, as opposed to 31.0% of those with normal TT (P < 0.000). TT was lower in diabetic men with ED compared to those without ED (12.04 ± 5.36 vs 17.11 ± 7.11 nmol/L, P < 0.001). Significant negative correlation was found between TT and age, body mass index, waist circumference, systolic and diastolic blood pressures, and HBA1c (P < 0.00). FSH, LH, and prolactin levels were within the normal reference range in all subjects. HbA1c was higher in patients who had LST with ED, compared to those with normal TT and without ED. However, multivariate logistic regression analysis did not reveal a significant association between HBA1c and LST levels.
Conclusion
LST, symptoms of androgen deficiency, and ED are common in the studied sample of Egyptian men with T2DM. Inappropriately normal FSH and LH in face of LST may denote a state of hypogonadotropic hypogonadism. HBA1c was found to be more significantly associated with ED than with LST.
Introduction
Male androgen deficiency is a global health issue. 1 The association between low serum testosterone (LST) and type 2 diabetes mellitus (T2DM) has recently drawn substantial attention.2,3 Epidemiological studies have reported that 30%–50% of men with T2DM have testosterone deficiency,4,5 and up to 75% of them have sexual dysfunction, particularly erectile dysfunction (ED). 6
Hypogonadism (HG) is defined as the presence of overt clinical symptoms besides LST. 7 Questionnaires have been developed to assess androgen deficiency. These were found to lack specificity but have reasonable sensitivity in the presence of LST level.8,9 Beyond sexual health, LST is linked to visceral adiposity and insulin resistance (IR), 10 vascular disease, 11 hypertension, and dyslipidemia, 12 as well as osteoporosis. 13 These metabolic derangements are linked to increased morbidity and mortality in patients with LST. 14
There is no clear consensus for an accepted lower limit of normal testosterone. Various guidelines for the diagnosis of HG are available including the International Society for Andrology, The European Association for Urology, and the Endocrine Society Clinical Practice guidelines.15,16 They recommended that patients with total testosterone (TT) <8 nmol/L should be considered hypogonadal and are candidates for testosterone therapy. For those with TT between 8 and 12 nmol/L besides hypogonadal symptoms, a trial of testosterone therapy may be considered, and others with TT >12 nmol/L should not be considered hypogonadal and should not be treated. The guidelines of the Endocrine Society of America recommended that testosterone level <10.4 nmol/L is consistent with HG. 17
ED and HG have recently been recognized as possible predictors of forthcoming metabolic diseases and cardiovascular events. 18 However, it is noted that the attention on male sexuality in diabetes is always centered on ED, while the frequency and relevance of HG are neglected. The present study had been carried out to verify the prevalence of androgen deficiency and ED within a sample of Egyptian men with T2DM and to assess its relation to glycemic control.
Subjects and Methods
Study Design and Population
This was a cross-sectional study. Seventy male patients having T2DM were randomly selected from those attending the diabetes outpatient clinic, Al-Zahraa University Hospital throughout the period from March 2014 to January 2015.
Inclusion Criteria
Male patients (age range, 30–50 years) having T2DM for ≥5 years were included. They should have normal renal and hepatic function.
Exclusion Criteria
Those included history of HG, panhypopituitarism, chronic debilitating disease, history of head or pelvic trauma, surgery or exposure to radiation, as well as history of psychiatric and neurological diseases. Moreover, patients with genital abnormalities or deformities and those on testosterone replacement therapy were excluded from the study.
Ethical Issues
All subjects were informed of the general aim of the study and their participation in the study was fully voluntary. Confidentiality of collected data was guaranteed to participants, and informed consent was obtained from each patient. The study was approved by the Ethical Committee of the Faculty of Medicine for Girls, AL Azhar University. This committee is compliant with the principles of the Declaration of Helsinki.
Methodology
Full demographic and medical history was taken from all subjects including age, smoking, duration of diabetes, type of treatment, and complications.
All patients underwent a complete physical examination including measurement of height, weight, and waist circumference (WC), and body mass index (BMI, kg/m2) was calculated. Blood pressure (BlPr) was also measured (mean of three measurements 5 min apart, in sitting position, with a standard sphygmomanometer). Thorough assessment of peripheral and autonomic diabetic neuropathy was also done. Secondary sexual characters including body hair distribution pattern and gynecomastia were assessed. Local genital examination with the assessment of testicular size and structure of the penis was also done.
All study participants were asked to complete the abridged and the validated Androgen Deficiency in Aging Males questionnaire (ADAMq), including morning erection to exclude psychogenic ED. A positive response denoted the presence of clinical HG based on a decrease in libido, strength of erections, or any three nonspecific questions that may include a decrease in muscle strength, fatigability, mood changes, and loss of height. 9 ED was assessed using the International Index of Erectile Function-5 (IIEF-5) Questionnaire. This addresses the relevant domains of male sexual function, namely, ED, orgasmic function, sexual desire, intercourse satisfaction, and overall satisfaction. A score of >21 denoted no ED while a score ≤21 denoted ED. 19
Blood samples for biochemical analysis was drawn between 8:00 and 10:00 am after an overnight fast. Measurement of TT was done by EIA Kit (EA 78; Oxford Biomedical Research, normal range 8.5–40 nmol/L), follicle-stimulating hormone (FSH) by ST AIA-PACK FSH (cat. no. 002565; normal range 2–14 mIU/mL), luteinizing hormone (LH) by ST AIA-PACK, LH II (cat. no. 0025296; normal range 1–12 mIU/mL), PRL by ST AIA-PACK PRL (cat. no. 0025255; normal range 3–28 ng/mL), and glycated hemoglobin (HBA1c) by B.S 280-Kintic Kit (Mindery, normal range 4–6%).
According to criteria of serum TT levels recommended by the Endocrine Society Clinical Practice Guideline, 16 LST was considered if TT level was <8 nmol/L. Normal level was considered when TT was >12 nmol/L and values between 8 and 12 nmol/L should be considered borderline cases.
Penile color duplex ultrasound (PCDU) had been performed for patients who complained of variable degrees of ED. Details of the procedure were explained to patients prior to examination. High-frequency transducer (7.5–9.0 MHz) was used for penile Doppler examination.
Preinjection measurements included examination in flaccid state to determine structural abnormality or calcified plaques, inner diameter of cavernosal artery (normal value: 0.3–0.5 mm), baseline peak systolic velocity (PSV cm–1sec) and end diastolic velocity (EDV cm–1sec).
Papaverine 60 mg was injected intracavernosally and 10–15 μg of prostaglandin E1 was also injected under sonographic guidance.
Postinjection measurements at 5, 10, 15, and 20 minutes included inner diameter of cavernosal artery (normal value: 0.6–1.0 mm), PSV cm–1sec and EDV cm–1sec. Levels of PSV>30 cm–1sec, EDV of <5 cm–1sec and a resistance index > 0.8, were considered normal. 20
Statistical Analysis
Data were collected, revised, coded, and entered in the statistical package for social science (IBM SPSS) version 20. The qualitative data were presented as numbers and percentages while the quantitative data were presented as means, standard deviations, and ranges. The comparison between two groups with qualitative data was done by using Chi-square test while the comparison between two groups with quantitative data and parametric distribution were done by using independent t-test. Spearman correlation coefficients were used to assess the significance between two quantitative parameters in the same group. Multivariate logistic regression analysis was used to assess the predictors of LST and the presence of ED. The confidence interval was set to 95% and the margin of error accepted was set to 5%. P-value was considered significant at the level of <0.05.
Results
Seventy men, having T2DM, were included in this study. The mean age of the patients was 41.44 ± 6.89 years with a mean duration of diabetes 7.96 ± 3.07 years. Clinical HG (positive response to ADAM q) was found in 58.6% of study subjects. Other demographic and clinical data are shown in Table 1.
Demographic and clinical characteristics of study participants in relation to TT levels.
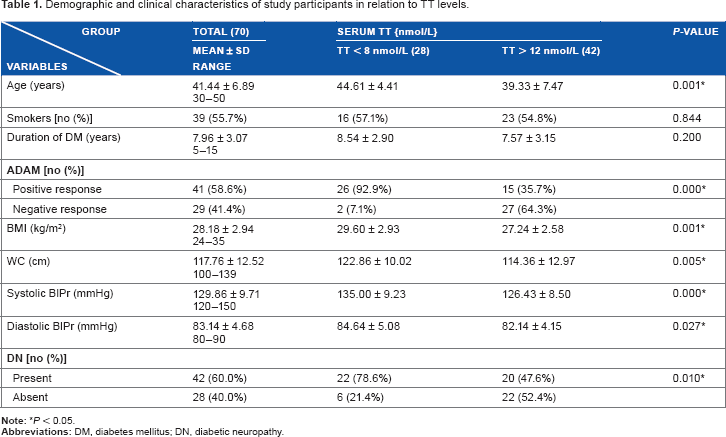
According to morning TT levels, 28 patients (40%) had biochemical HG (TT < 8 nmol/L) and 42 patients (60%) were considered eugonadal (TT > 12 nmol/L). None of our patients had serum TT between 8 and 12 nmol/L. Clinical HG assessed by ADAMq was detected in 24 out of 28 men with LST (92.9%) and in 15 out of 42 men with normal TT (35.7%).
Patients with LST had higher BMI and WC compared to those with normal TT (29.6 ± 2.93 vs 27.24 ± 2.58 and 122.86 ± 10.02 vs 114.36 ± 12.97, respectively). They had higher systolic and diastolic BlPr (135.0 ± 9.23 vs 126.43 ± 8.50, P < 0.00, and 84.64 ± 5.08 vs 82.14 ± 4.15, P < 0.02). DN was found in 78.6% of men with LST compared to 47.6% of those with normal TT (Table 1). Moreover, they had higher HBA1c (9.20 ± 0.74 vs 7.95 ± 0.60, P < 0.00).
No significant difference was found between those with low and normal TT as regards the concentration of FSH, LH, and Prolactin (Table 2).
Biochemical data of study participants in relation to TT levels.

Table 3 displays results of assessment of ED using IIEF-5. Patients with LST had lower IIEF-5 score compared to those with normal TT (15.96 ± 3.38 vs 21.26 ± 2.00, P < 0.00), 85.7% of patients with LST had ED opposed to 31.0% of those with normal TT (P < 0.00). PCDU was done only for patients who presented with ED (24 out of 28 patients with TT < 8 nmol/L, and 13 out of 42 of those with normal TT). Patients with LST had lower levels of PSV and EDV compared to those with normal TT (26.50 ± 4.14 vs 31.31 ± 2.59, P < 0.001) (2.46 ± 0.98 vs 3.38 ± 0.51, P < 0.003).
Parameters of ED in relation to TT levels.

P < 0.05 (significant).
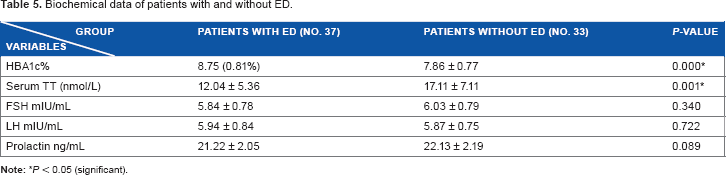
Patients with ED were older than those without ED (44.41 ± 6.15 vs 35.75 ± 4.13, P < 0.00). They had higher BMI and WC (29.31 ± 2.79 vs 26.01 ± 1.81, P < 0.00, and 122.22 ± 11.47 vs 109.21 ± 9.84, P < 0.00, respectively), higher systolic (133.26 ± 9.67, P < 0.00) and diastolic BlPr (85 ± 5.06 vs 80.42 ± 2.04, P < 0.00), and higher percent of DN (89.2% vs 39.4%, P < 0.00) (Table 4). They also had higher HBA1c levels (8.75 ± 0.81 vs 7.86 ± 0.77, P < 0.00) compared to those without ED. Patients with ED had lower TT than those without ED (12.04 ± 5.36 vs 17.11 ± 7.11, P < 0.001). Other parameters were not significantly different between the two groups (Table 5).
Demographic and clinical characteristics of patients with and without ED.

Biochemical data of patients with and without ED.
Spearman correlation coefficients revealed significant negative correlation between TT and age
Correlation between TT and some studied variables.

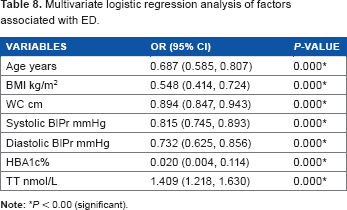
Multivariate logistic regression analysis indicated that age, BMI, and WC were the factors significantly associated with LST (Table 7). In addition, age, BMI, WC, systolic and diastolic BlPr, HBA1c, and serum TT were the main factors associated with ED (Table 8).
Multivariate logistic regression analysis of factors associated with LST.

Multivariate logistic regression analysis of factors associated with ED.
Discussion
Consensus recommendations issued by several international endocrinology and andrology societies recognized T2DM as a risk factor for male HG. 18 Many studies in men with DM have defined HG on the basis of serum testosterone only. Symptoms of HG have rarely been considered in combination with biochemical testosterone deficiency. 7 In the present study, the definition of HG as the combination of symptoms of androgen deficiency defined by ADAM q and low level of TT had been used. It was found that 40% (28 men) had TT < 8 nmol/L, 92.9% of them had symptoms of androgen deficiency, while 60% (42 men) had normal TT > 12 nmol/L. Unexpectedly, 35.7% of men with normal TT had symptoms of androgen deficiency.
It is important to note that the Arabic ADAMq had high sensitivity (86.5%) but a low specificity (24.3%) and is used only in the presence of low testosterone. 9 The original ADAMq also had low specificity (60%), 8 besides the observation that the symptoms of HG are nonspecific. The presence of symptoms was more closely linked to increasing age than to testosterone levels. 7 This further underlies the importance of measuring serum testosterone in diabetic men and not depending solely on clinical symptoms, especially when planning to implement androgen therapy.
In the present study, 40% of the studied subjects had LST, a prevalence rate which is comparable to several cross-sectional studies and systematic analyses from various countries that reported that T2DM is associated with LST. There is limited information about the prevalence of HG among men with T2DM in Arab countries, including Egypt. A recent study by Ghazy et al, 21 found that 33% of men with T2DM had HG. In Jordan, Al Hayek et al, 4 found that 36% of T2DM had LST, and 80% of them had symptoms of androgen deficiency. These differences may be due to difference in ages of the patients, duration of DM, presence of complications, as well as to the difference in the cut-off points used for testosterone.
To clarify if the type of hypogonadal state was due to a hypothalamic–pituitary–gonadal axis or due to a pure gonadal defect, levels of FSH and LH had been also assessed. Although the mean levels of FSH and LH were in the normal range, they were inappropriately lower in patients with LST than in those with normal TT. One would expect that LH levels should be higher in face of this low testosterone. These results would let us consider a state of hypogonadotropic HG, which is mostly functional secondary to a chronic disease such as T2DM. Obesity and IR, conditions that are commonly associated with T2DM, could be associated with impaired function of hypothalamic–pituitary–testicular axis (HPT). 22 Increased levels of tumor necrosis factor alpha and interleukin 1B in obesity was found to reduce hypothalamic GnRH and LH secretion in animal and in in vitro studies. 23
The reports on the levels of gonadotropic hormones in men with T2DM are conflicting. Dandona and Dhindsa 24 found that 25% and 4% of men with T2DM had hypogonadotropic and hypergonadotropic HG, respectively. Onahn et al 25 and Natah et al 26 reported higher FSH and LH in T2DM. Ando et al 27 reported normal FSH and LH in T2DM. However, Hussien and Al-Qatsi 28 reported low levels of FSH and LH.
Prolactin levels were in the normal range, which excludes any role for prolactin in HG-associating T2DM.
Aging is associated with decline in testosterone levels in men. 29 Testosterone begins to decline in the late third or early fourth decade and diminishes at a constant rate thereafter at a rate of about 1.6% annually. An increase in sex hormone–binding globulin (SHBG) by 1.2% per year was also reported. 22 Most of the studies were carried out on the age range 40–70 years. Moreover, Chandle et al 30 reported LST in young T2DM (age range 18–35 years). In agreement with these studies, we found that patients (age range 30–50 years) with LST were older than those with normal TT in addition to an inverse correlation between age and TT. This raises attention toward young and middle-aged patients with T2DM, not only the elderly ones, as this problem might affect their fertility throughout their reproductive years. It is not clear whether the age-related decline in TT levels is because of the chronic illnesses or is it due to aging process per se. Some studies have found that age-associated decline in TT is diminished or abolished in healthy men. 31 The age-related decline in TT is likely a combination of testicular/pituitary/hypothalamic defects, in addition to an age-related alteration in the feedback mechanism of HPT axis. 32
The present study revealed a negative correlation between TT and overall obesity as well as visceral adiposity as assessed by BMI and WC, respectively. Khan et al 33 reported similar results. A plausible explanation for this is the hypogonadal–obesity cycle. 34 Essentially, visceral adipocytes have a high activity of the enzyme aromatase, which converts testosterone to estrogen. In addition, leptin resistance at the HPT levels might cause reduced LH release and testosterone secretion. 35
A negative correlation between testosterone and systolic and diastolic BlPr had been found, which is in agreement with other studies. 36 Chignalia et al 37 presented new evidence on genomic and nongenomic mechanisms of testosterone action on vascular smooth muscle cells in arterial hypertension through modulating associated cellular events.
Diabetes is strongly associated with microvascular complications, including DN, which was more prevalent in patients with LST than in those with normal TT. Grossmann and Wu 38 found similar results.
The presence of sufficient amount of androgen is essential for adequate sexual function in men. Furthermore, androgens also act peripherally, maintaining the integrity of penile structures and facilitating the erectile mechanisms. 39 Androgen has a role in the regulation of intrapenile vasodilatation. 40
In the present study, 85.7% of T2DM patients with LST had ED versus 31% of those with normal TT. Moreover, patients with ED had lower TT than those without ED. This highlights the importance of LST, and consequently HG in the pathogenesis of sexual dysfunction in men having T2DM rather than incriminating diabetic autonomic neuropathy as the main and sole pathogenetic factor. Raising the awareness about this will, consequently provide patients with T2DM wider therapeutic approach that would definitely improve the outcomes. Ghazy et al 21 found that 94.4% of diabetic patients with HG had ED versus 61.0% of the eugonadal patients.
Numerous studies have shown that up to 75% of T2DM have sexual dysfunction, mainly ED. Approximately one-third of men with ED have androgen deficiency. 41 In the present study, the severity of ED had been assessed with IIEF-5 score. An inverse relationship between TT and IIEF-5 score was found, which is consistent with the results of Kapoor et al. 42 Wang et al 43 also confirmed that the lower the serum testosterone, the greater the severity of ED. Moreover, indices of PCDU (PSV cm–1sec and EDV cm–1sec) were lower among patients with ED and LST as opposed to those with ED and normal TT. Corona et al 44 also reported similar results.
Gurbuz et al 45 demonstrated the benefits of testosterone monotherapy on ED. Moreover, combined therapy with testosterone replacement and sildenafil in hypogonadal men converts 60% of sildenafil nonresponders to responders. 46
In the present study, it is noteworthy that patients with ED (46 men) had more comorbidities compared to those without ED (24 men). Rosen et al 47 reported similar results. The design of the present study would not let us delineate if the LST-associating ED was the cause of these comorbidities or the latter were the cause of the LST and ED. However, the devastating nature of T2DM as a chronic disease would probably be the generator of the LST, ED, and the disabling co-morbidities.
In the present study, mean level of HBA1c of all subjects was high (8.45 ± 0.90), patients with LST had higher HBA1c than those with normal TT. In addition, a negative correlation was found between TT and HBA1c. This is in agreement with previously published data.33,48,49 In the present study, multivariate logistic regression analysis of the studied variables revealed that HBA1c was not a significant predictor of LST, which is consistent with the results obtained by some studies.4,7,48 In addition, as reported before, levels of HBA1c were higher in patients with ED than in those without ED.
Chronic hyperglycemia could inhibit the HPT axis, 50 and as shown more recently, acute hyperglycemia could lower testosterone levels. 51 Results of studies assessing the effect of testosterone on glucose metabolism are inconsistent. 6 Although only few randomized controlled trials have been reported, testosterone therapy seems to improve glycometabolic control as well as fat mass.18,52
In the present study, multivariate logistic regression analysis indicated that age, BMI, and WC were the factors significantly associated with LST. In addition, age, BMI, WC, systolic and diastolic BlPr, HBA1c, and serum TT were the main associated factors with ED.
In conclusion, testosterone deficiency and ED, individually and together, are conditions commonly associated with T2DM in Egyptian men. Given the large number of diabetic patients in Egypt (11.4% of the population), the number of diabetic patients with LST is undoubtedly enormous.
Thus, screening of men having T2DM for LST levels is recommended, especially for those presenting with ED. Lifestyle measures with emphasis on weight loss, besides optimization of diabetic control and assiduous care of comorbidities would be very beneficial in preventing LST and thence HG in men with T2DM.
Study limitations include the cross-sectional nature of the study, which does not permit inferences on the direction of causality for the relationship of androgen deficiency, ED, and glycemic control. The small number of patients and lack of control group are other limitations to be considered when implementing data taken from the study.
Footnotes
Abbreviations
Androgen deficiency in aging male questionnaire body mass index blood pressure confidence interval Diabetic neuropathy European association of urology erectile dysfunction end diastolic velocity follicle stimulating hormone hypogonadism glycated hemoglobin hypothalamic pituitary testicular international index of erectile function insulin resistance international society of andrology interlukin-1beta low serum testosterone luteinizing hormone odd ratio penile color duplex ultrasound peak systolic velocity type 2 diabetes mellitus total testosterone tumor necrosis factor alpha waist circumference.
Author Contributions
Conceived and designed the experiments: EOAE, IME, SES. Analyzed the data. EOAE, OAF, IME. Wrote the first draft of the manuscript: EOAE, OAF. Contributed to the writing of the manuscript: SES, IME, LMAB, AG. Agree with manuscript results and conclusions: EOAE, OAF, SES, LMAB, AG. Jointly developed the structure and arguments for the paper: OAF, SES, IME, AG, LMAB. Made critical revisions and approved final version: OAF, EOAE. Penile color duplex ultrasound: LMAB, AG. All authors reviewed and approved of the final manuscript.
