Abstract
Background
Bleeding events after percutaneous coronary interventions (PCI) are associated with patients' age, gender, and the presence of chronic kidney disease, antithrombotic treatment, as well as arterial access site. Patients being treated by PCI using radial access site are associated with an improved prognosis. However, the safety of femoral closure devices has never been compared to radial compression devices following PCI. Therefore, the aim of this study is to evaluate the safety of femoral closure compared to radial compression devices in patients treated by PCI envisaging access site bleedings as well as short- and long-term prognostic outcomes.
Methods
The Femoral Closure versus Radial Compression Devices Related to Percutaneous Coronary Interventions (FERARI) study is a single-center observational study comparing 400 consecutive patients undergoing PCI either using radial compression devices (TR Band™) or femoral closure devices (Angio-Seal™) at the corresponding access site. The primary outcome consists of the occurrence of vascular complications at the arterial access site, including major bleedings as defined by common classification systems. Secondary outcomes consist of the occurrence of adverse cardiac events, including all-cause mortality, target lesion revascularization, and target vessel revascularization during 30 days and 12 months of follow-up.
Results
Study enrollment was initiated in February 2014. The enrollment phase is expected to last until May 2015.
Conclusions
The FERARI study intends to comparatively evaluate the safety and prognostic outcome of patients being treated by radial or femoral arterial closure devices following PCI during daily clinical practice.
Introduction
The occurrence of bleedings represents one of the most relevant complications significantly increasing short-term and long-term all-cause mortality in patients being treated by percutaneous coronary interventions (PCI).1–3 A recent meta-analysis of three randomized controlled trials (OASIS, OASIS-2, and CURE) evaluating patients with an acute coronary syndrome (ACS) without persistent ST-segment elevation (non-ST-elevation myocardial infarction [NSTEMI]) being treated by PCI and different consecutive antithrombotic therapeutic regimens (ie, heparin versus hirudin; clopidogrel in addition to aspirin) demonstrated an increased prevalence of all-cause mortality in those patients developing major bleedings compared to those without (mortality rates: 12.8% versus 2.5%; P < 0.00001). 1 Accordingly, pooling data from studies evaluating bivalirudin and heparin plus glycoprotein IIb/IIIa inhibitors (ie, REPLACE-2, ACUITY, and HORIZONS-AMI) in 17,034 patients undergoing PCI revealed an increased risk of all-cause mortality at 1 year being caused by an increase in non-CABG-related bleedings. 2
However, it is still unclear whether adverse bleedings are associated with access site or non-access site bleedings.3–5 Verheugt et al showed that a higher adjusted mortality risk was associated with non-access compared to access site bleedings (hazard ratio [HR]: 3.94, 95% confidence interval [CI]: 3.07-5.15, P < 0.0001 versus HR: 1.82, 95% CI: 1.17-2.83, P = 0.008). 3 In contrast, another meta-analysis pooled data from three studies, evaluating different antithrombotic therapies (ie, heparin versus hirudin, eptifibatide versus placebo, and lamifiban versus placebo) in a total of 26,452 patients, and demonstrated an increasing risk of mortality accompanied by similar rates of procedure and non-procedure related bleedings. 4 Accordingly, Vavalle et al proved that severe bleedings assessed by the “Global Use of Strategies to Open Occluded Arteries” (GUSTO) classification was associated with an increased risk of death or myocardial infarction at 6 months irrespective of the bleedings' origin (evaluated in patients with NSTEMI randomized either to enoxaparin or unfractionated heparin). 5
In the last decade, it was aimed to reduce bleeding risks both by improved medical therapies and with innovative interventional devices. For instance, bivalirudin was shown to significantly reduce bleeding events in patients with an ACS undergoing PCI.6,7 Smaller sheath diameters of cardiac catheters with better trackability represent an innovative device-related improvement.8,9 Additionally, the wider use of radial arterial access site leads to a reduction of both bleeding risks and major adverse cardiac events (MACE).10,11 Current European guidelines on myocardial revascularization recommend the use of the radial approach in patients with ST-elevation myocardial infarction (STEMI) based only on a class of recommendation IIb (level of evidence A). 12
However, using either the radial or femoral approach as well as using one of the different available vascular closure devices (VCD)13,14 depends highly on the preference of the selected PCI center and local experiences of the performing operators during clinical routine practice.15,16 Accordingly, a multinational observational study revealed that the radial approach was only assessed in 11% of PCI patients evaluated in 23 different countries of four continents. 17 In Germany, the radial approach was used in 24%, whereas the femoral approach was still applied in 76% of PCI patients with STEMI between 2008 and 2012. 18
Arterial Closure Devices Depending on the Arterial Access Site
Radial Approach
Due to the superficial anatomic course of the radial artery, the radial approach reveals an easier manual compression. Immediate remobilization after PCI additionally favors the radial approach compared to the femoral approach, because it is associated with an increase in the patient's comfort. 19 In a large-scaled analysis of the British Cardiovascular Intervention Society (BCIS) database, Mamas et al demonstrated a significant improvement of both short-term 30-day all-cause mortality (HR: 0.71, 95% CI: 0.52-0.97; P < 0.05) and access site complications (HR: 0.38, 95% CI: 0.19-0.75; P < 0.01) in 46,128 patients with STEMI using the radial approach. 11 In contrast, the radial approach is associated with an increased risk of arterial spasms or even occlusion. Additionally, the more tortuous course of the arteries of the upper extremity might sometimes necessitate to switch toward the femoral artery, which might also extend the duration of fluoroscopy.20–22
Different compression methods and devices are currently available on the market. Among these are simple circumferential short stretch bandages and the MAQUET AIR-BAND™ (MAQUET Holding B.V. & Co. KG), Seal One (Perouse Medical), HELIX (Vascular Perspectives Ltd.), Finale® (Merit Medical Systems, Inc.), as well as the TR Band™ (Terumo Corporation). The TR Band™ was recently evaluated as a safe compression device after radial PCI. 23
Femoral Approach
The femoral approach is associated with a faster and easier accuracy of arterial puncture, absence of arterial spasms, increased backup during PCI, and reduced PCI-related radiation time.10,22,24 Additionally, the femoral approach allows multiple punctures and bigger sizes of different sheaths. Less frequently occurring but nonetheless of considerable consequences are pseudo-aneurysms, arterio-venous fistulae and limb ischemia being caused by an occlusion of the femoral artery following PCI. 9 Risk factors increasing the risk of relevant femoral complications such as bleedings include aggressive antithrombotic therapies, 25 female gender, age, and chronic kidney disease.26,27
In order to reduce relevant bleeding rates after femoral PCI, VCD were developed in the mid-1990s. There are three basic types of VCD: collagen based (Angio-Seal™; St. Jude Medical, Inc.), suture based (Perclose ProGlide, Abbott Vascular, Inc.), and staples and clips. 13 Their common primary purpose is to provide an effective and rapid hemostasis in order to reduce relevant bleeding complications and time to discharge.28–30 However, conflicting data concerning their safety have recently been published. Koreny et al summarized results of 30 different studies reporting an increasing risk of developing hematomas compared to standard manual compression. 31 Hence, Tavris et al observed a reduced incidence of vascular complications and bleedings in patients treated with femoral closure devices (such as the Angioseal, Vasoseal, Duett, Perclose, Techstar, Prostar) (OR = 0.68 [0.65-0.70], P < 0.001). 32
Focused Perspective
Only retrospective data are available for directly comparing femoral VCDs with radial compression devices.33,34 Currently ongoing studies will specifically address radial access compared to femoral access using femoral VCD in NSTEMI patients. 35 However, a direct comparison of one specific radial versus one specific femoral closure device during daily routine practice in a real-world fashion has not yet been investigated. Therefore, this study aims to compare the safety of one femoral closure device with one radial closure device with specific regard to differences of access site bleedings, peri- and postprocedural treatment differences, as well as short- and long-term prognostic outcomes in patients with coronary artery disease treated by PCI during daily clinical workup.
Methods
Study Population
The “Femoral Closure versus Radial Compression Devices Related to Percutaneous Coronary Interventions (FERARI)” study is a single-center prospective, nonrandomized observational study being performed at the First Department of Medicine, University Medical Centre Mannheim (UMM) in Mannheim, Germany (clinicaltrials.gov identifier: NCT02455661). The study is carried out according to the principles of the Declaration of Helsinki and was approved by the medical ethics commission II of the Medical Faculty Mannheim, University of Heidelberg, Germany. Written informed consent is obtained from all participating patients or their legal representatives.
Patients being planned for PCI are screened at our cardiologic department and consecutively included in this study, when they are treated either using radial arterial access site in combination with one specific vascular compression device (TR Band™) or using femoral arterial access site in combination with one specific VCD (Angio-Seal™; french size 6F). Inclusion criteria of the FERARI study are listed in Table 1.
Eligibility criteria of the FERARI study.

Patients with diagnostic angiography, prior active bleeding, increased risk of bleeding (defined as relevant thrombocytopenia <50,000/μL or hemorrhagic diatheses), PCIs using femoral approach without or with a different VCD than the evaluated one, age <18 years, and relevant neurologic disorders (such as residual neurologic disability, dementia), as well as patients with language barriers are excluded.
The study is designed as an open-label, observational all-comers study in order to recruite a generalizable and representative study population comparable to the daily practice in other PCI centers.36,37 All patients are followed-up for 24 hours, 30 days, and 12 months after the index procedure both by initial personal in-hospital and final standardized telephone visits.
Primary Outcomes
The primary outcome is defined by all relevant arterial access site complications within 30 days after PCI. Access site complications are listed in Table 2 and mainly consist of bleedings, hematoma (graduated by diameters in centimeter, estimated deepness, and induration) pseudoaneurysms, arteriovenous fistulae, arterial occlusion, nerve injury, and need for vascular surgical repair. Bleedings are classified according to established criteria such as the “Bleeding Academic Research Consortium,” “The Thrombolysis in Myocardial Infarction” (TIMI), and “The Global Use of Strategies to Open Occluded Arteries” (GUSTO). 38 A study-specific classification system for PCI-related complications was developed (so called “FERARI” classification). In case of relevant bleeding complications, hemoglobin, hematocrit, and platelet counts as well as hemostaseologic parameters (such as international normalized ratio, INR) are assessed regularly after PCI.
Outcome definitions of the FERARI study

Secondary Outcomes
Secondary outcomes consist of major adverse events (MAE), such as death (cardiovascular or noncardiovascular), myocardial infarction, stroke, acute kidney injury (AKI), stent thrombosis, and target lesion revascularization (TLR) or target vessel revascularization (TVR), as well as any further bleedings occurring within the following 12 months after initial PCI as shown in Table 2. Further secondary outcomes consist of differences in radiation exposure between both groups. The height of arterial puncture in the femoral group is also evaluated.
Procedures
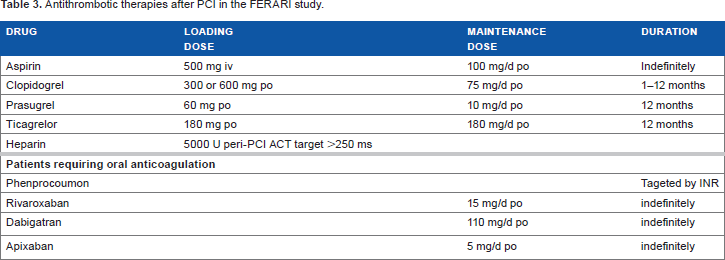
Patients fulfilling inclusion/exclusion criteria were considered for the study. The choice of PCI access site, interventional techniques, and materials such as sheath sizes is not influenced by the study investigators and solely belongs to the operators' discretion depending on the real-life setting in which the PCI is performed. Only four different experienced interventional cardiologists performed PCIs in this study with a minimum volume of 300 PCI cases per year and an equal distribution of experiences with both the radial and femoral approach of each operator. Operators had already implanted at least 100 AngioSeal(R) femoral closure devices per year. Patients were allocated to the access site group, in which the PCI is finally performed. Switching of crossover procedures from radial to femoral access site or vice versa is avoided. PCI and adjunct antithrombotic treatment are initiated as recommended by current European guidelines (Table 3). 39
Antithrombotic therapies after PCI in the FERARI study.
Echocardiographic results, standard laboratory parameters, such as hemoglobin, hematocrit, platelets, creatinine, glomerular filtration rate, and cardiac enzymes, such as troponin I, are assessed before PCI.
Radial Arterial Closure
The TR Band™ is used as the only radial compression device in this study. According to the instruction of use, the sheath is retracted by 2-3 cm after PCI. The green marker of the compression balloon is adjusted proximal to the puncture site, and the strap is fixed on the wrist with the fastener. About 15 mL of air is injected into the balloon, and contemporarily the sheath is removed making sure that there is no bleeding. After four hours of radial compression, the TR Band™ is removed after gradual deflation by 2-3 mL every 30 minutes until final hemostasis. After removal of the band, a sticking plaster is attached. During the process of motor function, vascularization and neurologic sensitivity at the access site are checked regularly.
Femoral Arterial Closure
This study only uses the Angio-Seal™ as the preferred VCD of the femoral access site after PCI in the absence of contraindications. Before placement, a selected angiography of the iliacofemoral region is recommended. In case of slight to normal arterial calcification and straight course of arteries, the decision to implant the AngioSeal™ is made.
The Angio-Seal™ is composed of an absorbable polymer anchor compressing the inner vascular wall and an absorbable collagen sponge compressing the outer vascular wall. According to the instructions of use, an insertion sheath and an arteriotomy locator are snapped together and positioned with an introducer right in the femoral artery noticeable of the blood reflow. After removing the insertion sheath and introducer, the Angio-Seal™ device is inserted through the locator. A clicking sound indicates that the anchor has left the sheath. Pulling back the device, the anchor was pressed against the inner vessel wall and further retreat released the collagen plug in the exterior puncture hole. After removing the whole components of the device, a suture tube appeared. Applying pressure downward and meanwhile pulling back the device hub, the collagen compacts the outer vessel wall. A black mark is released, which should be cut close to the skin. After femoral puncture is closed with Angio-Seal™, a circumferential compression bandage was applied for 6-12 hours and motor function, vascularization, and sensibility were checked regularly.
Statistical Analysis
According to the estimation of the power using the data of the first 100 patients, a sample size of 200 in each group has 90% power to detect an increase of 56% (increase of “0.16” = 16 percentage points) of femoral bleedings at a significant level (alpha) of 0.05 (two tailed). In this estimation of the power with the first 100 patients, the proportion of bleedings in patients with femoral access is 0.45 and that with radial access is 0.29. The difference between the proportions is 0.16 (16 percent points resulting in a 56% increase). In order to achieve balanced groups, patients will be consecutively included in the study until equal numbers of patients according to indications for PCI and relevant periprocedural anticoagulant therapies will be achieved in each treatment group (for instance, for ACS [STEMI/NSTEMI] patients and additional glycoprotein IIIa/IIb inhibitors). Predefined event rates will be compared by frequencies and group percentages, and chi-squared or Fishers's exact test will be applied. Continuous variables will be presented as means and standard deviation, standard error of mean, or median with interquartile ranges depending on the distribution of data. These will be compared by Student's t-test. Known risk factors for access site bleedings,30,40 such as age, gender, body mass index, renal function, hemoglobin, myocardial enzymes, diabetes mellitus, and different kinds of antithrombotic treatment, will be adjusted within multivariable logistic regression analyses for the above described predefined study endpoints. Statistical analyses are performed using SPSS (version 22.0; SPSS) and GraphPad InStat (GraphPad Software, Inc.).
Results and Discussion
The FERARI study starts enrollment in February 2014 and is intended to be finished in May 2015.
The following hypotheses are investigated comparing patients with radial PCI and TR Band™ with patients with femoral PCI and Angio-Seal™ closure device:
Evaluation of access site complications in hospital and within 30 days (primary outcome).
Evaluation of MACE such as death (cardiovascular or noncardiovascular), myocardial infarction, stroke, AKI, stent thrombosis, and TLR/TVR (secondary outcomes) at 30 days and 12 months.
Evaluation of MACE related to access site bleeding, MACE related to non-access site bleeding, and MACE related to total bleeding (secondary outcome).
Evaluations of differences in the height of arterial puncture related to device failure (secondary outcome).
Evaluation of differences between radiation exposures (secondary outcome).
Evaluation of failure rates of vascular closure and compression devices (secondary outcome).
Conclusions
No direct comparison of one specific radial compression with one specific femoral closure device has ever been investigated in patients following PCI. The FERARI study aims to close this lack of evidence by comparatively evaluating the safety and prognostic outcome of patients being treated with one specific radial and femoral arterial closure devices following PCI during daily clinical practice.
Author Contributions
Conceived and designed the experiments: IA, MBe. Analyzed the data: IA, MBe, MÜ, SL. Wrote the first draft of the manuscript: MÜ, MBe, IA. Contributed to the writing of the manuscript: CF, IE-B, KM, RL, MBo, UH. Agree with manuscript results and conclusions: MBo, KM, RL, UH. Jointly developed the structure and arguments for the paper: MBo, KM, RL, UH. Made critical revisions and approved final version: CF, IE-B, SL, MBo, KM, RL, UH. All authors reviewed and approved of the final manuscript.
Footnotes
Abbreviations
Acknowledgments
Thanks in advance to all patients who took part in this study.
