Abstract
Late development of left ventricular (LV) pseudoaneurysms after ventricular tachycardia (VT) catheter ablation is a rare phenomenon, and very few cases have been reported in the medical literature. We describe the case of a giant LV pseudoaneurysm as a late complication of multiple epicardial and endocardial VT ablations in a female in her 50s with known cardiac sarcoidosis.
Case Report
A woman in her 50s presented to the emergency department after receiving multiple shocks from her intracardiac defibrillator (ICD). She had a history of cardiac sarcoidosis with intermittent corticosteroid therapy because of noncompliance. She had frequent admissions at another center because of unstable ventricular tachycardia (VT), which resulted in ICD implantation 13 years prior to presentation. Nine years after initial ICD implantation, the patient developed multiple episodes of hemodynamically unstable VT, necessitating VT ablation procedure. She underwent endocardial radiofrequency ablation (RFA) after scar mapping using CARTO system (Biosense Webster Inc.). Multiple lesions were created in the midanterior left ventricular (LV) wall extending up to the mitral annulus. There was recurrence of VT, and one month later, the patient had a repeat endocardial and epicardial VT ablation in the same anatomic region. This procedure was complicated by the development of cardiac tamponade treated by sternotomy and pericardial window. Owing to continuous development of VT, four months later, another attempt was made using intra-arterial balloon pump-assisted endocardial approach. Multiple lesions were created in LV inferior-septal area extending from midapex to mitral ring, resulting in termination of tachycardia during ablation, but at the conclusion of procedure, VT could be induced. The patient was maintained on medical therapy until two years later when there were multiple recurrences of VT. She underwent successful epicardial ablation focused on an area of scar identified in lateral wall of LV adjacent to mitral ring as well as the inferior-posterior wall. Eight months prior to admission to our institution, the patient had multiple shocks from ICD because of recurrence of VT. She has an endocardial RFA over a scar in the inferior-posterior wall with extension to the lateral wall. An echocardiogram prior to this last procedure showed mild left atrial dilatation, mild diffuse LV hypokinesis, and no evidence of cardiac mass or aneurysm.
On admission to our center, she was found to be in atrial fibrillation with rapid ventricular response. This was interpreted as VT by the ICD resulting in multiple inappropriate shocks. She was otherwise asymptomatic. Chest X-ray on admission revealed retrocardiac opacity (Fig. 1A). On transthoracic echocardiogram, a large 6.1 cm fluid-filled structure was observed along the LA, which compressed it during systole, and some evidence of flow from the LV into the structure (Fig. 1B). A CT angiogram of the heart, performed to better characterize the lesion, showed a large LV pesudoaneurysm involving the inferior wall extending into posterolateral wall. The pseudoaneurysm measured 9 cm in its largest diameter with a 3 cm neck (Fig. 1C). There was evidence of dissection into the myocardium of the inferior wall of the LV adjacent to the neck of the pseudoaneurysm (Fig. 1D). 3D reconstructions and volume-rendered images showed the anatomic relationship of the pseudoaneurysm to the cardiac structures (Fig. 1E and F). She underwent successful surgical repair of the large pseudoaneurysm.
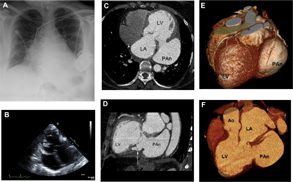
(
Discussion
Catheter ablation of VT in patients with cardiac sarcoidosis has been reported to be effective in eradicating VT as well as reducing the complications associated with recurrent VT1,2; however, rare late complications of this procedure have to be considered in this patient population given their propensity for progressive ventricular remodeling. Common causes of LV pseudoaneurysms include myocardial infarction, cardiac surgery, surgical or external trauma, and rarely, infections.3,4 Small asymptomatic LV pseudoaneurysms have been reported as delayed complications of VT ablation, especially in patients after a myocardial infarction with residual wall weakness and remodeling.2,5 Frances et al reported that the most common symptoms of presentation of larger pseudoaneurysms include congestive heart failure, chest pain, dyspnea, cerebrovascular accident, and arrhythmias. 4
In prior studies, Koruth et al described unusual complications of epicardial ablation of cardiac arrhythmias such as coronary spasm, coronary artery and vein lacerations, intra-abdominal bleeding, hepatic hematoma, and right ventricular pseudoaneurysm. 6 Mansour and collaborators reported the case of a 50-year-old male who presented with syncope and was found to have a 3 × 4 cm LV pseudoaneurysm under the mitral annulus 15 years after ablation for symptomatic Wolff–Parkinson–White syndrome. 7 The case presented here is unique because the patient developed a very large pseudoaneurysm over the area of VT ablation lesions after her last procedure. An echocardiogram performed prior to the last ablation confirmed that there were no major cardiac lesions present. It is interesting that such a large aneurysm remained asymptomatic for a period of about eight months until she was admitted with atrial fibrillation resulting ininappropriate shocks.
To our knowledge, there have been no cases reported of very large asymptomatic LV pseudoaneurysms in a patient with cardiac sarcoidosis after multiple VT ablations. In patients who undergo multiple ablative procedures, especially with combined approaches of endocardial and epicardial ablation, awareness of asymptomatic development of life-threatening complications may be warranted.
Conclusion
This case demonstrates the importance of considering silent, yet potentially life-threatening rare complications after multiple VT catheter ablation, such as large LV pseudoaneurysm in patients with infiltrative heart disease.
Author Contributions
Conceived and designed the experiments: FR, EP. Analyzed the data: KEK, FR, AG, EP, RF. Wrote the first draft of the manuscript: KEK, FR. Contributed to the writing of the manuscript: AG, EP, RF. Agree with manuscript results and conclusions: KEK, FR, AG, EP, RF. Jointly developed the structure and arguments for the paper: KEK, FR. Made critical revisions and approved final version: KEK, FR, AG, EP, RF. All authors reviewed and approved of the final manuscript.
