Abstract
The successful treatment of acute decompensated heart failure continues to evolve with an increasing utilization of nondurable mechanical support devices. Indications for acute support have broadened to include their use as a bridge to recovery or decision (for durable ventricular assist devices [VADs] or heart transplant). Available devices have improved in terms of effectiveness, ease of insertion, and reduction in complications. The commonly used devices (intra-aortic balloon pump, TandemHeart, Impella, and extracorporeal membrane oxygenation circuit), together with their mechanisms of action, are reviewed. Current considerations for support, specific to each device, are examined and future directions and indications for percutaneous VADs are explored.
Why Percutaneous Ventricular Assist?
Acute decompensated heart failure is a growing concern, with an increasing volume of cases driven by an aging population and improvements in myocardial infarction and heart failure survival. In the not-too-distant past, the options for treatment of the advanced heart failure patient, refractory to medical therapy with evidence of low output and hypoperfusion, were limited. Heart transplantation remains a limited resource and durable ventricular assist devices (VADs), although improving in terms of efficacy, safety, and reduction in complications, remain restricted. Improvement in survival is critically dependent on the timing of circulatory support. 1 The attitude toward, and application of, nondurable mechanical circulatory support (MCS) for the short-term support of acute heart failure continues to rapidly evolve and has assumed an increasing role for acute circulatory support and as a bridge to decision. In this algorithm, nondurable MCS can be implemented as a hoped-for bridge to recovery (eg, postcardiotomy syndrome, myocarditis, and acute myocardial infarction). 2 MCS also allows time for clinical evaluation and potential recovery of multisystem organ dysfunction, which may improve the candidacy of the patient with persistent severe cardiac dysfunction for a durable VAD either as destination therapy or as a bridge to transplant. 3
Advances in technology have dramatically improved both the effectiveness and ease of use of percutaneous VADs relative to the long-time standby of the intra-aortic balloon pump (IABP). These advances have led to the earlier and more-frequent application of MCS over a much broader spectrum of patients. Nondurable mechanical support today is more frequently initiated prophylactically in situations associated with a high likelihood of hemodynamic compromise and at an earlier stage of heart failure, before the downward cycle of progressive shock with associated end-organ compromise and inflammatory activation has been established. At the other end of the spectrum, demonstrated success in enhancing survival in otherwise-hopeless resuscitation attempts has seen the extension of mechanical support to selected cases of cardiac arrest with circulatory failure, wherein restoration of spontaneous circulation could not otherwise be established.
The initial devices implanted by cardiac surgeons have commonly been supplanted by percutaneous devices inserted by cardiologists as the ease of deployment and efficacy of these devices for circulatory support has improved (Table 1). Current devices work either in series or in parallel with the heart to provide circulatory support. A recent study by Dr Stretch 4 reported the use of short-term MCS devices in the United States from 2004 to 2011 using the Nationwide Inpatient Sample from the Health Care Cost and Utilization Project. He reported a 1,511% increase in percutaneous devices between 2007 and 2011; nonpercutaneous devices increased by 101% over the same time interval. A comparison of the initial 2004-2007 time interval to the interval 2008-2011 demonstrated a significant 19.1% reduction in observed mortality, from 41.1% to 33.4% (P for trend = 0.027). The subset of patients with cardiogenic shock also demonstrated a significant 16.5% reduction in mortality during this time interval, from 51.6% to 43.1% (P for trend = 0.012).
Common options for nondurable percutaneous MCS.

The most common primary diagnosis in patients receiving MCS was acute myocardial infarction. The frequency of this diagnosis increased from 32.0% in 2004-2007 to 37.8% in 2008-2011 (P = 0.003). Heart failure comprised 11.9% of baseline primary diagnosis, increasing to 14.4% over these time intervals (P = 0.055). The length of stay during these periods declined significantly for patients with congestive heart failure, from 58.7 ± 65.5 to 32.3 ± 35.9 days (P = 0.002). Acute myocardial infarction-related length of stay decreased from 18.5 ± 27.3 to 14.3 ± 19.5 days (P = 0.09). There was an associated significant decrease in the cost of treating heart failure (from $217,144 to $190,612; P = 0.01) and of treating acute myocardial infarction (from $142,176 to $97,134; P = 0.015). The cost of treating cardiogenic shock also declined significantly from $171,509 to $146,942 (P = 0.034).
Acute decompensated heart failure may be due to right or left heart failure or both. Left ventricular (LV) decompensation may occur acutely due to a variety of events, including postcardiotomy shock, myocarditis, ischemia/infarction (ST-segment elevation myocardial infarction or STEMI), valvular failure (eg, acute mitral regurgitation). Acute LV decompensation in patients with chronic heart failure occurs commonly and may be precipitated by ischemia, arrhythmias, renal or pulmonary dysfunction, and dietary indiscretion, but often, it has no clear precipitating event. Isolated right ventricular dysfunction may occur secondary to severe pulmonary events (pulmonary embolism, pneumonia, and acute respiratory distress syndrome), but right ventricular dysfunction is also commonly seen secondary to LV dysfunction or in the context of right ventricular infarction due to acute right coronary occlusion.
The objectives of VADs are to unload the failing ventricle (decreasing myocardial oxygen demand and promoting favorable remodeling) and to maintain an adequate systemic pressure and output to provide end-organ perfusion and function (including particularly the head, heart, and kidneys). Achieving these objectives should translate into a decrease in short- and long- term morbidity, a decrease in length of stay and 30-day readmissions, and an improvement in survival. Considerations of the degree of support required, whether right, left, or both ventricles require support, and whether pulmonary function is adequate for effective gas exchange (oxygenation and CO2 removal) determine the device(s) selected.
Many of the complications of earlier devices have substantially decreased with the development of smaller percutaneous devices that are increasingly effective in providing circulatory support. To varying degrees, all the percutaneous support devices may compromise circulation distal to the site of insertion in relation to the catheter size and have associated concerns of vascular damage, thrombosis, hemolysis, bleeding (due to necessary anticoagulation), and infection. Aortic insufficiency is of particular concern with the IABP and Impella support devices.
Patient Selection
In patients with cardiogenic shock (“crash and burn,” INTERMACS [Interagency Registry for Mechanically Assisted Circulatory Support] profile 1), placement of a percutaneous assist device can provide immediate stabilization and effectively improve short- and long-term survival. Timely placement of a nondurable percutaneous VAD (pVAD) results in rapid improvement of hemodynamics, decreases the need for inotropes/pressors/diuretics, and lessens the risk of progressive multisystem organ failure. Use of pVADs as a bridge to recovery has been particularly effective in patients with cardiogenic shock due to conditions in which significant recovery of ventricular function is often seen (eg, postcardiotomy syndrome, myocardial infarction, acute fulminant myocarditis). In patients demonstrating failure to recover, timely nondurable pVAD placement improves candidacy for more durable VAD support by improving hemodynamic stability and cardiac output, decreasing the degree of multiorgan dysfunction. Percutaneous VAD placement may also be reasonable in patients in whom the eventual treatment plan is undecided (bridge to decision). By providing continued circulatory support in critically ill patients with uncertain neurologic status and undetermined candidacy, pVAD placement allows for potential optimization/reversal of multisystem organ dysfunction and allows time for the appropriate assessment of potential risks and benefits for durable VAD or heart transplant.
Contraindications
The use of percutaneous support devices as a bridge to decision has considerably widened the window of suitable patients. However, end-of-life considerations are still pertinent if known. Do not resuscitate/do not intubate (DNR/DNI) status, terminal illness with a limited prognosis, candidacy for VAD or transplant if cardiac recovery is unlikely, duration and quality of antecedent cardiopulmonary resuscitation (CPR), presence of multiorgan system dysfunction (particularly neurologic), and baseline quality of life may all enter into the decision to provide support. All patients receiving a percutaneous support device require anticoagulation therapy (to prevent thrombosis) and close monitoring of access sites with chronic indwelling catheters (large cannula may cause vascular damage, downstream ischemia). Consideration must therefore also be given to relative contraindications such as conditions increasing the risk of bleeding (recent surgery, cerebrovascular accident (CVA), active bleeding), severe peripheral vascular disease increasing the likelihood of limb ischemia, presence of bacteremia increasing the likelihood of difficult-to-treat line sepsis and septic shock.
Axial support devices are associated with mechanically induced hemolysis and an acquired von Willebrand syndrome with decreased platelet aggregation. 5 In patients undergoing chronic treatment with continuous flow devices, bleeding from cerebral and gastrointestinal arteriovenous malformations may be seen and complicate durable VAD therapy. Anticoagulation must be closely monitored for adequacy to prevent thrombosis and to maintain surveillance for heparin-induced thrombocytopenia if heparin is used as the anticoagulant. With the exception of the IABP, the commonly used percutaneous VADs provide continuous flow.
Nondurable Percutaneous VADs
There are effective VADs that require surgical placement (Thoratec PVAD, Abiomed AB5000, BVS 5000, centrifugal pumps requiring median sternotomy such as CentriMag) and have been used for both short- and longer-term support.
The IABP (multiple vendors, including Arrow, Datascope/Maquet, Teleflex) has a long tradition of use as an emergency aid in the treatment of acute heart failure associated with acute myocardial infarction. It can be readily inserted in the cardiac catheterization laboratory percutaneously via the femoral artery, with the balloon placed in the proximal descending aorta. In emergent cases, it can be placed at the bedside with confirmatory X-ray after placement. Inflation of the balloon, synchronized with diastole, provides diastolic augmentation of arterial pressure and an increase in the mean arterial pressure. Because coronary blood flow occurs predominately in diastole when the aortic-ventricular (transmyocardial) pressure gradient is greatest, diastolic augmentation of aortic pressures increases coronary artery pressures and flow and may recruit collaterals, improving myocardial perfusion in acute coronary ischemic syndromes. Balloon deflation provides a measure of presystolic unloading of the left ventricle and increased LV ejection time. The increase in cardiac output associated with IABP use is modest. The first clinical trial of IABP use in cardiogenic shock demonstrated an augmented cardiac output of 0.5 L/min. 6 The IABP often has an immediate effect of stabilizing patients and improving systemic pressure, in addition to improving coronary, cerebral, and peripheral perfusion.
Case series comparing the use of IABP before versus after primary percutaneous coronary intervention (PCI) in patients with cardiogenic shock complicating acute myocardial infarction have reported dramatic differences in in-hospital mortality (19% vs 59%; P = 0.007) and major adverse cardiac events (23% vs 77%; P = 0.0004), favoring early IABP placement. 7 Randomized trial demonstration of benefits have, however, been largely disappointing in the era of PCI treatment of STEMI with cardiogenic shock. 8 The IABP functions as a serial device with the heart, with inflation and deflation timed off the electrocardiogram or aortic pressure tracing. The dependence on intrinsic electrical and mechanical cardiac function is a major drawback in patients with significant tachycardias (heart rate >120 beats/minute typically leads to ineffective diastolic augmentation) or arrhythmias affecting timing or output (eg, ventricular tachycardia, ventricular fibrillation) or those with electromechanical dissociation.
The incidence of major complications associated with the IABP placed for cardiogenic shock complicating myocardial infarction has been reported at 2.6%. The minor complication incidence of 4.2% included major limb ischemia in 0.9% and bleeding requiring transfusion or surgical intervention in 0.8% of patients. 9
The
The
Proper positioning of the Impella axial pump centered across the aortic valve is essential for correct functioning. Falling flow rates postinsertion can be due to several concerns, including catheter migration/malposition, inflow obstruction due to “suckdown,” thrombus, and catheter kinking. Suckdown may have multiple causes with a common theme of left ventricular underfilling: right ventricular failure/pulmonary hypertension, hypovolemia, and an excessive reduction in preload. Catheter repositioning can usually be accomplished with imaging and use of the displayed ventricular pressure tracing but may at times require device replacement. During suckdown, the catheter/cannula coming in apposition with the ventricular wall or other structures reduces device inflow and increases power consumption. Due to the multiport pigtail catheter design, inflow compromise of the Impella is usually not as dramatic or as severe as that seen with an LVAD cannula. Suspected suckdown is initially treated with a reduction in pump speed and fluid resuscitation, while a transthoracic echocardiogram (TTE) is obtained for further clarification of the cause of dysfunction. TTE provides key information regarding left and right ventricular chamber sizes and function, catheter placement and function, and presence of thrombus. Due to its noninvasive nature and bedside portability, TTE is the initial modality of choice for determining and treating device concerns. TTE also plays an indispensable role in assessing recovery of LV function, determining the timetable and potential for weaning, or the need for a prolonged support strategy (eg, LVAD). 13 When indicated, transesophageal echocardiography (TEE) provides consistent and superior imaging to TTE and is of particular utility in detecting cardiac thromboembolic concerns (eg, atrial shunts, intracardiac thrombus), endocarditis, cannula course, positioning and complications.
Special Considerations
Because of their ease of insertion and ability to provide substantial circulatory support, the Impella 2.5 and CP are frequently used to support procedures with an associated high risk of acute heart failure and shock in both the catheterization laboratory and electrophysiology suite. Offering greater circulatory support than the IABP, the Impella is frequently used in higher-risk interventional cardiology procedures such as intervention on the last remaining vessel and for multivessel and left main disease interventions in patients with severe LV compromise. 14 While providing demonstrably better hemodynamic support and a trend toward fewer major adverse events, clear outcome superiority over the IABP has not been demonstrated (with the Impella 2.5). The Impella is also used in the electrophysiology laboratory for the support of patients undergoing ventricular tachycardia mapping and ablation. In this cohort of patients, often with severe LV dysfunction and chronic heart failure, the Impella adds a substantial margin of safety and tolerability to an often-prolonged procedure. 15 The common use of arteriotomy site preclosure (Perclose ProGlide, Abbott) allows for safe removal of the Impella immediately postprocedure.
Extracorporeal membrane oxygenation circuit (
ECMO can provide the greatest degree of cardiopulmonary support in terms of maintaining oxygenation, systemic blood pressure, and cardiac ouput. By maintaining systemic blood pressure ECMO may also enhance coronary blood flow (depending on the transmyocardial pressure gradient). When used alone, however, it has significant limitations in treating severe left heart failure due to its effects on loading conditions. By increasing afterload without significantly affecting preload, progressive increases in ventricular distention and diastolic pressures may result in worsening LV function and intractable pulmonary edema/hemorrhage. Options that have been used for treatment include decreasing flow rate (to decrease afterload), dobutamine administration (to improve contractility and decrease afterload), and adjunctive use of IABP or Impella devices (for afterload and preload reduction, respectively).
The site of ECMO arterial return is also important, with the highest oxygenation of blood near the site of the arterial return cannula. Arterial return via the subclavian or brachiocephalic delivers oxygenated blood closer to the heart and head. Central cannula can also support patient mobilization, while femoral access will mandate continued bed rest. A reperfusion line distal to the cannula insertion is often routinely placed to supply oxygenated blood to the limb beyond the often-obstructing cannula. The flow produced with ECMO is limited by the diameter of the cannula, particularly that of the venous intake (flow is proportional to the fourth power of cannula diameter, by Poiseuille's law).
Due, in part, to the comorbidity of the indicated patient population, venoarterial ECMO has reported substantial complications. These include major/significant bleeding (40.8%), significant infection (30.4%), lower extremity ischemia (16.9%), lower extremity amputation (10.3%), and fasciotomy/compartment syndrome (10.3%). 18
Special Considerations
The IABP has been used in conjunction with ECMO to provide augmentation of coronary blood flow/myocardial perfusion and afterload reduction. 19 The IABP used in conjunction with ECMO has very different effects on delivery of oxygenated blood to the head and heart depending on the placement of the arterial cannula. When arterial return is proximal to the IABP (ie, subclavian), oxygenation of the head and heart is improved with adjunctive use of the IABP; when the arterial return is distal to the IABP (ie, femoral), IABP use decreases the delivery of oxygenated blood to the head and heart.20,21
The “Harlequin syndrome” occurs when deoxygenated blood is delivered to the head and upper extremities (blue head) and the lower extremities remain oxygenated (red legs). This can indicate LV function recovery, with the recovering heart competing with ECMO flow in the aorta. It can also indicate further pulmonary deterioration, causing an increasing proportion of deoxygenated blood delivered to the upper body (head and coronary perfusion of particular concern). If the cause is determined to be further deterioration of pulmonary function, it may be treated by returning oxygenated blood to the superior vena cava (SVC) (important to minimize recirculation through the collecting cannula). However, the added preload may further compromise a failing left ventricle. An IABP proximal to the ECMO femoral artery cannula placement can also cause the Harlequin syndrome. Post-ECMO placement oximetry of the right upper extremity or ear lobe (cerebral oxygenation is also sometimes monitored by near-infrared spectroscopy) is routinely used to ensure adequacy of oxygenation and monitor for this occurrence.
Venoarterial ECMO is increasingly used in the treatment of refractory cardiac arrest (extracorporeal CPR or ECPR), with field deployment by mobile units and in the Emergency Department (ED) setting. Deployed by critical care physicians in the ED, in conjunction with hypothermia and primary PCI, if indicated, this technique has led to reports of high survival rates (54%) with neurologically intact patients. 22 The 2014 Extracorporeal Life Support Organization International Summary reported 38% of ECPR-treated adults surviving extracorporeal life support and a survival to discharge or transfer of 29% (http://elsonet.org/registry/statistics/limited). ECPR survival rates were even higher in neonates and pediatric patients. Data from multiple experiences suggest that the best results for venoarterial ECMO for refractory cardiac arrest (ECPR) occur when therapy is initiated within 30 minutes of the arrest.
Choosing a Device
VADs have potential benefits in increasing myocardial blood flow (O2 supply), decreasing O2 demand (by decreasing afterload and preload/wall stress, but not directly affecting contractility), and supporting cardiac output. In terms of outcomes, maintaining an adequate perfusion pressure is as important as the cardiac output. 23 The cardiac power output (CPO) (watts), a combined measure of output and end-organ perfusion pressure (CO X mean blood pressure/451) may be more useful than cardiac output alone in assessing end-organ perfusion. The CPO has been used to predict worsening heart failure at 30 days (CPO <0.6watts) and mortality in cardiogenic shock (CPO <0.53 watts). 24
The IABP, by providing diastolic augmentation and increasing the transmyocardial pressure gradient, is effective in improving myocardial oxygen supply, with small effects on decreasing afterload and increased cardiac output. The TandemHeart devices have a balanced effect on myocardial oxygen demand. The TandemHeart decompresses the ventricle, by decreasing LV end-diastolic pressure and volume (decreases myocardial oxygen demand), but increases afterload by returning oxygenated blood from the left atrium to the aorta, maintaining arterial pressure, and increasing afterload (increases demand). The TandemHeart is clearly more effective than the IABP in increasing cardiac output and CPO and may increase oxygen supply by increases in cardiac output and perfusion pressure. The Impella devices (2.5 [maximum cardiac output 2.5 Lpm], CP [maximum cardiac output 4.00 Lpm]) are similar to the TandemHeart in decreasing preload but do not appreciably effect afterload, resulting in a significant decrease in myocardial oxygen demand, in addition to being equally effective in increasing myocardial oxygen supply, cardiac output, and cardiac power index. By contrast, ECMO provides the greatest increase in cardiac output and CPO but does not decrease myocardial oxygen demand (increases afterload and LV distention/wall stress) or promote favorable remodeling.
The initial choice of device will depend on whether cardiac support alone is needed or whether cardiopulmonary support is necessary. If cardiopulmonary support is needed, ECMO in the venoarterial configuration provides necessary oxygenation of blood in addition to circulatory support. As baseline cardiac function and organ perfusion become increasingly compromised, greater degrees of support are indicated. The IABP is the easiest device to place but provides little systemic support and requires a cardiac trigger (electrical or mechanical). In many catheterization laboratories, the Impella has become the device of choice, because of its ease of insertion and its effectiveness in decreasing preload, improving cardiac perfusion, and maintaining systemic circulatory support. Other advantages of the Impella 2.5 and CP are the decrease in the caliber and number of cannulas required, bearing in mind that larger catheters are associated with greater difficulty in placement (may require surgical/graft placement) and more vascular injury and bleeding complications.
Clinical Trials
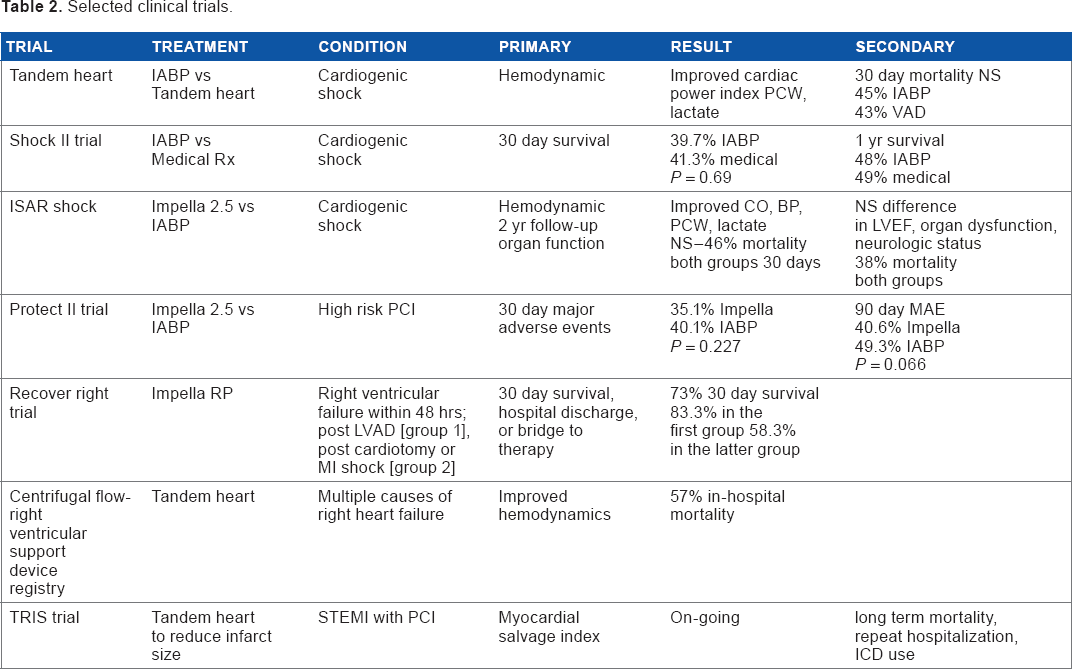
The newer percutaneous support devices are clearly effective in supporting the heart and circulation. They decrease LV filling pressures and end-diastolic volume, decrease myocardial ischemia, and maintain both cardiac output and perfusion of the vital organs. In the catheterization laboratory, they allow safer and more prolonged procedures in patients with severe LV dysfunction and/or large potential or ongoing ischemic burdens. By providing circulatory support, the new devices, deployed in a timely manner, effectively lessen the risk of a downward spiral of multisystem deterioration, often seen in cardiogenic shock patients in the past. Despite the clearly beneficial immediate hemodynamic effects, which include a decrease in filling pressures, improved cardiac power index, increased cardiac output, and decrease in lactate levels, demonstration of survival benefit has proved a more elusive end point. Randomized trials to date (Table 2)25–33 have only been able to demonstrate trends toward improved short- and long-term survival and decreases in major adverse events and end-organ dysfunction. There are probably a number of reasons for the inability to demonstrate clear superiority of the newer devices over medical therapy or IABP placement. These include study size, enrollment, timing of intervention, significant comorbidity masking intervention effectiveness, and studies preceding the currently improved devices.
Selected clinical trials.
What Does the Future Hold?
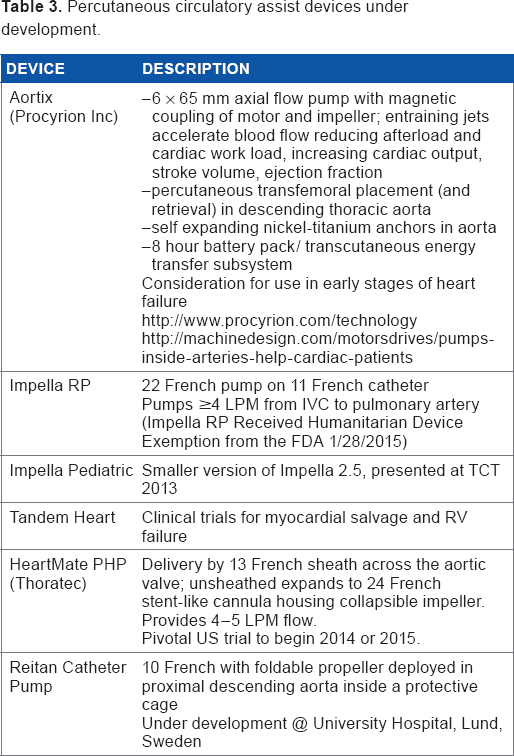
The field of percutaneous mechanical support is continuing to evolve with the development of easier-to-deploy devices that are smaller, safer, and more effective in providing hemodynamic support (Table 3; devices under development). As a consequence, treatment will be initiated earlier, in anticipation of cardiac decompensation, lessening the risk of circulatory compromise and multisystem organ failure. The incorporation of a bridge to decision rationale has significantly increased the potential candidates for percutaneous support. Axial (serial) devices are increasingly dominating circulatory support wherein the problem is confined to cardiac dysfunction, while parallel devices (ECMO) remain necessary when gas exchange is inadequate. Percutaneously deployed, self-contained circulatory assist units acting in series with the circulation and that are capable of induction recharging (Aortix) are already under development and may blur the lines between durable and nondurable mechanical support devices. Right heart failure support devices promise to significantly improve our ability to treat this currently frustrating condition with limited treatment options. Finally, the significant improvement in neurologically intact survival demonstrated with early ECMO as part of a resuscitation and support strategy for appropriate CPR patients without return of spontaneous circulation may well lead to a much wider implementation of mechanical support initiated in the ED and an increasing use of mobile ECMO teams.
Percutaneous circulatory assist devices under development.
Author Contributions
Wrote the first draft of the manuscript: WL. Contributed to the writing of the manuscript: WL, MK. Agree with manuscript results and conclusions: WL, MK. Jointly developed the structure and arguments for the paper: WL, MK. Made critical revisions and approved final version: WL, MK. Both authors reviewed and approved of the final manuscript.
