Abstract
As we continue to care for an older and sicker end-stage heart failure population, it has become challenging to evaluate patients based on current risk scores that mainly focus on subjective symptoms and patient disability. For generations, geriatricians have sought to identify the body's underlying vulnerabilities that characterize frailty. More recently, cardiologists have begun to recognize this entity in their own practice. Several studies have suggested rates of frailty as high as 50% in patients with cardiovascular disease. However, despite recognizing frailty, it remains difficult to define. Like heart failure, frailty is a biologic syndrome that affects multiple organ systems. Measures of frailty are shown to strongly correlate with adverse outcomes in the health care system.
Keywords
Introduction
The United States population living with heart failure (HF) is aging. As of 2013, 80% of patients with HF were aged ≥65 years, and 25% were aged ≥80 years. 1 Mortality is high, with 50% of Medicare beneficiaries surviving <3 years following hospitalization for HF. In the United States, costs associated with HF exceed $35 billion annually. Future costs estimated using an American Heart Association model predict that by 2030, 1 of 33 people will have HF, with direct costs of caring for these patients projected to rise to $53 billion. 1 The high mortality and costs associated with caring for HF makes this one of the most clinically challenging chronic diseases to treat. As we continue to care for an older HF population, we are limited by our current assessment tools that primarily focus on subjective symptoms and patient disability. For generations, geriatricians have sought to identify the body's underlying vulnerabilities that characterize frailty. More recently, cardiologists have begun to recognize this entity in their own practice. Several studies have suggested rates of frailty as high as 50% in patients with cardiovascular disease. 2 Like HF, frailty is a biologic syndrome that affects multiple organ systems. This review emphasizes the importance of measuring and assessing frailty in the evaluation of HF patients by summarizing the existing literature uniting HF with frailty. Ideally, we aim to offer a fresh perspective on integrating frailty tools into our current risk stratification of HF patients evaluated for cardiac resynchronization, mechanical circulatory support, aggressive medical regimens, and perhaps transplantation. In addition, some of the referrals we make to HF management programs and cardiac rehabilitation may be better served through more sensitive tools utilizing this concept. This review was compiled from a PubMed search including all items related to HF, cardiovascular disease, and frailty.
Definition of Frailty
The concept of frailty currently lacks a formally accepted definition; however, it is widely recognized in the health care literature. In essence, frailty is a biological syndrome of decreased homeostatic reserves, resulting in increased vulnerability to stressors. 3 Frailty clinically manifests when a physical or psychological stressor causes a disproportionate change in health status. For example, an elderly subject with reduced ejection fraction undergoes elective same-day implantation of a defibrillator, which subsequently becomes complicated by a hematoma, resulting in an extended hospitalization and recovery period. An important principle is that frailty arises from an accumulation of subclinical deficits across multiple physiological systems, which, eventually, as an aggregate, surpass a threshold and become clinically apparent. The systems involved in the frailty pathobiology include the musculoskeletal (sarcopenia/osteopenia), endocrine (excessive catabolism), gastrointestinal (micronutrient deficiencies), and immune (a proinflammatory state) systems. 4
Tools to Describe Frailty
Frailty tools.

Frailty can also be assessed as a multidimensional accumulation of deficits as opposed to a single clinical phenotype. This approach is best represented by the Frailty Index, which was developed as a part of the Canadian Study of Health and Aging, wherein 92 baseline variables consisting of signs, symptoms, disabilities, and laboratory values were used to define frailty.7,8 The index is calculated as the proportion of variables present to the total assessed and is strongly linked to risk of death and institutionalization. Subsequent work has demonstrated that the Frailty Index can be simplified to a 30-item bedside assessment tool without the loss of predictive validity. 9 The Changes in Health, End-stage disease and Signs and Symptoms (CHESS), the MacArthur Study of Successful Aging (MSSA), and the Resident Assessment Instrument (RAI 2.0) scores employ the accumulation of deficits model. This scoring system, as compared with the phenotypic model, examines a gamut of deficits including impairment, disability, and comorbidities.10,11
Our current HF subjective assessment tools, the Kansas City Cardiomyopathy Questionnaire and the Minnesota Living with Heart Failure Questionnaire, assess quality of life; they do not assess the entire phenotypic picture that defines frailty.
Frailty, Disability, and Heart Failure
The field of geriatrics leads clinical medicine in the ability to evaluate and manage patients with complex chronic diseases by acknowledging the interrelated but distinct concepts of disability, frailty, and comorbidity. While an understanding of the concept of comorbidities (the co-occurrence of multiple diseases) is self-evident to most, the distinction between disability and frailty is not well appreciated in the health care field. Disability is commonly defined as difficulty with either activities of daily living or mobility, whereas frailty represents a state of heightened vulnerability that places an individual at risk for adverse outcomes. Although frailty and disability often overlap, they should be considered distinct as they carry independent prognostic implications
4
(Fig. 1).
Overlap of disability, comorbidity and frailty. Reproduced with permission from Fried LP et al. J Gerontol A Biol Sci Med Sci. 2001;56:M146-57.
Similar to geriatrics, effective care of HF requires a comprehensive approach that expands beyond the traditional disease-oriented model. 12 Despite increased emphasis on comprehensive management of HF, current assessment of HF focuses solely on disability. In fact, the most commonly used tools for evaluation of HF assess functional capacity: the New York Heart Association (NYHA) Class, the cardiopulmonary exercise test, and the 6-minute walk test. 13 These tools, which, in essence, assess disability, have been incredibly valuable as they carry strong prognostic ability, form the inclusion criteria into clinical trials of HF, and therefore guide therapy. 14 Nonetheless, by focusing solely on disability, there may be a missed opportunity in HF, as illustrated by the successful incorporation of frailty into geriatric assessments.
Summary of trials in heart failure and frailty.
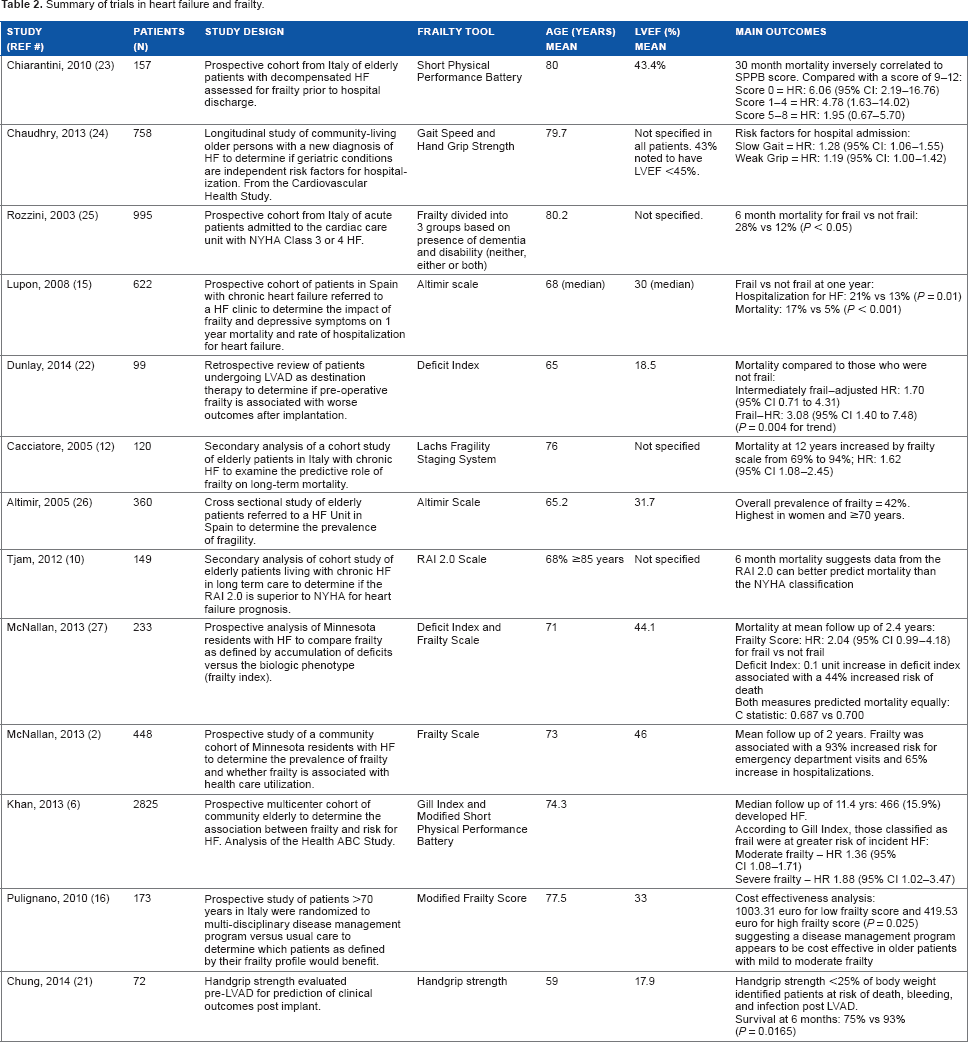
Frailty for Selection of Invasive Therapies in HF
Therapy for HF has been dictated by clinical trials in which the average age of the patients enrolled tended to be <65 years; this stands in contrast to the HF population in the United States with an average age in the mid-70s. 17 Furthermore, there has been a rapid rise in device-based therapies for the treatment of HF, which raises the issue of whether the adverse-event profile demonstrated in clinical trials translates appropriately to the “real-world” patient. The use of frailty measures in patients considered for invasive therapies is beginning to gain recognition in the non-HF literature. It is generally agreed upon that the population with HF, in particular, is vulnerable to poor outcomes if not carefully selected. It is therefore logical to assume that frailty may be an important tool in risk stratification for device therapy in the older HF patient.
In 2011, Matlock et al. 18 described an interesting practice pattern. His group surveyed 1124 cardiologists in regard to their current attitudes and recommendations for implantable cardiac defibrillators (ICD). The investigators found that physicians in high-utilization areas were more likely to recommend an ICD to frail patients or patients with life expectancies <1 year – a group less likely to garner benefit and more likely to have complications. 18 Unfortunately, the criteria used to describe frailty in their paper relied upon age. The investigators are to be commended for raising the issue of frailty as a prognostic tool before ICD use; however, their study is limited by their misconception that aging is an equivalent to frailty.
The evidence for cardiac resynchronization therapy stems from studies of patients in their 60s. 19 In order to assess the effectiveness of cardiac resynchronization therapy (CRT) in an octogenarian population, Foley et al. 19 evaluated patients before and after CRT implantation with a 6-minute walk test and the Minnesota Living with Heart Failure Questionnaire. There was an improvement in both scores for patients older or younger than 80 years, a finding that emphasizes that age alone is an inadequate prognostication tool. The two aforementioned studies are important investigations that allude to our current gap in risk stratification of elderly HF patients undergoing ICD placement and/or CRT – further studies examining whether frailty can fill this gap are necessary.
The cardiac surgery literature offers precedents for frailty assessment as a risk stratification tool. The short-distance 5-m gait speed is a simple-to-perform tool that has been adopted by many preoperative research protocols. Afilalo et al. 20 prospectively demonstrated that a slow 5-m gait speed before cardiac surgery was associated with a three-fold increase in postoperative morbidity and mortality. Similarly, hand grip strength has been shown to predict outcomes after left ventricular assist device (LVAD) placement. 21 In addition, in a retrospective analysis, categorization of frailty via the deficit index was associated with mortality after LVAD implantation. 22 These data in cardiac surgery demonstrate the potential of frailty assessment in patient selection for invasive therapies.
Future Directions
While the addition of frailty tools to the management of the HF patient holds promise, multiple topics must be clarified and validated before mainstream clinical application. These include identifying which of the many tools provides the best combination of performance and facility. Furthermore, cohort studies to define standardized cutoff values of frailty within these tools, based on age and sex, are necessary. This is particularly relevant for the younger population with HF because frailty may coexist, but identifying it based on values validated in an older population may underestimate its prevalence. In this regard, integration of biomarkers of frailty may increase the sensitivity of current tools. The next step would be to design intervention trials based on frailty assessment in HF. These could include the following: (1) selection for invasive therapies, whereby the outcomes would include not simply procedural success but also quality of life and cost-effectiveness; (2) larger prospective validation of referral to an intensive disease management program, wherein geriatric interventions are combined with those of the HF field; and (3) referral to cardiac rehabilitation and nutritional supplementation based on frailty status.
Conclusion
It is intriguing to speculate that integration of frailty assessment into the clinical evaluation of HF may refine our understanding of this disease. Ultimately, incorporation of frailty into HF algorithms may be the next advancement in the management of this complex disease by allowing providers to make more informed decisions and make more responsible and efficient use of health care resources.
Author Contributions
Conceived the concepts: SP. Analyzed the data: RJ. Wrote the first draft of the manuscript: RJ. Contributed to the writing of the manuscript: RJ. Agree with manuscript results and conclusions: SP. Developed the structure and arguments for the paper: RJ. Made critical revisions and approved final version: SP. Both authors reviewed and approved of the final manuscript.
