Abstract
Lipid goal attainment studies in Asian patients with acute coronary syndrome (ACS) are limited. The objectives of this study were to determine low-density lipoprotein cholesterol (LDL-C) goal attainment rate at 4 months, and to examine prescription behavior influencing lipid goal attainment in Asian patients with ACS. A retrospective analysis of 267 patients with ACS was performed. The mean follow-up duration was 41.2 ± 10.7 months. LDL-C goal attainment rate was highest at 4 months (36.7%) but declined progressively throughout follow-up. More than 85% of patients were discharged with equipotent statin dose of 2 (equivalent to simvastatin 20 mg) or less. In patients who did not attain LDL-C goals, the statin dose remained low throughout follow-up because of a lack in responsive dose titration. Aggressive lipid-lowering therapy should be initiated early to improve goal attainment in these high-risk patients.
Keywords
Introduction
Extensive evidence suggests that aggressive lowering of low-density lipoprotein cholesterol (LDL-C) with 3-hydroxy-3-methylglutaryl coenzyme A reductase inhibitors (also referred to as statins) can reduce cardiovascular events in both the primary and secondary prevention settings. Landmark randomized controlled trials have demonstrated that additional reductions in LDL-C with intensive therapy significantly reduce cardiovascular events in high-risk individuals with acute coronary syndrome (ACS).1–4
Based on data from clinical trials, the National Cholesterol Education Program (NCEP) Adult Treatment Panel III (ATP III) first established treatment targets for LDL-C in 2001, which were later revised in 2004. For patients with established coronary heart disease (CHD), an LDL-C target of less than 2.6 mmol/L (100 mg/dL) is recommended, and when risk is considered very high, the recommended LDL-C target is less than 1.8 mmol/L (70 mg/dL). 5
Previous effectiveness studies have reported LDL-C attainment rates of 18% to 56%6–13 and the clinical benefits in patients who achieved better cholesterol control were parallel to those seen in clinical trials.12,13 These studies included patients with heterogeneous cardiovascular risk factors. Similar observational studies on goal attainment rates in patients with ACS are, however, limited, especially in Asia.
The objectives of this study were (1) to determine LDL-C goal attainment rates and predictors of goal attainment at 4 months, and (2) to describe prescription behavior that influences lipid goal attainment in Asian patients with ACS.
Methods
Study design and population
This is a single-center, retrospective observational study involving consecutive patients (> 18 years of age) admitted between January 2006 and July 2006, and discharged with a diagnosis-related group code of ACS or equivalent terms such as “acute myocardial infarction (MI)”, “ST elevation MI”, “unstable angina”, or “non-ST elevation MI”. Patients who died during admission and individuals who did not return for subsequent clinic visits were excluded from the analysis.
Lipid profiles were taken within 24 hours of the event, after a fast of at least 10 hours. Total cholesterol and triglycerides were measured directly by enzymatic methods. High-density lipoprotein cholesterol (HDL-C) was separated from LDL-C and very low density lipoproteins (VLDL) by precipitation and measured by enzymatic methods as for total cholesterol. LDL-C was calculated by Friedewald's equation. Lipid-lowering therapy was initiated during admission at the discretion of the physician, if there were no contraindications.
This study was reviewed and approved by the Singapore General Hospital Institutional Review Board. Informed consent was waived.
Data collection
Baseline demographics and characteristics of patients, LDL-C levels on admission and during follow-up visits, and lipid-lowering medications and dosages were extracted from electronic hospital medical records. Lipid-lowering statins were expressed as equipotent doses to simvastatin 10 mg according to Maron's formulation. 14
LDL-C goal attainment rates and prescription behavior measures
In high-risk patients with ACS, lipid goal attainment was defined as an LDL-C less than 1.8 mmol/L. LDL-C levels were determined at 4 months, 12 months, and at the end of the final follow-up period. In patients without lipid profiles at the defined time points, data closest to the specified time were used. Prescription behavior was assessed according to the equipotent doses of statins prescribed on discharge and the efforts made at dose-titration during follow-up.
Statistical analysis
Categorical data are presented as frequencies and percentages and continuous variables were reported as means ± standard deviation (SD).
Clinical predictors associated with LDL-C goal attainment were evaluated using both univariate and multivariate logistic regression. To avoid overfitting of the logistic model, the presence of 3 or more covariates including hypertension, diabetes mellitus, smoking, and previous history of coronary revascularization, were considered as a single variable coronary artery disease risk factor (CADRF). Odds ratios (OR) of LDL-C goal attainment at 4 months and their 95% confidence intervals (CI) were reported.
Multivariate analysis was performed to adjust for age at admission, sex, ethnicity, the presence of CADRF, and LDL-C level on admission.
Results were considered statistically significant if the P-value was < 0.05 on a 2-tailed test. Analyses were carried out using STATA/SE software version 11.0 for Windows (Stata Corporation, College Station, TX, USA).
Results
Patient characteristics
A total of 267 patients were included in the analysis, with a mean follow-up duration of 41.2 ± 10.7 months. There were more males (57%), and the mean age for all participants was 69 years. A large proportion of patients had a concomitant history of either hypertension (82%), diabetes mellitus (51%), or hyperlipidemia on statins prior to admission (49%). More than 30% of patients had a history of ischemic heart disease with previous coronary revascularization using either percutaneous intervention or coronary artery bypass. The mean LDL-C concentration on admission was 2.36 ± 0.97 mmol/L (Table 1).
Baseline characteristics of patients.

Pravastatin (n = 1), ezetimibe (n = 1), combination therapy (n = 2), fenofibrate (n = 3).
LDL-C attainment rates and predictors of goal attainment
Lipid goal attainment rate was highest at 4 months (36.7% at 3.7 ± 1.9 months), but declined progressively at 12 months (23.4% at 12.0 ± 3.1 months) and at the end of the follow-up period (14.5% at 41.2 ± 10.7 months). Among patients who achieved their LDL-C goal at 12 months (n = 40), 62.5% did so within the first 4 months of therapy. Similarly, among patients who achieved LDL-C goal at the end of follow-up (n = 27), 59.3% did so within the first 4 months of therapy (Fig. 1).
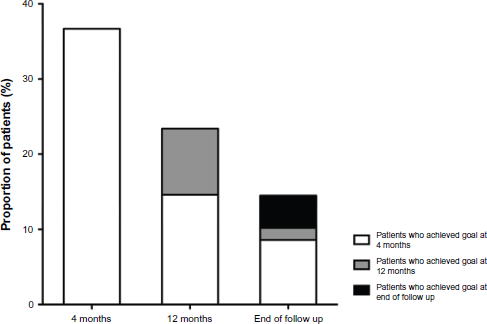
Proportion of patients who achieved low-density lipoprotein cholesterol (LDL-C) goals at 4 months, 12 months, and at the end of the follow-up period.
Univariate and multivariate logistic regression analyses showed that lower LDL-C levels on admission were predictive of LDL-C goal attainment at 4 months. For every 1 mmol/L increase in baseline LDL-C, the odds of attaining LDL-C goal in 4 months decreased by about 35%. Other factors were not associated with LDL-C goal attainment at 4 months (Table 2).
Univariate and multivariate analysis: clinical predictors to LDL-C goal attainment at 6 months.

CADRF is a single variable, defined by the presence of 3 or more of the following: hypertension, diabetes mellitus, smoking and previous history of coronary revascularization.
Prescription behavior
Most patients were prescribed simvastatin (76.0%) or lovastatin (8.6%) on discharge. The mean equipotent dose of statins prescribed on discharge in the entire cohort was 1.57 ± 0.87, with more than 85% of patients receiving an equipotent dose of 2 (corresponding to simvastatin 20 mg) or less.
Among patients who had not achieved lipid goals, the dose of statins prescribed was low throughout the follow-up duration. Specifically, the equipotent doses prescribed at 4 months, 12 months, and at the end of follow-up were 1.53 ± 0.89, 1.58 ± 0.93, and 1.90 ± 1.10, respectively.
In the first year, the equipotent dose of statins remained relatively unchanged. This was likely the result of failing to up-titrate doses in more than 2/3 of patients, as well as an increased rate of discontinuation and down-titration of statins in the first year (Fig. 2). Although there were more aggressive efforts in up-titration at the end of follow-up period, more than 1/3 of patients did not have their statin doses changed since hospital discharge. On the other hand, the rates of discontinuation and down-titration of statins remained stable at the end of follow-up duration (Fig. 2). This resulted in a higher mean equipotent dose of statins at the end of follow-up period, but not sufficient to impact goal attainment.
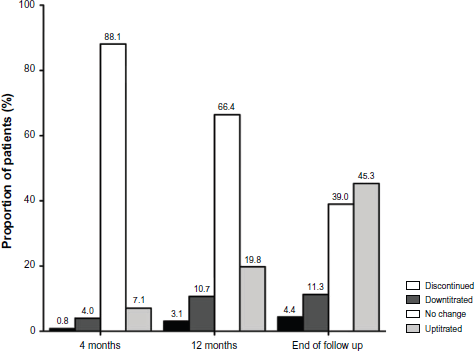
Distribution of statin dose titration in patients who did not achieve low-density lipoprotein cholesterol (LDL-C) goals.
Discussion
Patients with ACS are at very high risk for cardiovascular events. Compared to patients with few or no cardiovascular risk factors, patients at higher risk have lower rates of LDL-C goal attainment.6–10 In patients with ACS, the LDL-C attainment rates are even lower. 11
In our study, 36.7% of patients attained the LDL-C target of less than 1.8 mmol/L at 4 months. Patients were prescribed low potency statins, and in those who did not achieve LDL-C goal, a responsive dose-titration was usually not performed. Our study showed that for patients who achieved lipid goals at 1 year and at the end of the follow-up period, most did so within the first 4 months. Furthermore, a higher baseline LDL-C reduced the likelihood of attaining lipid goals, a finding that is also consistent with previous studies.6,8,10,13 These results suggest that an intensive lipid-lowering strategy should be considered at the start of therapy to achieve LDL-C goals early.
The challenges of achieving cholesterol treatment goals had led to the development of specific lipid-lowering strategies. One treatment approach is to treat all high-risk patients with an intensive highdose, lipid-lowering statin instead of treating to a lipid target. 15 This strategy is consistent with a meta-analysis suggesting that the mortality benefits of aggressive lipid lowering in high-risk patients are evident regardless of baseline LDL-C levels. 16 Although intensive high-dose statin therapy has been well tolerated in clinical trials, its use in a more heterogenous group of patients in the real-world setting, especially in Asians, is not commonly practiced.
One of the primary concerns of intensive therapy relates to potential adverse effects with high-dose or more potent statins. Although numerous large-scale clinical trials have demonstrated statin efficacy and safety in a variety of populations, studies evaluating the efficacy and safety of statins in Asians are limited. Another concern relates to the higher cost of newer and more potent statins. This is particularly true in a public healthcare system such as Singapore. Generic medications are usually prescribed, as they are made more affordable by patients after substantial government subsidies. However, with the current availability of potent generic statins, cost will be less of an issue if such a management strategy is considered.
Another strategy utilizes dosing algorithms to select an initial starting dose of a potent statin based on the patient's baseline LDL-C level and/or cardiovascular risk.17–22 Clinical studies have demonstrated that a significant proportion of patients were able to achieve their LDL-C goal within 4 to 6 weeks using this strategy.17–22 In addition, the rate of adverse events was relatively low. However, one major limitation of these studies was the short duration of follow-up; because of this, long-term outcomes could not be assessed.
Regardless of the treatment strategy adopted, aggressive lipid-lowering therapy should always be balanced by a consideration of the adverse effects associated with such treatment. This risk-benefit profile has to be assessed individually.
Limitations
There are several limitations to our study. This was a retrospective study, thus we were not able to ascertain factors such as medication compliance or reasons for down-titration, discontinuation, or failure to up-titrate statin doses. The combined rates of down-titration and discontinuation were about 15% at the end of follow-up, compared to down-titration rates of 3% to 40% and discontinuation rates of 1% to 18% reported in other studies.6–11 These low rates of down-titration and discontinuation reflect the low statin doses prescribed and the diligence in medication adherence. We postulate that the reasons for discontinuation and down-titration were due to side effects of statins, while the failure or delay in up-titrating statin dose could be due to either the patient's reluctance to have medications changed or the physician's behavior in treating to target goals.
In addition, although the characteristics of the study population are similar to patients in many Asian countries, we acknowledge that the results reflect clinical practice from a tertiary center, and that may be different compared to other countries or clinical settings.
Conclusions
A large proportion of patients with ACS did not achieve LDL-C levels of less than 1.80 mmol/L. These individuals constitute a challenging group of high-risk patients for aggressive secondary prevention. LDL-C goal attainment can be improved with more potent or high-dose statins and responsive dose-titration. This can be achieved through formal lipid management programs.
Author Contributions
Conceived and designed the experiments: RST, TTL. Analyzed the data: FG. Wrote the first draft of the manuscript: CWC. Contributed to the writing of the manuscript: FG, TTL, RST. Agree with manuscript results and conclusions: CWC, FG, TTL, RST. Jointly developed the structure and arguments for the paper: CWC, FG, TTL, RST. Made critical revisions and approved final version: CWC, FG, TTL, RST. All authors reviewed and approved of the final manuscript.
Funding
This study was supported by an education grant by Pfizer, Inc.
Competing Interests
The author(s) disclose no potential conflicts of interests.
Disclosures and Ethics
As a requirement of publication author(s) have provided to the publisher signed confirmation of compliance with legal and ethical obligations including but not limited to the following: authorship and contributorship, conflicts of interest, privacy and confidentiality and (where applicable) protection of human and animal research subjects. The authors have read and confirmed their agreement with the ICMJE authorship and conflict of interest criteria. The authors have also confirmed that this article is unique and not under consideration or published in any other publication, and that they have permission from rights holders to reproduce any copyrighted material. Any disclosures are made in this section. The external blind peer reviewers report no conflicts of interest.
Footnotes
Acknowledgements
This manuscript has been edited by BioMed Proofreading.
