Abstract
This systematic review assessed the impact of atrial fibrillation (AF) and pharmacotherapy on health-related quality of life (HRQOL) in elderly patients. Highly prevalent in the elderly, AF is associated with morbidity and symptoms affecting HRQOL. A PubMed and EMBASE search (1999-2010) was conducted using the terms atrial fibrillation, elderly, quality of life, Medicare, and Medicaid. In all, 504 articles were identified and 15 were selected (studies examining pharmacotherapy [rate or rhythm control] and HRQOL in AF patients with a mean age > 65 years). Information, including study design, cohort size, and HRQOL instruments utilized, was extracted. Five observational studies, 5 randomized trials comparing rate and rhythm control, 3 randomized trials investigating pharmacologic agents, and 2 trials examining HRQOL, depression, and anxiety were identified. Elderly AF patients had reduced HRQOL versus patients in normal sinus rhythm, particularly in domains related to physical functioning. HRQOL may be particularly affected in older AF patients. Although data do not indicate whether a pharmacologic intervention or single treatment strategy—namely rate versus rhythm control—is better at improving HRQOL, either of these strategies and many pharmacologic interventions may improve HRQOL in elderly AF patients. Based on reviewed data, an algorithm is suggested to optimize HRQOL among elderly patients.
Introduction
Atrial fibrillation (AF) is the most common cardiac arrhythmia seen in clinical practice. 1 Each year in the United States, AF contributes to approximately 5 million office visits, 500,000 outpatient and emergency department visits, and 350,000 hospitalizations. 2 Atrial fibrillation predominantly affects the elderly population. Approximately 70% of AF patients are between 65 and 85 years of age, with a median age of 75 years. 1 The high prevalence of AF in the elderly has been associated with age-related changes within the atrial myocardium and conducting tissues3,4 and with structural heart disease. 5 The prevalence of AF increases with advancing age; approximately 5% of the population older than 65 years and almost 8% of the population older than 80 years are affected.1,6,7 Because of the aging of the US population and improved survival of patients with hypertension, diabetes, and heart failure,4,8–10 the projected prevalence of AF is about 8 million Americans by the year 2050. 11
Atrial fibrillation is associated with significant morbidity and mortality, resulting in an increased risk of stroke, thromboembolism, chronic heart failure (CHF), and sudden death.12–15 Older age is an independent risk factor for stroke4,16 and strokes attributable to AF tend to be associated with a higher mortality rate and level of disability.9,12 Furthermore, patients demonstrate a greater cognitive decline in the presence of AF compared with patients without arrhythmia.17,18 Stroke prevention is critical in patients with AF and has received prominent attention with the introduction of newer anticoagulants. Most elderly patients with chronic AF do not develop stroke even after 30 years of follow-up, 19 yet all AF patients may have impaired health-related quality of life (HRQOL).
The negative effect that aging has on HRQOL is a highly relevant issue, in particular when considering management options because most patients consider HRQOL to be the most important outcome of therapy. 20 Many patients experience considerable impairment from symptoms and report lower HRQOL levels, increased illness intrusiveness, less than total functional capacity, and lower global life satisfaction than do healthy individuals or asymptomatic patients. 21 The 36-Item Short Form Health Survey (SF-36) is a health scale that measures HRQOL across numerous domains, including Physical Functioning, Role-Physical, Bodily Pain, General Health, Vitality, Social Functioning, Role-Emotional, and Mental Health. 22 Patients with AF report significantly below-average HRQOL scores on various SF-36 domains, falling between patients with CHF and myocardial infarction in terms of the level of HRQOL impairment. 21
Over the last 20 years, hospital admissions for AF have increased by 66%. This is attributable to several factors, including an aging population, the increased prevalence of chronic heart disease, and increased diagnosis rates owing to more frequent electrocardiogram monitoring.1,6,23 As health care utilization increases in AF patients, HRQOL is negatively affected. 24
The impact of AF on HRQOL has not been extensively evaluated and many studies that do assess HRQOL have significant limitations. The majority of studies examining HRQOL in patients with AF and following either rate or rhythm control strategies are conducted in highly symptomatic patients, highly selected patient populations, or subgroups of clinical trial patients, populations with the potential for bias and inadequate statistical power.25,26 Interventions studied have included ablation and pacing procedures, the Maze procedure, pulmonary vein isolation, internal or external cardioversion, different pacing modalities, and pharmacologic therapy. To date, most of the studies examined the impact of ablation procedures on HRQOL, with relatively few studies assessing the impact of pharmacologic therapies that are used in the majority of AF patients.
A previous systematic review found that HRQOL in the segment of the general population diagnosed with AF was inferior to that of healthy controls, the general population, and patients with coronary heart disease. The same study found that both rate and rhythm control strategies had a beneficial effect on HRQOL. 25 As aging and its associated disorders (eg, hypertension, stroke) independently reduce HRQOL, the greater prevalence of AF in the elderly warrants a better understanding of the impact of AF on HRQOL for this specific patient population. Reductions in HRQOL seen in aging populations complicate any analysis of a potential relationship between an asymptomatic disorder such as hypertension and HRQOL in elderly patients. Generally, decreased HRQOL with age is seen in almost all areas, with older patients with hypertension reporting more stress, worries about health, and difficulties with coping. 27 To date, AF clinical trials examining HRQOL issues in the elderly versus younger patient populations are not available. This is further compounded by the lack of a generally accepted definition for the term elderly, either chronologically or physiologically. However, an age of 65 years or greater is generally accepted as a reasonable chronologic definition. A better understanding of the impact of AF on HRQOL in elderly patients and the influence of various pharmacotherapies on HRQOL may help physicians to better determine an individual patient's optimal treatment plan.
Methods
Objective
This systematic review evaluated HRQOL in patients with AF and assessed the effects of pharmacologic interventions on HRQOL, with a focus on older patients (mean age > 65 years).
Search strategy and study selection
A literature search limited to studies conducted in humans and published in English between January 1, 1999, and September 27, 2010, was performed using PubMed and EMBASE. Both Medical Subject Heading (MeSH) terms and text word searches were used in the following search strategy (MeSH terms identified):
(Atrial Fibrillation [MeSH] OR paroxysmal atrial fibrillation OR persistent atrial fibrillation OR permanent atrial fibrillation OR chronic atrial fibrillation OR lone atrial fibrillation OR silent atrial fibrillation OR atrial fibrillation/drug therapy [MeSH]) AND (quality of life [MeSH] OR well being OR personal satisfaction [MeSH] OR palpitation) AND (rate control OR rhythm control OR intervention OR pharmacologic treatment OR non-pharmacological interventions) AND (elderly OR aged [MeSH] OR Medicare [MeSH] OR Medicaid [MeSH])
Once articles were identified as eligible for inclusion, the reference lists from those articles were reviewed to identify any additional studies meeting inclusion criteria.
Inclusion and exclusion criteria
Observational HRQOL studies, non-interventional HRQOL studies, and randomized clinical trials examining pharmacotherapy (rate or rhythm control) and HRQOL in AF patients with a mean age of 65 years or greater were included. To be all-inclusive, studies assessing HRQOL through any HRQOL measurement instruments were eligible for inclusion, as were studies with any duration of follow-up. Studies that did not examine HRQOL were excluded, as were studies examining the effects of radiofrequency ablation and pacing therapy, internal or external direct current cardioversion, implantable defibrillators on HRQOL, or arrhythmia surgery.
Quality assessment and data abstraction
Each study eligible for inclusion was then reviewed to record the duration of the study, size of cohort, study sample representation of overall patient population, study design, and HRQOL instruments utilized. Limitations of each study were assessed based on information included in each article's discussion section.
Results
The literature search identified 504 English-language citations (Fig. 1) published between January 1999 and September 2010. Removal of duplicate records left 379 citations. After reviewing the titles and abstracts of the 379 citations based on the inclusion and exclusion criteria, 357 were excluded. Thereafter, 22 full-text articles were assessed for eligibility, 6 were added based on review of the full-text article bibliographies, and 13 were excluded because HRQOL was not measured.
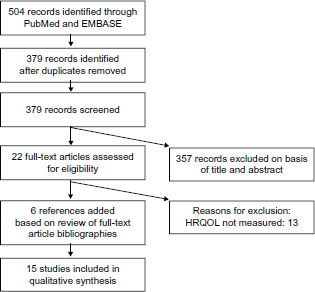
Literature search flow diagram.
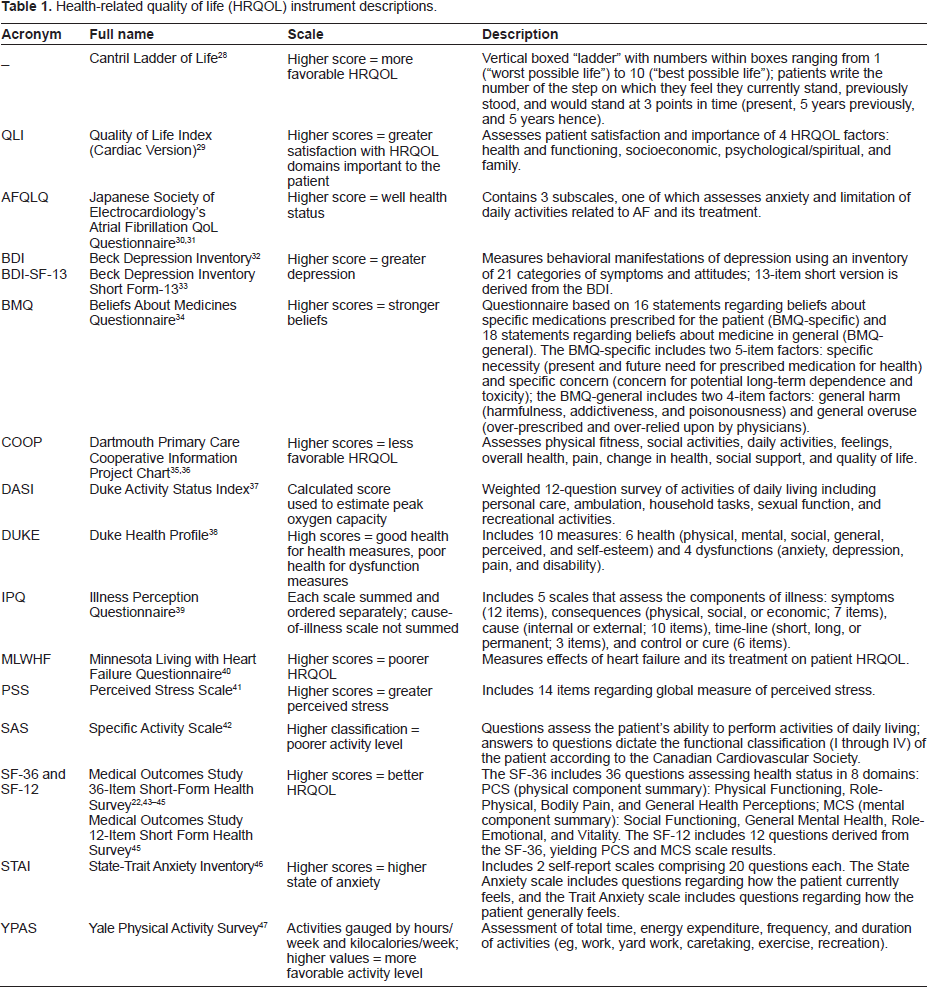
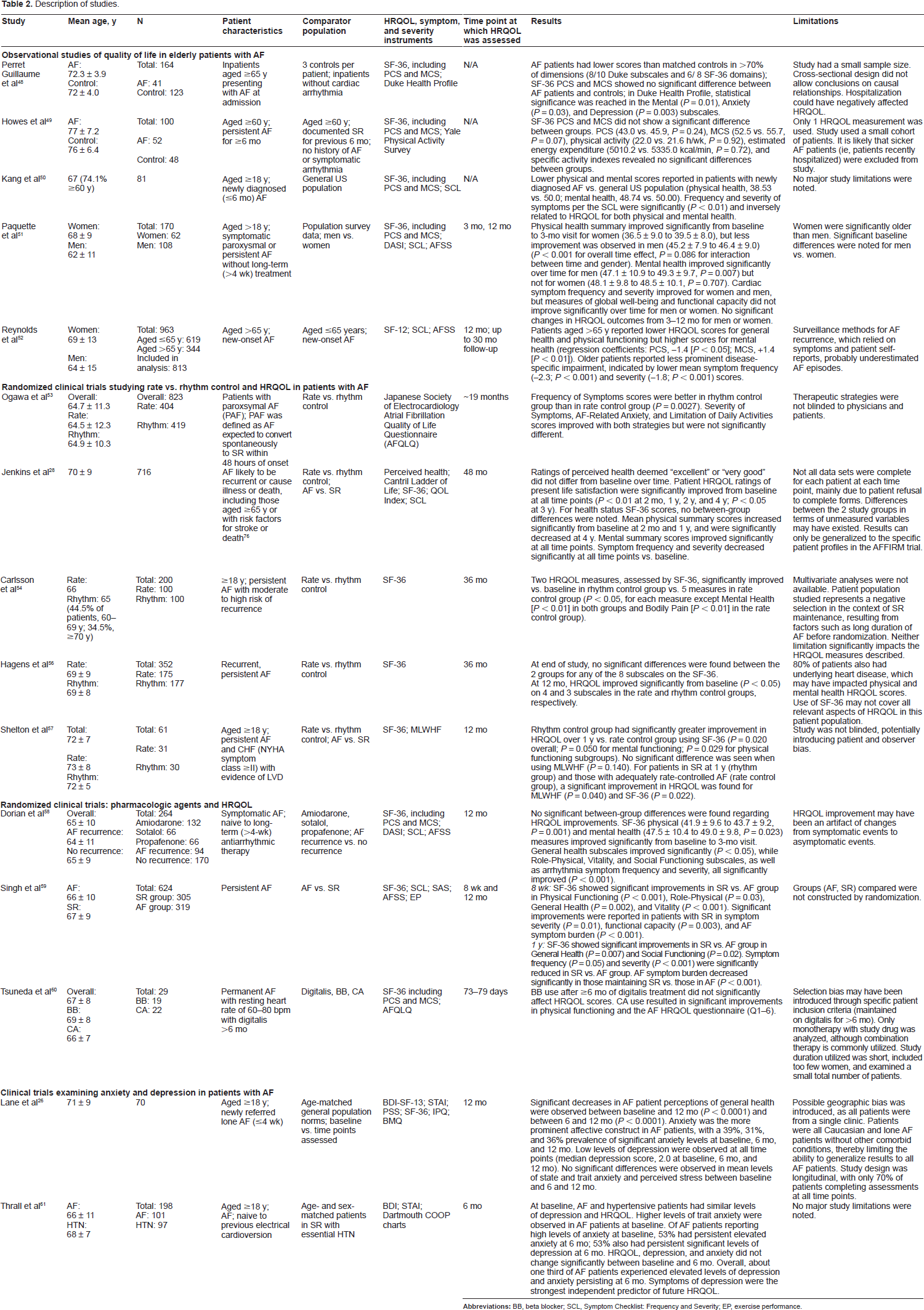
A total of 15 studies remained and were included in this review: 5 observational studies, 5 randomized trials comparing effects of rate versus rhythm management strategies on HRQOL, and 3 randomized trials investigating the effects of specific pharmacologic agents on HRQOL. Additionally, 2 clinical trials examining HRQOL, depression, and anxiety in patients with AF, as well as the relationship between depression/anxiety and HRQOL, were included. The most common instrument utilized for HRQOL assessment in these 15 studies was the SF-36, including the physical component summary (PCS) and mental component summary (MCS) scales. The SF-36 measures health status in 8 domains: Physical Functioning, Role-Physical (limitations due to physical problems), Vitality, Mental Health, Role-Emotional (limitations due to emotional problems), Social Functioning, Bodily Pain, and General Health. A description of all the HRQOL instruments used in these studies is provided in Table 122,28–47 and summary data from the 15 studies are presented in Table 2.
Health-related quality of life (HRQOL) instrument descriptions.
Description of studies.
Observational quality-of-life studies in elderly patients with AF
A cross-sectional study (part of the CliniQual Vie Program) compared HRQOL in elderly inpatients with AF aged 65 years or older (n=41) with that of age-matched controls (n = 123). 48 Two HRQOL tools were utilized: the SF-36 and the Duke Health Profile questionnaire. The Duke Health Profile questionnaire measures HRQOL through 6 health measures—physical, mental, social, general, perceived health, and self-esteem—and 4 dysfunctions: anxiety, depression, pain, and disability. 38 After adjustment for coronary artery disease or chronic respiratory failure, patients with AF had lower scores than matched controls in more than 70% of dimensions, namely 8 of 10 Duke Health Profile subscales and 6 of 8 SF-36 domains. Differences reached statistical significance for the Mental (P = 0.01), Anxiety (P = 0.03), and Depression (P = 0.003) subscales in the Duke Health Profile. No significant differences were observed between the 2 groups in the PCS or MCS of the SF-36. Limitations of this study include the small sample size, a cross-sectional design which did not allow conclusions to be made regarding causal relationship, and the cohort of hospitalized patients because hospitalization itself may have had negatively impacted HRQOL.
In another study, patients with AF (n = 52) reported overall HRQOL scores similar to those of population controls in normal sinus rhythm (SR) (n = 48). The PCS and MCS scores revealed a trend toward lower scores in patients with AF compared with controls, but differences did not achieve statistical significance (PCS, 43.0 versus 45.9, P = 0.24; MCS, 52.5 versus 55.3, P = 0.07). 49 However, significantly lower scores were reported in AF patients for the Social Functioning (77.16 versus 89.32, P = 0.01) and Role-Emotional (82.05 versus 94.44, P = 0.01) domains. There was no difference in scores in the Physical Functioning domain, as assessed by the Yale Physical Activity Survey, which estimates the amount of time an individual spends performing physical activities and equivalent kilocalorie expenditures. 47 This study was limited by reduced power due to a small cohort size and the fact that sicker AF patients (ie, those who were recently hospitalized) were excluded. Patients in this study had a mean age of 77 years; these patients are generally less active and could perhaps have fewer cardiovascular demands than younger patients.
Another study observed lower physical and mental scores in patients with newly diagnosed AF (n = 81) compared with the general US population (physical health, 38.53 versus 50.0; mental health, 48.74 versus 50.00). 50 The frequency and severity of symptoms (as reported on the Symptom Checklist: Frequency and Severity scale) were inversely related (P < 0.01) to HRQOL for both physical and mental health, although causality could not be determined. This study had a relatively focused population of newly diagnosed elderly patients; however, it had reduced power due to small sample size and lack of follow-up.
The
A multivariate analysis of composite HRQOL in 813 patients from the
Randomized clinical trials studying rate versus rhythm control and HRQOL in elderly patients with atrial fibrillation
The Japanese Rhythm Management Trial for Atrial Fibrillation 53 (J-RHYTHM) was a randomized comparative study of patients with paroxysmal AF treated with either rate control (n = 442) or rhythm control strategies (n = 443). Patient HRQOL, a secondary endpoint, was assessed using the Japanese Society of Electrocardiology's Atrial Fibrillation Quality of Life Questionnaire (AFQLQ). This questionnaire comprises 3 subsets, including 26 questions regarding the occurrence frequency of 6 symptoms (palpitations, dizziness, shortness of breath, chest discomfort, irregular pulse, and pulse deficit), the severity of these symptoms, and anxiety and limitation of daily activities related to AF and AF treatment.30,31 Frequency of Symptoms subset scores were better in the rhythm control group (P = 0.0027), whereas Severity of Symptoms, AF-Related Anxiety, and Limitation of Daily Activities subset scores improved with both treatment strategies and were not significantly different between groups. In this study, therapeutic strategies were not blinded to physicians and patients, which could have led to bias. As Japanese patients may differ physiologically and/or culturally compared with patients in other countries, the results of this study may not be generalized to other patient populations.
A substudy of the
The open, randomized, pilot
In the
A randomized, controlled study of rate versus rhythm control in patients with chronic AF and CHF (CAFÉ-II study) found HRQOL to be significantly improved in the rhythm control group (n = 30) versus the rate control group (n = 31) after 1 year, as assessed by overall (P = 0.020), MCS (P = 0.050), and PCS (P = 0.029) scores on the SF-36 scale. 57 The greatest improvements occurred when SR was maintained, leading to the conclusion that restoring SR in patients with CHF may improve HRQOL compared with a rate control strategy. Post hoc analyses of patients achieving adequate treatment response (defined as SR and adequate rate control) with their respective treatment strategies found significant differences using both measurements. This study contributed prospective as well as post hoc data on a specific patient population—those with AF patients and comorbid CHF. However, the study was limited by its small sample size and unblinded study design.
Randomized clinical trials: pharmacologic agents and HRQOL in elderly patients
In the CTAF study (n = 264), 58 significant improvements in both physical and mental health HRQOL occurred over the first 3 months of therapy regardless of whether patients were assigned to amiodarone, sotalol, or propafenone (PCS: 41.9-43.7, P = 0.001; MCS: 47.5-49.0, P = 0.023), but no differences in HRQOL improvements were observed between treatment groups. 58 However, AF burden was improved to a greater extent in patients treated with amiodarone compared with sotalol or propafenone (P = 0.001, interaction between time and treatment). No significant differences in HRQOL scores were noted at 3 and 12 months between treatment groups. Patients had relatively new AF without exposure to long-term antiarrhythmic therapies; therefore, the investigators were able to study a population without the bias of drug resistance or nonresponse. However, the small sample size diminished the power to compare HRQOL assessments over time or between treatment groups.
Another study examined HRQOL and exercise performance in patients with persistent AF converted to SR (n = 305) compared with patients remaining in or reverting to AF (n = 319).
59
Patients were randomized to receive amiodarone, sotalol, or placebo as part of the
In a crossover study, AF patients treated with digitalis who had a resting heart rate between 60 and 80 beats per minute (bpm) for more than 6 months (n = 29) were randomized to monotherapy with either a beta blocker or a calcium antagonist (CA). 60 Patients consenting to continue (n = 12) were switched to the other therapy; efficacy was determined once adequate heart rate was achieved for at least 1 month. Patients received beta blocker therapy for a mean duration of 79.3 ± 34.5 days and CA therapy for a mean duration of 72.8 ± 27.4 days. Calcium antagonists, but not beta blockers, showed improvement over digitalis baseline therapy in the Role-Physical domain of the SF-36 scale (48.6 versus 54.1, P < 0.05), as well as in the Frequency and Severity of Symptoms domain of the AF HRQOL Questionnaire (15.1 versus 17.0, P < 0.05). The authors concluded that CA, rather than beta blocker monotherapy, may be preferable to digitalis to improve HRQOL in permanent AF patients. However, this study was limited by a small, predominantly male (n 25/29) sample and a brief study duration. In addition, the narrow selection of patients treated with digitalis may limit generalizability of the findings. The study of monotherapy rather than combination therapy may also have influenced the results.
Clinical trials examining anxiety and depression in patients with atrial fibrillation
HRQOL, depression, and anxiety during the first 12 months following a diagnosis of lone persistent or permanent AF (N = 70) were examined in a study. 26 Low levels of depression were reported by patients, and although SF-36 domain scores were lower in AF patients compared with age-matched population controls (with the exception of Mental Health over time), no significant differences in HRQOL (with the exception of General Health) were reported over 12 months. Anxiety was more prevalent than depression—it was reported by 38.5%, 30.9%, and 35.7% of patients at baseline, 6 months, and 12 months, respectively, with no significant differences between time points. This was one of the few studies evaluating the effects of AF on anxiety and depression in addition to HRQOL; however, the study population comprised patients from a single center, all of whom were Caucasian, had AF, and were without comorbid conditions.
Another study found that patients with AF (n = 101) and hypertension (n = 97) reported similar levels of depression and HRQOL; however, AF patients exhibited significantly higher anxiety scores (37.4 versus 33.3, P = 0.02). 61 Quality of life, depression, and anxiety scores were unchanged at 6 months. Overall, approximately one third of AF patients had elevated levels of depression and anxiety that persisted at 6 months. Depression and anxiety were both significantly associated with a patient's perceived HRQOL (P < 0.001), and improvement in depression symptoms was a significant predictor of HRQOL scores at 6 months (P < 0.001). Although this study was the first to examine the association between the affective status of AF patients and HRQOL, it was limited by its small sample of AF patients recruited from a single cardiology clinic.
Discussion
The data examined in this systematic review suggest that HRQOL may be particularly affected in older patients. An algorithm drawn from these data to aid in decision-making is provided to optimize HRQOL among elderly patients with AF (Fig. 2). The algorithm describes the HRQOL parameters that should be assessed in all patients with AF. The 2010 Canadian Cardiovascular Society AF Guidelines also recommend that assessment of patient quality of life be part of the evaluation of every patient with AF. 62

An algorithm designed to optimize HRQOL among elderly patients with AF.
Five observational studies with a predominantly elderly population were reviewed. In one study, 49 no statistically significant difference between patients with AF and controls in overall HRQOL was observed. However, based on several methodological weaknesses, the relevance of this study is low. Interestingly, this was the only study with a population aged greater than 75 years. Three other studies reported decreased HRQOL in the elderly with AF compared with age-matched48,51 and general population 50 controls. The FRACTAL subanalysis 52 indicated slightly lower HRQOL scores (General Health and Physical Functioning domains) in patients older than 65 years compared with those aged 65 years or less.
Two clinical trials26,61 examined anxiety and depression in addition to HRQOL. Both studies were substantially limited by their designs, primarily the small, single-center patient populations. While they yielded interesting data on anxiety and depression and correlations with HRQOL, changes in HRQOL scores were not significantly different from those of controls.
Several clinical trials (AFFIRM, STAF, RACE, J-RHYTHM, and others28,53,54,56,57) examined the effects of 2 different treatment strategies—rate control and rhythm control—on HRQOL in patients with AF. While the AFFIRM and STAF trials reported significant improvements from baseline in patients treated with both rhythm and rate control strategies, these trials reported no significant changes in HRQOL between treatment groups.28,54 Results in the RACE trial revealed significant improvements in several components of HRQOL in the rate control group, but not in the rhythm control group, when compared with baseline scores. 56 Conversely, investigators reported significant improvement in HRQOL in the rhythm control group versus the rate control group, but only in a specific patient population with comorbid CHF. 57 In the J-RHYTHM study, only the Frequency of Symptoms subset scores were better in the rhythm control group than in the rate control group; Severity of Symptoms, AF-Related Anxiety, and Limitation of Daily Activities improved with both strategies and were not significantly different between groups. 53
The effects of various pharmacologic agents to positively influence HRQOL in patients with AF was explored in 3 randomized clinical trials. Agents included amiodarone, sotalol, propafenone, digitalis, beta blockers, and CAs. In the CTAF trial, amiodarone was not significantly different from sotalol in measures of HRQOL; however, AF burden was improved to a greater extent in patients treated with amiodarone than those treated with sotalol or propafenone. This is in contrast to results of the SAFE-T trial, where patients in SR showed significant improvement in HRQOL regardless of treatment group (amiodarone or sotalol). Several domains of the HRQOL questionnaire improved following use of CAs—but not beta blockers—when compared with digitalis therapy. 60 This study, however, was limited by its small sample size and biased patient selection and is therefore of limited relevance.
Other recent randomized clinical trials and registries add important data for the management of patients with AF. The J-RHYTHM study was designed to determine the optimal strategic approach for AF patients (rate or rhythm control) and emphasized patient-reported experience and perception of AF-specific disability. This study showed that in patients with paroxysmal AF, the primary outcome— first occurrence of all-cause mortality, symptomatic cerebral infarction, systemic embolism, major bleeding, heart failure hospitalization, or physical/ psychological disability requiring alteration of treatment strategy—was significantly reduced with a rhythm control strategy compared with a rate control strategy (P = 0.0128). 53
Results from a substudy of the RACE II trial 63 suggest that the method of rate control (strict [n = 207] or lenient [n = 230]) has no difference on HRQOL in patients with permanent AF. Strict rate control was defined by a resting heart rate of less than 80 bpm and heart rate during moderate exercise of less than 110 bpm. Lenient rate control was defined as a resting heart rate of less than 110 bpm. 64 HRQOL was assessed using the SF-36 scale, the AF severity scale, the Minnesota Living with Heart Failure (MLHF) questionnaire, and the Multidimensional Fatigue Inventory-20 (MFI-20) at baseline, 1 year, and study conclusion. Over a median follow-up of 3 years, results of the MLHF questionnaire and AF severity scale did not differ between lenient and strict rate control groups from the baseline to the end of study. In the SF-36 scale, Physical Functioning domain scores decreased in both the lenient (P = 0.01) and strict control groups (P = 0.04) by study completion. A history of heart failure and an age older than 75, but not strict or lenient rate control, were associated with decreased HRQOL. These data show that changes in HRQOL appear to be influenced by age, symptoms, gender, and underlying heart disease rather than a strict or lenient rate control strategy. 63
Registry data are valuable since they represent real-life situations. Two large registries containing HRQOL data—RECORD-AF (Registry on Cardiac Rhythm Disorders Assessing the Control of Atrial Fibrillation)65,66 and AFFECTS (The Atrial Fibrillation: Focus on Effective Clinical Treatment Strategies) 67 —document that the initial treatment strategy assigned by cardiologists to the majority of AF patients is rhythm control.65,67 This preference decreased with age in both registries. When compared with patients assigned a rate control strategy, patients managed with a rhythm control strategy were more frequently symptomatic and more likely to have recently diagnosed AF.65,67 In the RECORD-AF registry, a rate control strategy was more frequently chosen for heart failure, valve disease, or persistent AF, 65 and in the AFFECTS registry, patients in the rate control group tended to be older, had a longer mean duration of AF, and were more likely to have a family history of AF. 67 At 1 year, 81% of RECORD-AF patients treated with a rhythm control strategy were in SR compared with only 33% treated with a rate control strategy. Additionally, only 13% of rhythm control patients progressed to permanent AF compared with 54% of rate control patients. 65 Therapeutic success—defined as SR for patients receiving a rhythm control strategy or a resting heart rate lower than or equal to 80 bpm with a rate control strategy, no crossover of treatment strategy, and no incidence of clinical outcomes 68 — was significantly different between patients assigned a rhythm control (60%) and a rate control strategy (47%). 65 Rhythm control patients were more likely to be hospitalized for arrhythmias and rate control patients were more likely to be hospitalized for heart failure, but the difference in total number of clinical events between these patients did not reach statistical significance at 1-year follow-up in RECORD-AF. 65 In the AFFECTS registry, there were too few deaths and cardiovascular-related events for a meaningful comparison between rhythm control and rate control strategies. 67
Data obtained from RECORD-AF and AFFECTS provide a “real-world” look at a physician's choice of treatment for the management of patients with AF. Registry data are valuable because randomized controlled trials often do not completely represent real-life situations. RECORD-AF data demonstrate that when physicians are able to select a treatment strategy, they are successful at meeting the goals of that strategy 81% of the time. Results from registries such as these can provide important insight to physician-based management strategies, particularly treatment strategies for AF patients with decreased HRQOL.
Limitations
This analysis is limited by several factors. The study duration varied considerably in the studies analyzed, ranging from 6 months to 4 years. Many of the studies assessing HRQOL were subanalyses of larger clinical trials and, with the exception of the AFFIRM substudy, 28 may not have had adequate statistical power. Many of these studies used generic HRQOL questionnaires, most commonly the SF-36 scale, which may not detect subtle but significant HRQOL changes in patients with AF. In addition, statistically significant changes in HRQOL may have gone unobserved between treatment groups using non-disease-specific questionnaires, as elderly populations are likely to have comorbidities. Importantly, no studies have specifically examined HRQOL in an exclusively elderly population; therefore, this review focused on trials wherein the mean population age was 65 years.
Future Directions
Both aging and AF are associated with impaired HRQOL and more data are needed on HRQOL in the elderly AF population. Since few interventions have shown decreased morbidity and mortality rates, the treatment of AF in clinical trials remains focused on controlling symptoms and improving HRQOL. Thus, HRQOL is a critical issue in the treatment of AF as well as in the study of new therapies for AF.
New treatment strategies may improve HRQOL in patients with AF. Dronedarone is an antiarrhythmic agent approved by the US FDA for the management of patients with AF. Dronedarone is indicated in the US to reduce the risk of hospitalization for AF in patients in SR with a history of paroxysmal or persistent AF. 69 In the post-marketing setting, cases of hepatocellular liver injury and hepatic failure in patients receiving dronedarone have been reported, including 2 reports of acute hepatic failure requiring transplantation and new-onset or worsening heart failure.70,71 The revised prescribing information recommends obtaining periodic hepatic serum enzymes, especially during the first 6 months of treatment. 69 Cases of increased international normalized ratio with or without bleeding events have also been reported in patients on warfarin and dronedarone.69,72 Dronedarone is contraindicated in patients with symptomatic heart failure showing recent decompensation requiring hospitalization or NYHA class IV heart failure, as well as in patients with AF who will not or cannot be cardioverted into normal SR. 69
Vernakalant (intravenous formulation) has been approved in the European Union, Iceland, and Norway for the rapid conversion of recent-onset AF to SR in nonsurgical adult patients with AF of 7 days or less and for postcardiac-surgery adult patients with AF of 3 days or less. 73 In the AVRO study (A Phase III Superiority Study of Vernakalant vs. Amiodarone in Subjects With Recent Onset Atrial Fibrillation), treatment with vernakalant resulted in a significantly greater improvement in patient perception of state of health (as measured by the EQ-5D QOL assessment visual analog scale) at hour 2 compared with amiodarone (mean adjusted increase from baseline of 10.9 points vs. 5.6 points; P = 0.0006, respectively). 74 Further studies are needed to compare these treatment options with currently available agents as well as to assess any potential effects on HRQOL. Additionally, the impact of AF type on HRQOL has not been well studied as it is confounded by comorbidities and type of treatment.
A validated, AF-specific HRQOL questionnaire would be ideal in enabling clinicians to make well-informed decisions regarding treatment strategies for patients with AF (ie, rate or rhythm control). Recently, the 20-item long Atrial Fibrillation Effect of Quality-of-Life (AFEQT) questionnaire was developed and validated in a prospective observational study. 75 This instrument provides a 4-item symptoms score, an 8-item daily activities score, a 6-item treatment concerns score, and a 2-item treatment satisfaction score. In contrast to generic HRQOL instruments, disease-specific instruments allow patients to quantify the extent to which their limitations are attributable to a specific disease. In this study, AFEQT was shown to be reliable as indicated by the high Cronback α coefficients, valid by demonstrating adequate convergent and divergent correlations, and sensitive in discriminating the severity of patients' AF. These preliminary findings support its use in following patients with AF; however, future studies with this instrument will provide an understanding of how to best evaluate the efficacy of AF therapy and the quality of care for AF patients. 75 HRQOL information from large registries also provides a representation of real-world AF management. The data in this review indicate that HRQOL should be strongly considered when designing AF treatment strategies for individual patients, especially the elderly.
Conclusions
Atrial fibrillation (AF) is a chronic illness disproportionately affecting the elderly population. An increase in prevalence is anticipated over the next several decades. Because of the broad range of symptoms and sequelae associated with AF, health-related quality of life (HRQOL) is of increased interest to clinicians caring for this patient population. The articles selected for this systematic review demonstrate that HRQOL is impaired to some degree in all patients with AF and may particularly affect older patients. Although the data do not demonstrate a particular pharmacologic intervention to be superior over others at improving HRQOL, many pharmacologic interventions have been shown to improve HRQOL in the elderly patient with AF. More research is warranted to address the limitations of the available data. HRQOL-specific registry data will also be important to provide real-world information. As new therapies become available, further research regarding effect on patient HRQOL is warranted to allow clinicians to select therapies that take into account the physical, emotional, and social well-being of patients.
Acknowledgments
Editorial support was provided by Vrinda Mahajan, PharmD, of Peloton Advantage, LLC, Parsippany, NJ.
Author Contributions
Conceived and designed the experiments: CJP. Analysed the data: CJP. Wrote the first draft of the manuscript: VM. Contributed to the writing of the manuscript: CJP. Agree with manuscript results and conclusions: CJP. Jointly developed the structure and arguments for the paper: CJP. Made critical revisions and approved final version: CJP. The author reviewed and approved the final manuscript.
Funding
Editorial support was funded by Sanofi-Aventis U.S. The opinions expressed in the current article are those of the author. The author received no honorarium or other form of financial support related to the development of this manuscript.
Competing Interests
Carl J. Pepine, MD, has received grant support through the University of Florida from Baxter Healthcare, Berlex Labs/Bayer HealthCare, Boehringer Ingelheim, Cardium Therapeutics, Daiichi Sankyo, Lilly, Forest Laboratories, GlaxoSmithKline, Novartis, Pfizer, Reliant, Sanofi-Aventis, Schering-Plough, and Viron Therapeutics. He has provided consultancy services for Abbott Laboratories, Angioblast Systems, Athersys, Baxter Healthcare, Boehringer Ingelheim, Gilead, Medtelligence, NicOx, Pfizer, Sanofi-Aventis, Schering-Plough, Servier, and Slack.
Disclosures and Ethics
As a requirement of publication, the author has provided to the publisher signed confirmation of compliance with legal and ethical obligations including but not limited to the following: authorship and contributorship, conflicts of interest, privacy and confidentiality and (where applicable) protection of human and animal research subjects. The author has read and confirmed his agreement with the ICMJE authorship and conflict of interest criteria. The author has also confirmed that this article is unique and not under consideration or published in any other publication, and that he has permission from rights holders to reproduce any copyrighted material. Any disclosures are made in this section. The external blind peer reviewers report no conflicts of interest.
