Abstract
Mechanical loading provides indispensible stimuli for growth and development of the articular cartilage and bone. Interestingly, depending on loading conditions loads applied to the joint can be beneficial as well as harmful to skeletal maintenance and remodeling. Moderate loads to the synovial joint, for instance, suppress the expression levels of matrix metallproteinases (MMPs), while loads above a threshold tend to increase their destructive activities. This report focuses on two recently developed loading modalities from animal studies, joint motion and joint loading. Their unique characteristics and potential usages for maintenance of the articular cartilage and stimulation of bone remodeling are reviewed. Also described are biophysical and molecular mechanisms which likely are responsible for the load-driven maintenance of cartilage and bone, and a possibility of developing load-mediated treatments of osteoporosis and osteoarthritis.
Loading Modalities
In vitro studies allow for well-controlled experimental loading conditions to be applied to chondrocytes. The effects of mechanical loads on the biological response of articular cartilage have been investigated using cell culture systems incorporating a broad range of mechanical stimuli. 2 In those in vitro studies using cultured chondrocytes, the expression of matrix metalloproteinases (MMPs) such as MMP-1 and MMP-13 were altered by dynamic and static mechanical stimuli in an intensity-dependent manner. Moderate shear stress (2–5 dyn/cm2) reduced MMP expression levels, while high shear stress (10–20 dyn/cm2) increased them. Similarly, moderate hydrostatic pressure (1–5 MPa) suppressed MMP-1 expression, while higher loads (10 MPa) elevated it. 10 These in vitro results indicated an important role of mechanical stimulation in the regulation of MMPs in the articular cartilage. However, these conditions may be significantly different to stimuli experienced by chondrocytes in vivo. Therefore, animal models are better positioned to investigate the physiological relevance of mechanical stimuli in the maintenance of cartilage and the regulation of MMPs. This article focuses on two in vivo modalities used to stimulate chondrocytes in articular cartilage of diarthrodial joints in small animal models. The principle of these in vivo modalities lies on applying moderate stimulus to chondrocytes via motion (joint flexion) or loading (compressive force) to the joints such as the knee and elbow.
Joint Motion
This controlled joint flexion modality was developed to investigate the role natural physiological joint motions play in the maintenance of the articular cartilage. 1 Flexion of the knee joint produced by a computer controlled system is similar to the natural knee motion of the animal hindlimb (Fig. 1). Note that the natural motion induces not only rotation but also translation (sliding) of the articular cartilage at the interface of the femur and the tibia. This joint motion device is able to select loading parameters such as rotation angle (30–150°), angular speed, and motion frequency in Hz (typically 0.1–1 Hz). To evaluate the effects of joint motion, two additional conditions can be employed. First, joint motion can be combined with axial loading of the limb, which presumably increases the level of compressive and shear stresses to the articular cartilage. Second, the absence of motion (joint immobilization) can be used as a negative control.

Joint motion.
Joint Loading
In the joint loading modality, mild forces are applied in the medio-lateral direction in the form of cyclic loading, typically for 3–5 min per day at 1–20 Hz, to major synovial joints such as the knee (knee loading), elbow (elbow loading), and ankle (ankle loading)3,4 (Fig. 2). In the knee loading model, for instance, loads are transmitted to both the distal femur and the proximal tibia. Loading effects such as enhanced bone formation and accelerated fracture healing are observed throughout the lengths of the femur and the tibia. 5 The required magnitude of loads for joint loading is in general smaller than that for axial loading (e.g. 0.5 N for elbow loading and 2–3 N for ulna axial loading in mice). Bone is less stiff in a lateral direction than an axial direction.
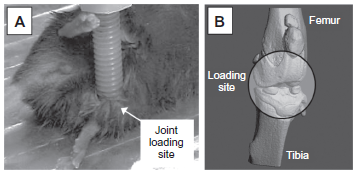
Joint loading.
According to a currently proposed mechanism, joint loading does not require load-driven in situ strain for enhancing bone remodeling. It has been proposed that joint loading periodically alters the pressure in the medullary cavity and activates molecular transport in a lacunocanalicular network in cortical bone. 6 That is, a pressure gradient in the medullary cavity generates oscillatory fluid flow in the porous bone cortex. It has been speculated that induced fluid flow then enhances molecular transport in the lacunocanalicular network and applies shear stress to osteocytes residing in lacunae. 7 Modulation of the intramedullary pressure with knee loading is exerted throughout the length of the tibia and the femur. Thus, this joint loading modality has potential for stimulating bone remodeling at a location distant from the loading site at a joint.
Load-Driven Effects on Articular Cartilage and Bone
Results from joint motion and joint loading demonstrated that mechanical loads to the joint can have a significant impact on maintenance of the articular cartilage and remodeling of bone. In particular, based on in vivo data collected from animal models the two loading modalities described above can be potentially beneficial to the preservation of joint tissues and the prevention of bone loss if rotation and loading conditions are properly selected.
Maintenance of Articular Cartilage with Joint Motion
Moderate joint motion can suppress inflammatory responses and reduce tissue degeneration caused by MMPs.
Suppression of inflammatory responses—Proinflammatory cytokines such as IL-1β upregulate the expression and activity of MMP-1 and MMP-13. It has been shown using cultured chondrocytes that mechanical stimulation, given in a form of fluid flow shear stress, can suppress the IL-1β-induced upregulation of MMP-1 and MMP-13. 2 In accordance with those in vitro results, joint motion in vivo is able to reduce inflammatory responses in a murine collagen-induced arthritis model. Additionally, in an antigen-induced arthritis model in rabbits, continuous passive motion suppressed transcription of IL-1β and synthesis of inflammatory mediator COX-2 and MMP-1. These mechanical signals also induced IL-10 synthesis, suggesting that moderate joint loading can generate anti-inflammatory signals. 8
Reduction in tissue degeneration—It has been shown that a lack of joint motion (immobilization) increases the expression and activity of MMP-3. 9 Interestingly, passive movement of these immobilized limbs suppresses expression of this degradative enzyme and prevents histologically detectable decreases in cartilage matrix integrity. 10 However, the same flexion of the joint in the presence of axial loads (5 N) increased the level of MMP-13 mRNA and its activity. 1
In an osteoarthritis model in which the misalignment of articular surfaces is a cause of damage and degradation of the articular cartilage, normal joint motion is hardly achieved. Thus, although studies with moderate exercise in osteoarthritis patients show a beneficial effect with joint motion and loading, it is important to evaluate a potential outcome of joint motion for individual osteoarthritis patients.
Stimulation of Bone Remodeling with Joint Loading
Animal studies using mice and rats have demonstrated that joint loading can stimulate bone formation, accelerate wound healing, and facilitate lengthening of long bones. 11
Bone formation—Joint loading can increase bone formation throughout the length of long bones. Knee loading, for instance, is capable of elevating the rate of new bone formation in the tibia and the femur. 12 Bone formation was observed not only at the site close to the knee but also in the proximal femur and the distal tibia. In those areas distant from the loading site, strain on the periosteal surface by joint loading was in the order of 10 µ strain.
Wound healing—Joint loading was also able to accelerate healing of bone wounds. In a healing study in which surgical holes were created in the cortical shaft of the tibia and the femur, joint loading was shown to increase the closure rate of these surgically generated holes. 13
Bone lengthening—When knee loading was applied to one leg, the loaded tibia and femur were reported to be longer than the non-loaded contralateral bones. Histological analysis revealed that in response to knee loading, the number of cells in the growth plate of the proximal tibia increased and their cellular shape was altered. 14 The result suggests a possibility of using knee loading for treating limb length discrepancies in children.
Although the effects of joint loading on bone have been well documented, its potential effect on the articular cartilage is less clear. For a case of osteoarthritis in which misalignment of the articular cartilage induces an irregular joint motion, an intriguing question is whether joint loading is capable of providing moderate mechanical stimulation and reducing activities of proteolytic enzymes. Recent data indicate that like joint motion, moderate loads applied with joint loading can decrease the expression level of MMP-1 and MMP-13 (unpublished data). Furthermore, it is important to evaluate the effects of loading on other joint tissues such as the meniscus and synovium. In our previous study, we reported that moderate mechanical stimulation to synovial cells reduced the mRNA level of MMP-13. 15
Signaling Pathways
Many studies have been conducted to identify signaling pathways in the mechanotransduction of bone and joints.16,17 In this article we highlight data for CITED2-and NF-κB-mediated transcription for joint motion, and genome-wide analysis and an integrated stress response for joint loading.
Cited2-Mediated Transcription
CITED2 (CBP/p300-interacting transactivator with ED-rich tail 2) is a member of the CITED family of nuclear regulators. It is inducible by various stimuli including lipopolysaccharide, hypoxia, and cytokines. 18 It has been shown in cultured chondrocytes that CITED2 mRNA and protein levels are increased by moderate flow shear (5 dyn/cm2), intermittent hydrostatic pressure (1–5 MPa), and joint motion, which down-regulated MMP-1 and MMP-13 expression levels as well as enzyme activities. 2 The induction of CITED2 in vivo by physiologic loading was correlated with the downregulation of MMP-1 and the maintenance of cartilage matrix integrity. 10 Consistent with the above observations, overexpression of CITED2 repressed MMP-1 and MMP-13 mRNA levels and activities. 2 Competitive binding and transcription assays demonstrated that CITED2 suppresses MMP-1 expression by displacing MMP transactivator Ets-1 from its coactivator p300. Moderate mechanical stimulation specifically phosphorylates the p38δ isoform, which is required for CITED2 upregulation. 10 Together, these studies suggest that CITED2 plays a critical role in mediating the anticatabolic effects of moderate loading.
NF-κB, a transcription factor induced by pro-inflammatory mediators such as IL-1β and TNF-α, regulates transcription of multiple genes including pro-inflammatory cytokines and mediators nitric oxide synthase (NOS-2), COX-2, MMPs, TNF, and IL-1.19–23 It is reported that IL-1β-induced transcriptional activity of NF-κB can be blocked by biomechanical signals by interfering with multiple steps in the NF-κB signaling cascade. 24 In chondrocytes and bone cells, it has yet to be investigated whether crosstalk exists between the CITED2 and NF-κB pathways.
Genome-Wide Analysis
Using tibia samples with and without ankle loading, signaling pathways linked to joint loading were predicted from whole-genome microarray data. 25 Four pathways highlighted in the analysis, include phosphoinositide 3-kinase (PI3K), extracellular matrix (ECM)-receptor interactions, TGF-β signaling, and Wnt signaling. First, various extracellular signals activate PI3K, which affects cellular growth and movement. Second, ECM-receptor interactions include interactions of molecules including many types of collagens, integrin, fibronectin, and laminin. Third, TGF-β signaling is involved in embryogenesis, angiogenesis, inflammation, and wound healing, and is important to development and maintenance of cartilage and bone metabolism. Lastly, Wnt signaling is one of the central pathways in bone metabolism and mechanotransduction. Mice with nonfunctional Lrp5 receptor in this pathway respond poorly to mechanical loading with significant reduction in bone mass.
Integrated Stress Response
An integrated stress response is caused by a number of cellular insults including hypoxia, nutrient deprivation, viral infection, oxidation, and stress to the endoplasmic reticulum. 26 Whether mechanical loading can suppress or induce the integrated stress response is largely dependent on the loading intensity. This stress response leads to translational de-activation by a mechanism involving phosphorylation of eIF2α, with preferential translational activation of a particular set of proteins linked to cellular survival or apoptosis. In cultured chondrocytes, administration of thapsigargin and tunicamycin induces stress to the endoplasmic reticulum, which triggers an integrated stress response. 27 In this response, the level of phosphorylated eIF2α was elevated together with the expression of MMP-13. Interestingly, joint loading reduced the level of phosphorylated eIF2α by suppressing activity of Perk, one of the four known eIF2α kinases. 28 In contrast, unloading a hindlimb by tail suspension increased the level of eIF2α phosphorylation. 29 Thus, existing data supports the notion that mechanical stimulation in a proper loading condition can be a suppressor of Perk-mediated integrated stress responses and cell death.
Future Studies and Concluding Remarks
Existing studies support the beneficial role of mechanical loading to joints. Joints are a complex anatomical structure, which delicately integrate articular cartilage and subchondral bone together with meniscus and synovium. 30 It is necessary to evaluate a potential outcome of moderate mechanical intervention in connection to osteoarthritis and osteoarthritis-related cartilage lesions. With regard to potential clinical applications of joint motion and joint loading for the maintenance of the articular cartilage and the enhancement of bone remodeling, several questions for future studies are offered:
Does joint motion alter the expression and activities of aggrecanases? To prevent tissue degeneration in articular cartilage, it is important to regulate not only MMPs but also a family of ADAMTS (a disintegrin and metalloproteinase with thrombospondin motifs). In osteoarthritis, for instance, suppression of the activity of ADAMTS-4 (aggrecanase 1) and ADAMTS-5 (aggrecanase 2) appears to be critically important for its treatment.31,32
Does joint loading provide tissue protection in the articular cartilage? Osteoarthritis is often caused by misalignment of articulating surfaces, and for those patients joint motion does not protect their cartilage tissue. Although a primary application of joint loading has been directed at increasing bone mass, it also deforms the articular cartilage. An open question is whether joint loading in a proper mechanical condition has a possibility to provide cartilage protection. It is also important to evaluate the stress/strain distributions in response to joint motion and joint loading (Fig. 3).
Does mechanical loading of joints activate molecular interactions between the articular cartilage and the subchondral bone? Homeostasis of the articular cartilage is affected through interactions with the subchondral bone underneath the cartilage. For instance, both MMPs and ADAMTS need to be post-translationally activated, and this activation process is regulated by many factors including MMPs themselves and many proteoglycans. 33 It has not yet been investigated whether mechanical loading to joints regulates activities of MMPs and ADAMTS through interactions between the articular cartilage and the subchondral bone.
What is a molecular switch that controls an outcome towards stimulatory or inhibitory responses? This molecular switch may act at the level of transcriptional, translational, or post-translational regulation. Some of the molecules involved in this switch include CITED2 as a transcription regulator, eIF2α as a general translational factor, and syndecan 4 as an activator of ADAMTS-5. 34 It is possible that multiple elements comprise an integrated molecular switch in response to loads to the joint.

Micro CT image of mouse knee.
In summary, mechanical loading to joints can be beneficial to prevent tissue degeneration in the articular cartilage and strengthen bone. Since loading effects are sensitive to loading conditions, determination of an appropriate loading procedure is important. Answering the above questions and identifying key signaling pathways should aid in developing load-mediated treatments of bone and joint diseases such as osteoporosis and osteoarthritis.
Disclosure
This manuscript has been read and approved by all authors. This paper is unique and is not under consideration by any other publication and has not been published elsewhere. The authors and peer reviewers of this paper report no conflicts of interest. The authors confirm that they have permission to reproduce any copyrighted material.
Footnotes
Acknowledgements
The authors appreciate critical reading of the manuscript by George Malacinski and Daniel Leong, and thank funds from National Institutes of Health, AR52743 (HS), AG034198 (LC), AR52144 (HY), and National Science Foundation, NSF MRI0723027 (LC).
