Abstract
Interleukin (IL)-10 overexpression inhibits joint inflammation, however the effect of high local concentrations of IL-10 on chondrocyte homeostasis remains unclear. The aim of this study was to determine the effects of IL-10 overexpression on cartilage matrix production in three-dimensional (3D) chondrocyte cultures.
Human articular chondrocytes were transduced with adenoviral vectors alone (adv/empty) or by vectors either overexpressing enhanced green fluorescence protein (adv/EGFP) or human IL-10 (adv/hIL-10) before their transfer to a 3D culture system. Non-transduced chondrocytes were used as controls. The expression of IL-10 or EGFP was confirmed using ELISA or flow cytometry. Chondrocytes synthesis of collagen types II and I, aggrecan, fibronectin and β1-integrin was determined over a period of 14 days post transduction using flow cytometry or immunohistochemistry.
adv/EGFP or adv/IL-10 transduced chondrocytes expressed EGFP or secreted IL-10 detectable over the 2 weeks culture period. No suppression of collagen type II, aggrecan or β1-integrin synthesis by IL-10 overexpression was found and the deposition of collagen type I and fibronectin remained unaffected compared to the controls.
IL-10 overexpression does not impair key features of chondrocytes differentiated phenotype (e.g. collagen type II and aggrecan expression) suggesting the potential use of IL-10 for gene therapeutic approaches in the joint.
Introduction
The immunomodulatory cytokine (IL-10) is a promising anti-inflammatory agent for treating inflammatory joint diseases. Several experimental in vivo studies revealed that the application of recombinant IL-10 or IL-10 overexpression vectors in the joint reduced inflammation (Joosten et al. 1997; Lechman et al. 1999; 2003; Neumann et al. 2002; Van de Loo and van den Berg, 2002; Zhang et al. 2004; Kuroda et al. 2006). However, the direct effects of IL-10 on articular chondrocyte homeostasis remain still unclear. The application of viral vectors in the joint does not usually lead to the direct transduction of articular cartilage chondrocytes but rather it acts directly on the cells of the synovialis since the vectors do not penetrate the dense extracellular cartilage matrix surrounding the chondrocytes (Cucchiarini and Madry, 2005). Therefore, high virus doses are necessary for in vivo transduction strategies. This approach requires the dissiminated transduction of adjacent tissues in the joint; systemic distribution of the vectors in the patient and systemic adverse effects of the vector such as immunosuppression (Van de Loo and Van den Berg, 2002; Lechman et al. 2003). For these reasons, ex vivo transfection strategies are necessary to achieve sufficient IL-10 or anabolic growth factors overexpressing chondrocytes which might be implanted in cartilage defects as a strong localized transgene source to inhibit post-traumatic joint inflammation and to support cartilage repair (Gelse et al. 2003; Gelse and Schneider, 2006). Adenoviral overexpression of IL-10 might be a tool to achieve initial high and sustained local levels of IL-10, which can not be efficiently achieved using recombinant IL-10. However, adenoviral vectors produce only transient transgene expression as the cells usually loose the transgene during cell proliferation (Dinser et al. 2001). The 3D chondrocyte culture systems mimic more closely cartilage in vivo compared with monolayer cultures since monolayer chondrocytes usually rapidly dedifferentiate and loose their specific synthetic capacities (Benya and Shaffer, 1982; Bonaventure et al. 1994; Häuselmann et al. 1994; Martin et al. 1999; Schulze-Tanzil et al. 2004). Expression of cartilage-specific collagen type II is usually rapidly down-regulated in monolayer culture. The synthesis of cartilage-specific collagen type II and cell-matrix receptors β1-integrins which mediate the essential chondrocyte matrix interaction are the precondition for chondrocyte survival (Hirsh et al. 1997; Cao et al. 1999; Yang et al. 1999). IL-10 is involved in extracellular matrix remodelling in other connective tissue cell types (Reitamo et al. 1994; Yamamoto et al. 2001; Moroguchi et al. 2004). 3D culture systems might be a useful tool to store autologous chondrocytes in vitro for in vivo implantation in cartilage defects. The aim of the present study is to determine the direct effects of IL-10 overexpression on chondrocyte extracellular matrix production in 3D culture.
Materials and Methods
Construction of a Recombinant Adenovirus Expressing Human il-10
A derivative of human adenovirus serotype 5 was used as the source of viral DNA backbone. This modified adenoviral vector backbone contains a deletion of bp 355–4021, resulting in a loss of the E1a, E1b, and protein IX polypeptides. In addition, there is a deletion of bp 28,592–30,470, which results in a loss of 1.9 kb of DNA from the E3 region. The EGFP and human IL-10 transgenes contain the full-length translated region and the human cytomegalovirus immediate early enhancer/promoter (CMV). The cDNA constructs were produced using standard recombination methods. Additionally, a recombinant adenovirus containing an empty gene expression cassette was constructed for use as control. The particular procedure is published elsewere (Graham and Prevec, 1995; Minter et al. 2000).
Chondrocyte culture
Primary human articular chondrocytes were isolated enzymatically as previously described (Schulze-Tanzil et al. 2002, 2004). Non-arthritic human femoral head articular cartilage which was macroscopically free from osteoarthritic alterations (obtained from four patients between the age of 50–80 years undergoing joint replacement surgery for femoral neck fractures) was cut into small slices followed by digestion with 1% pronase deriving from Streptomyces griseus at 10 mg/mL in Ham's F-12 (7 units/mg, Roche Diagnostics GmbH, Mannheim, Germany) for 2 hrs at 37 °C and subsequently with 0.2% (v/v) collagenase type I deriving from Clostridium histolyticum at 2 mg/mL (266 units/mg, Sigma-Aldrich, Munich, Germany) for 4 hrs at 37 °C. Each donor tissue was strictly handled separatively and used for independent experiments. Isolated cells were resuspended in growth medium (Ham's F-12/Dulbecco's modified Eagle's medium [50/50] containing 10% fetal calf serum [FCS], 50 IU/mL streptomycin, 50 IU/mL penicillin, 2.5 μg/mL amphotericin B, essential amino acids, L-glutamine [all: Biochrom-Seromed, Munich, Germany]), 25 μg/mL ascorbic acid [Sigma-Aldrich, Munich, Germany], and cultured at a concentration of 2.5 × 10 6 cells/mL in alginate culture (Sigma-Aldrich, Munich, Germany) as previously described (Schulze-Tanzil et al. 2004). After a few days of culture, some chondrocytes migrated from the alginate forming a monolayer (Schulze-Tanzil et al. 2002, 2004; Oberholzer et al. 2007). These cells were seeded at 20 000 cells/cm2 in 6-well plates in monolayer culture, incubated at 37 °C with 5% CO2 for 24 hrs in serum-reduced growth medium (0.5% FCS as a minimum to allow chondrocyte growth) before using in transduction experiments.
Cfda-se Proliferation assay
Chondrocytes deriving from three independent cartilage donors were incubated for 15 min in the dark with 5 μM/mL 5-(6) carboxyfluorescein diacetate succinimidylester (CFDA-SE) solution (Molecular Probes, Invitrogen, Ca, USA) at 37 °C. After washing in PBS the excessive staining was removed, the cells seeded in 6-well plates at 20 000 cells/cm2 or embedded in alginate and further cultivated for 2–8 days.
Adenoviral Transduction of Chondrocytes and 3d culture
Chondrocytes were either transduced with a replication-deficient recombinant adenoviral vector producing EGFP (adv/EGFP), human IL-10 (adv/hIL-10), or nothing (adv/empty) at 5000 virus particles/cell for 4 hrs at 37 °C in 5% CO2 or remained non-transduced (controls). 24 hrs post transduction, adenovirally transduced chondrocytes were either embedded in alginate or introduced in high-density culture and cultured for further 4 weeks. For preparation of high-density cultures 8 μl of a chondrocyte cell sediment was pipetted onto a membrane filter with a pore diameter of 0.2 μm (Sartorius, Göttingen, Germany) on the top of a stainless steel grid at the medium-air interface in a Petri dish (Schulze-Tanzil et al. 2002). The EGFP expression rate in the alginate cultured chondrocytes was evaluated flow cytometrically in three independent experiments using chondrocytes derived from three human donors.
Quantification of il-10 Secretion by ELISA
Culture supernatants of alginate embedded chondrocytes deriving from two human cartilage donors were collected and tested in 4 independent ELISA experiments for chondrocytes IL-10 release using a commercially available human IL-10 ELISA kit (BDOptEIA, BD Bioscience, Heidelberg, Germany) according to the manufacturer's instructions.
Flow Cytometric Analysis of Collagen Type ii and β1-Integrin expression
Alginate was depolymerized after 2, 5, 7 or 14 days of alginate culture using 50 mM EDTA/10 mM HEPES solution. Chondrocytes were washed with PBS and resuspended in staining buffer (1% bovine serum albumine/PBS/0.01% NaN3) and phenotypically analysed using primary antibodies for rabbit-anti-human collagen type II (1:30 in staining buffer) (Chemicon) or mouse-anti-human β1-integrin (1:30 in staining buffer) (Chemicon) and their respective isotype control (mouse IgG1, 1:30 in staining buffer, Caltag) for 30 min on ice. After washing the cells (2% fetal calf serum/PBS/100 U/mL polymixin B sulfate) cells were immunolabelled with donkey-anti-rabbit FITC- or allophycocyanine (APC)-coupled secondary antibodies (Dianova) diluted 1:200 in staining buffer for 30 min on ice. Cells were washed and fluorescence was evaluated using a FACSCalibur flow cytometer and CellQuest software (Becton Dickinson, Heidelberg, Germany). Further flow cytometric data analysis was performed using FlowJo software for PC (Tree Star Inc., Ashland, Ore., USA). Chondrocytes deriving from three different human donors were used for six (collagen type II) or three (β1-integrin) independent experiments.
Indirect immunofluorescence
For immunofluorescence microscopy, alginate spheres and high-density cultures were embedded in O.C.T. (optimal cutting temperature) embedding medium (Sakura, Finetech, U.S.A.) and 4 μm thick cryosections were prepared. After fixation using 4% paraformaldehyde in phosphate buffered saline (PBS) for 15 min, cryosections were washed with tris buffered saline (TBS: 0.05 M Tris, 0.015 M NaCl, pH 7.6), overlaid with protease-free donkey serum (5% diluted in TBS) for 10 min at room temperature (RT) rinsed and incubated with primary antibodies (rabbit anti-human-collagen type I [1:40], rabbit anti-human-collagen type II [1:40] rabbit anti-human-aggrecan [1:40] [all: AcrisAntibodies Hiddenhausen, Germany], mouseantihuman-fibronectin [Dianova, Hamburg, Germany, 1:30], mouse-anti-human-β1-integrin [1:30] [Chemicon, International, Temecula, CA, USA] and respective isotype control antibodies [1:30 or 1:40] [Caltag, Hamburg, Germany] diluted in TBS in a humid chamber 1 hr at RT. Cells were subsequently washed with TBS before incubation with donkey-anti-rabbit-fluoresceinisothiocyanate (FITC) (Dianova) or donkey-anti-mouse-Cyanine-3 (Cy3) (Invitrogen, Ca, USA) coupled secondary antibodies (diluted 1:200 in TBS) for 1 hr at RT. Cell nuclei were counterstained using 4′,6′-diamidino-2-phenylindol (DAPI) and mounted with fluoromount mounting medium. Three independent experiments with chondrocytes deriving from three different human cartilage donors were performed.
Statistical analysis
Results were expressed as the mean and standard error of mean (mean ± SEM). Differences between experimental groups were considered significant at p < 0.05 as determined by Student's t-test.
Results
Proliferation of Chondrocytes Decreases in Alginate Compared with Monolayer culture
CFDA-SE proliferation assay was used to compare proliferation of chondrocytes in monolayer and alginate culture for a time period of 8 days. As expected the CFDA-SE staining intensity of the cells decreased with culturing time due to the chondrocyte proliferation, however, the loss of staining intensity was more pronounced in the monolayers compared with the alginate cultures indicating a higher proliferation rate in the monolayer chondrocytes (Fig. 1). At day 8 a slight second faster proliferating chondrocyte population was evident in alginate (Fig. 1, arrow).

Proliferation of chondrocytes in monolayer and alginate culture. CFDA-SE proliferation assay and flow cytometry were used to compare proliferation of chondrocytes in monolayer (red) and alginate culture (blue) for a time period of 8 days. Arrow: a small second more rapidly proliferating chondrocyte population was evident in the alginate culture on day eight.
EGFP Expression Rate in Adenovirally Transduced Chondrocytes Cultured in Alginate and High-Density Culture Differs to that in Monolayer culture
Adv/EGFP transduced chondrocytes were either embedded 24 hrs post transduction in alginate beads; introduced in high-density culture or remained in monolayer culture for at least 4 weeks. Cells from the monolayer or the alginate embedded chondrocytes were investigated for the EGFP expression rate using flow cytometry analysis, on a daily basis during the first week, followed by weekly analysis for the remaining 3 weeks in culture. 48 hrs post transduction, a transduction efficacy of 92,74% and 66,49% was evident in the monolayer and alginate embedded chondrocytes, respectively (Fig. 2). The percentage of EGFP expressing cells was significantly higher for the monolayer chondrocytes which was maintained until day 4 (p < 0,001). However, at days 7 (p = 0,043) and 14 (p < 0,001) the alginate embedded cells revealed significantly higher expression rates for EGFP compared to the monolayer cultured cells.

EGFP expression rate in adenovirally transduced chondrocytes cultured in alginate compared with monolayer culture. Chondrocytes were adenovirally transduced using an EGFP overexpressing vector and then either embedded 24 hrs post transduction in alginate beads or cultured in monolayer for 2 weeks. At day 2, 4, 6, 7 and 14 monolayer or alginate cultured chondrocytes were investigated for the EGFP expression rate using flow cytometric analysis.
Cryosections of the high-density cultures were investigated for fluorescence intensity. Most chondrocytes were EGFP positive at day 2 (Fig. 3A) however the fluorescence intensity decreased gradually (Fig. 3A-D) during the 14 days culture period.
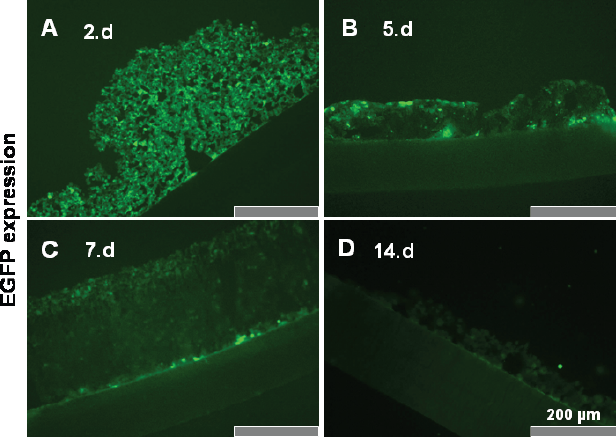
EGFP expression in high-density culture. Chondrocytes were adenovirally transduced using an EGFP overexpressing vector and then embedded 24 hrs post transduction in high-density culture. Cryosections of these cultures were investigated at day 2 (A), 5 (B), 7 (C), and 14 (D) for EGFP expression using fluorescence microscopy. Scale bars 200 μm.
Chondrocytes Transduced with the il-10 Overexpressing Vector and Cultured in Alginate Secreted High Levels of il-10 for Several Weeks as Determined by ELISA
Chondrocytes overexpressing IL-10 and embedded in alginate beads 24 hrs post transduction released high levels of IL-10 with a peak at 48 hrs post transduction. In the non-transduced control cultures, no IL-10 secretion was detectable using the ELISA assay since the IL-10 secretion was below the detection minimum (sensitivity ≥7 pg/mL). IL-10 secretion by IL-10 transduced chondrocytes decreased after day 2 but was still elevated after 2 weeks post transduction (Fig. 4).
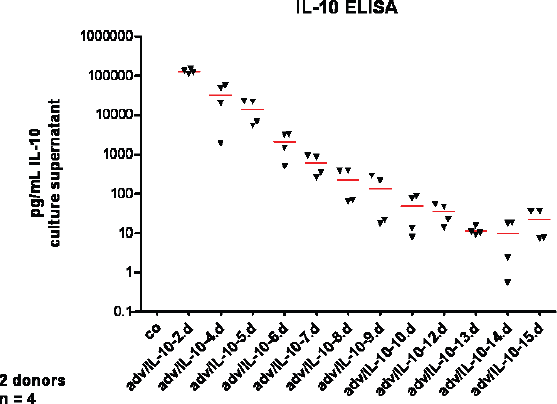
IL-10 release into the culture supernatant by IL-10 overexpressing chondrocytes embedded in alginate for two weeks. Chondrocytes deriving from two different cartilage donors were either adenovirally transduced using an IL-10 overexpression vector (adv/IL-10) or remained non-transduced (co: control) before they were embedded in alginate 24 hrs later. IL-10 secretion in the culture supernatant was measured during two weeks using a human IL-10 ELISA.
IL-10 Overexpression Did Not Impair β1-Integrin and Collagen Type ii Expression by Chondrocytes Embedded in alginate
Chondrocytes remained either non-transduced (control) or were transduced with the adv/empty or the adv/hIL-10. Flow cytometric analysis was used to semiquantify the expression of the cartilage specific matrix protein collagen type II and the cell matrix receptor β1-integrin on chondrocytes surface at days 2, 5, 7 and 14. Despite secretion of high amounts of human IL-10 by IL-10 transduced chondrocytes in the alginate cultures, cell membrane associated collagen type II (Fig. 5A) and β1-integrin (Fig. 5B) expression were not significantly affected during the two weeks culture period, compared with the non-transduced or the adv/empty chondrocytes. The mean fluorescence intensity for collagen type II revealed only a slight non significant increase from days 2 to 5, irrespective of the treatment course. The adenoviral transduction with the empty or the IL-10 vector had no effect on the mean fluorescence intensity of β1-integrins, compared with the non-transduced controls (Fig. 5B). The β1-integrin expression (MFI and percentage of integrin positive cells) was higher at day 5 compared to day 2 and peaked at day 7 in relation to levels at day 14, irrespective of the treatment course.

Cell surface collagen type II and β1-integrin expression in IL-10 transduced and non-transduced chondrocytes embedded in alginate. Expression of the cell membrane attached collagen type II (A) and the β1-integrins (B) by chondrocytes which were either adenovirally transduced with an IL-10 overexpression vector (adv/IL-10), an empty vector (adv/empty) or remained non-transduced (co: control) and embedded in alginate for 14 days was evaluated by immunolabelling with specific antibodies and flow cytometric analysis. The mean fluorescence intensities (MFI) are shown.
Collagen type II, aggrecan, collagen type I, and fibronectin expression by chondrocytes embedded in alginate or high-density culture was not affected by IL-10 transduction
Indirect immunofluorescence labelling with specific antibodies directed against important extracellular matrix proteins such as collagen type II, aggrecan, collagen type I and fibronectin was performed using cryosections of alginate (Fig. 6) and high-density cultures (Fig. 7) at days 2, 7 and 14. At day 2 transduced and non-transduced chondrocytes produced cartilage-specific collagen type II (Fig. 6A1-C1) and the matrix protein aggrecan (Fig. 6A3-C3), but higher amounts of collagen type I (Fig. 6A5-C5) and fibronectin (Fig. 6A7-C7). The immunolabelling of collagen type II and aggrecan was more pronounced at days 7 (not shown) and 14 (Fig. 6A2-C2, collagen type II; Fig. 6A4-C4, aggrecan), compared to day 2 in alginate culture, whereas the labelling for collagen type I and fibronectin decreased from days 2 to 14 in alginate (Fig. 6A6-C6; Fig. 6A8-C8).

Effect of IL-10 overexpression on collagen type II, aggrecan, collagen type I and fibronectin deposition in alginate culture as shown by immunofluorescence microscopy. Collagen type II (A1-C2), aggrecan (A3-C4), collagen type I (A5-C6) and fibronectin (A7-C8) expression was evaluated on chondrocytes either non-transduced (control, A) or transduced with the adv/empty vector (B) or the adv/hIL-10 vector (C). Cells were embedded for 2 (A1-C1, A3-C3, A5-C5, A7-C7) or 14 days (A2-C2, A4-C4, A6-C6, A8-C8) post transduction in alginate beads and matrix protein expression was assessed by labeling cryosections of alginate beads with specific antibodies and indirect immunofluorescence microscopy. Cell nuclei were counterstained with DAPI. Scale bars 20 μm.
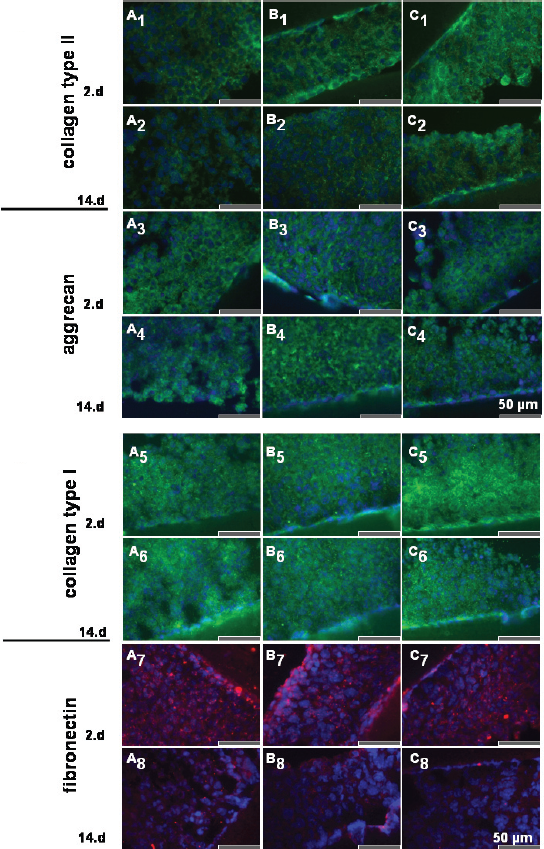
Effect of IL-10 overexpression on collagen type II, aggrecan, collagen type I and fibronectin deposition in high-density culture as shown by immunofluorescence microscopy. Collagen type II (A1-C2), aggrecan (A3-C4), collagen type I (A5-C6) and fibronectin (A7-C8) expression was demonstrated on both non-transduced chondrocytes (control, A) as well as chondrocytes transduced with the adv/empty (B) or the adv/hIL-10 (C) adenoviral vector. Cells were introduced for 2 (A1-C1, A3-C3, A5-C5, A7-C7) or 14 days (A2-C2, A4-C4, A6-C6, A8-C8) post transduction in high-density culture and cryosections were stained with specific antibodies for indirect immunofluorescence microscopy. Cell nuclei were counterstained with DAPI. Scale bars 50 μm.
In high-density culture the collagen type II labelling decreased from day 2 to day 14 whereas the aggrecan deposition remained unaltered (Fig. 7A3-C4). The staining intensity of the chondrocytes for collagen type I did not differ during the observation period of 14 days (Fig. 7A5-C6) but the fibronectin synthesis decreased from day 2 (Fig. 7A7-C7) to day 14 (Fig. 7A8-C8). No major differences in the deposition of collagen type II, aggrecan, collagen type I and fibronectin were evident in chondrocytes overexpressing IL-10 (Fig. 6,7C1–8) in alginate and high-density cultures, compared with their respective controls (Fig. 6,7 A1–8,B1–8).
Discussion
Over the last decade the anti-inflammatory potential of IL-10 in arthritis has been proposed through in vivo animal studies indicating that this immunoregulatory cytokine (IL-10) might be a promising candidate for gene therapy in the inflamed joint (Lechman et al. 1999, 2003; Neumann et al. 2002; Van de Loo and van den Berg, 2002; Gelse et al. 2003; Zhang et al. 2004; Kuroda et al. 2006). Only differentiated chondrocytes can be used for chondrocyte implantation to produce a stable cartilage-specific matrix and it remains unclear whether high concentrations of IL-10 might affect the differentiated phenotype of chondrocytes. The present study uses an in vitro overexpression model of human IL-10 in 3D chondrocyte cultures to investigate the direct effects of elevated concentrations of IL-10 on cartilage-specific matrix synthesis in differentiated chondrocytes. Overexpression of human IL-10 by the adenovirally transduced chondrocytes introduced in the 3D alginate culture systems was confirmed using ELISA analysis of the culture supernatants. Results showed that IL-10 transduced chondrocytes released high amounts of IL-10.
As previously reported by Häuselmann et al. (1994) and others (Bonaventure et al. 1994), our results indicate that irrespective of adenoviral transduction with the empty or IL-10 overexpression vector, increasing amounts of collagen type II and aggrecan deposition from days 2 through to 14 in alginate culture are synthesized, suggesting the stabilization of the differentiated phenotype of chondrocytes in these cultures. In contrast, expression of the ubiquitous matrix component fibronectin and the unspecific matrix protein collagen type I, which are both known to be produced in increasing amounts by dedifferentiating chondrocytes (Martin et al. 1999; Goessler et al. 2005, 2006), decreased in all treatment courses after the second day of alginate cultures. Collagen type II and aggrecan production were also found in the adenovirally transduced and non-transduced high-density cultures over the culture time indicating their differentiated phenotype which was not affected by transduction. One problem in using adenoviruses for gene transfer and gene therapy is that these vectors lead only to transient transduction as the transgene is usually lost during cell proliferation. Cell proliferation analysis using a CFDA-SE proliferation assay revealed clearly that monolayer chondrocytes had a higher proliferation rate compared with the alginate embedded cells, a possible explanation for prolonged EGFP expression in alginate and high-density culture for more than two weeks which is in agreement with the finding of Dinser et al. (2001). As previously reported by Dinser et al. (2001), in addition to the slow growing chondrocyte population in alginate culture, a second more rapidly growing population appeared at day 8 which might contribute to the gradual decrease of transgene expression in alginate culture. In alginate culture the EGFP expression rate was lower compared with monolayer chondrocytes. One might speculate that the synthetic activity of chondrocytes in alginate is generally lower. In the present study, we found that the expression of collagen type II and aggrecan was maintained in chondrocytes overexpressing IL-10 and transduced with the adv/empty which served as a control for the isolated effect of the adenovirus. The empty vector revealed a slightly non significant stimulatory effect on collagen type II expression. The fact that adenoviruses alone did not affect cartilage matrix deposition is in agreement with reports of Nixon et al. (2000) and Ikeda et al. (2000). There was no difference in collagen types I and II and in fibronectin synthesis between non-transduced chondrocytes and chondrocytes transduced with the IL-10 overexpression or the empty vector. The synthesis of collagen type I and fibronectin decreased in alginate culture from days 2 through to 14 but collagen type I remained unaffected in high-density culture. A precondition for cartilage homeostasis is the communication between chondrocytes and their specific extracellular matrix which is mediated via integrin receptors and essential for chondrocyte survival and differentiation (Hirsh et al. 1997; Yang et al. 1997; Cao et al. 1999). In the present study a broad expression of β1-integrins was detectable on around 90% of the alginate cultured chondrocytes which increased until 7 days of culture. β1-integrin cell surface expression remained unaffected by IL-10 overexpression in the alginate cultures.
Taken together key features of the differentiated phenotype are maintained in chondrocytes overexpresssing IL-10 in 3D cultures suggesting that anti-inflammatory gene therapy in the joint using IL-10 or implantation of IL-10 overexpressing chondrocytes in cartilage defects might not affect cartilage homeostasis.
Footnotes
Acknowledgement
The authors would like to acknowledge the support of the Deutsche Arthrosehilfe e.V, the Hypathia program of the University of Applied Science and the Rahel Hirsch program of the Charitέ Medical Schools Berlin and the Sonnenfeld foundation.
