Abstract
Osteoarthritis (OA) might affect chondrocyte culture characteristics and complement expression. Therefore, this study addressed the interrelation between macroscopical and microscopical structure, complement expression, and chondrocyte culture characteristics in non-OA and OA cartilage. Femoral head cartilage samples harvested from patients with femoral neck fractures (FNFs) and OA were analyzed for macroscopical alterations using an in-house scoring system, graded histologically (Mankin score), and immunolabeled for complement regulatory proteins (CRPs) and receptors. Morphology of monolayer cultured chondrocytes isolated from a subset of samples was assessed. The macroscopical score distinguished the FNF and OA cartilage samples and correlated significantly with the histological results. Chondrocyte phenotype from FNF or OA cartilage differed. Complement receptor C5aR, CRPs CD55 and CD59, and weakly receptor C3AR were detected in the investigated FNF and OA cartilage, except for CD46, which was detected in only two of the five investigated donors. The in-house score also allows inexperienced observers to distinguish non-OA and OA cartilage for experimental purposes.
Keywords
Introduction
Osteoarthritis (OA) is a chronic disease, which affects, depending on age, a large number of patients. More than 60% of the German population suffers from OA by the time of their 65th birthday. In combination with demographic changes, OA has led to an enormous rise of health-care costs over the past two decades.
1
Various nonmodifiable entities such as age, genetic, metabolic, biochemical, biomechanical, and modifiable factors, such as obesity, are all suspected to support this degenerative change.2,3 It has been observed that OA is initiated by multiple microtraumas to the joint or one single traumatic event.
4
However, the exact etiology remains unclear. Unfortunately, there is currently no effective biological and/or pharmacological therapy available for OA, despite some efforts in OA-related research. Within OA, there is a shift of the tightly regulated anabolic and catabolic process toward the catabolic state resulting in the upregulation of inflammatory markers, most notably interleukin-1β (IL-1β) and tumor necrosis factor-α (TNF-α).
3
Numerous animal model studies have shown the role of IL-1β in the upregulation of catabolic enzymes matrix metalloproteinases (MMP)-1, -3, and -13, while also identifying its ability to suppress the anabolic process, within the extracellular matrix (ECM), by inhibiting type II collagen and proteoglycan expression in chondrocytes.
5
Furthermore, IL-1β may initiate apoptosis via production of nitric oxide or reactive oxygen species within the chondrocyte.
6
Similarly, TNF-α exhibits enhanced expression not only in traumatic articular cartilage but also in OA synovium, inducing the same MMP enzymes as IL-1β, which suggests a synergistic catabolic effect during OA.
7
This loss of homeostasis between anabolic and catabolic factors might contribute to activation of the complement cascade as recently demonstrated for OA pathogenesis.
8
Complement activation results in the secretion of chemotaxins arising from cleaved complement factors, such as anaphylatoxins C3a and C5a. In addition, it induces opsonization of pathogenic particles, and finally, pathogen or cell lysis by membrane attack complexes (MACs).
8
The complement split fragments C3a and C5a bind to anaphylatoxin receptors, C3aR and C5aR, and induce a multiple inflammatory response, including cytokine release, vasoconstriction and permeability, histamine release, and leukotaxis, as well as platelet aggregation.9–11 Complement regulatory proteins (CRPs) protect cells from lysis induced by complement activation. The anaphylatoxin receptors are expressed on various cell types.
12
Interestingly, C5aR expression was observed as normal in OA and rheumatoid cartilage, while its upregulation was noted in normal, rheumatoid arthritis samples, but not in OA cartilage, when chondrocytes were stimulated by IL-1β.
13
In addition to C5aR, we wanted to demonstrate the expression of C3aR and crucial CRPs in OA and non-OA cartilage. We hypothesized in 2007 that complement expression could be involved in OA
14
and demonstrated the complement regulation by cytokines in cultured chondrocytes, in cartilage
Femoral neck fracture (FNF) is a typical feature of older patients suffering from sarcopenia and osteoporosis.18,19 An FNF usually cannot be treated by conservative strategies due to loss of femoral head vascularization. Therefore, endoprothetic joint replacement of the hip is usually performed. OA diagnosis is based mainly on clinical mobility and pain scores, eg, Western Ontario and McMaster Universities Arthritis Index, visual analog scale, and radiologic measurements, such as joint space narrowing (eg, Kellgren score). Histology provides more detailed information about OA progression in cartilage, but it cannot be applied in the clinical routine. When human articular cartilage is recruited for
Therefore, the aim of this study was to compare joint cartilage derived from FNF and hip OA patients using both microscopic and, a novel in-house, macroscopic scoring system while further analyzing chondrocyte culture characteristics. In addition, the expression of specific cell surface-located complement pathway-associated glycoproteins and complement anaphylatoxin receptors was assessed.
Materials and Methods
Samples
Samples of 37 patients (17 males, 20 females; 18 FNF, 19 hip OA; Table 1) were harvested during joint replacement surgery in accordance with the institutional ethics committee of the Charitέ-Universitätsmedizin Berlin, Campus Benjamin Franklin. The research was conducted in accordance with the principles of the Declaration of Helsinki, and patients gave their written, informed consent to participate. The average age of all donors included in the study was 68.72 years. The age range was between 30 and 91 years. In a subset of samples (15 donors), primary human chondrocytes were isolated from the femoral heads of patients who underwent joint replacement due to OA (4 donors) and FNF (11 donors).
Donor characteristics of samples included.

Macroscopical scoring
All femoral heads included in this study were scored macroscopically using the self-designed score before the joint cartilage was scratched off for chondrocyte isolation. Observers (one or two observers per time point, but different observers at different time points) were blinded for the diagnosis (FNF/OA). If two observers were included, the results were averaged. The topics such as color, extent of lesions (quadrants affected), consistency, and cartilage thickness were addressed during the scoring process as summarized in Table 2.
Self-designed macroscopical scoring system.

Isolation and culturing of articular chondrocytes
Apart from a 1-cm2 area reserved for histology, cartilage chips were harvested from macroscopically, mostly unaffected cartilage areas of human femoral head articular cartilage with a macroscopical score of ≤9, since at higher scores, the amount and quality of the remaining cartilage appeared too low for successful cell isolation. The rationale to also include OA-affected cartilage was to study the effect of the OA milieu in the joint on chondrocyte phenotype. The harvested cartilage was weighed and cut into small slices (∼1 mm2) followed by digestion with pronase derived from
Culture morphology
For the measurement of cell morphology and the number of adherent cells, freshly isolated cells (passage 0) of 15 donors were seeded into Petri dishes (35 mm diameter) at 52,083.33 cells/cm2 and cultured for 72 hours. Measurement was taken using cellD software at ×100 magnification under a light microscope (Axioskop 40, Zeiss), while cellular morphology, including length (μm), width (μm), and overall cell area (μm2), was assessed at ×200 magnification. Images were taken at ×100 using a XC30 camera system (Olympus, Europa Holding).
Decalcification and embedding of cartilage bone cylinders
A 1-cm2 cartilage–subchondral bone sample was fixed using 4% paraformaldehyde (PFA) solution (USB, USA) for three days. Twenty-two samples were recruited for histology (derived from 11 female and 11 male donors, 16 of them suffering from FNF and 6 from OA). The samples were placed in a volume 20 times their own volume of formic acid to ensure satisfactory decalcification. The acid solution was changed every 24 hours, while a chemical end point test was also carried out on the supernatant to access the rate of decalcification. When chemical testing exhibited no significant residual calcium, the samples were rinsed in water and incubated in 5% ammonia solution (Merck KGaA) for 30 minutes to ensure neutralization. Samples were then rinsed under running tap water for 24 hours to remove any residual acid. Dehydration involving a series of ascending ethanol solutions was completed over a 48-hour period. Following this, each sample was embedded in paraffin at 60 °C with subsequent sectioning using a microtome Microm HM 325 (Thermo Fisher Scientific).
Histological staining
The histological staining was conducted on paraffin sections (thickness between 3 and 5 μm). Paraffin was removed using a series of descending alcohol solutions. For hematoxylin and eosin (H&E) staining, the sections were incubated for 4 minutes in Harris hematoxylin solution (Sigma-Aldrich), briefly rinsed in tap water and then counterstained for 1.5 minutes in eosin Y (Sigma-Aldrich). For the safranin-O staining, the slides were incubated for 10 minutes in Weigert's iron hematoxylin (Carl Roth GmbH) and afterward rinsed under running tap water for 10 minutes. Subsequently, each section was stained in 0.001% fast green solution (Sigma-Aldrich) for five minutes. After rinsing the slides briefly in 1% acetic acid, the slices were stained in 0.1% safranin-O solution (Merck KGaA) for up to five minutes. For alcian blue (AB) staining, the sections were incubated for 3 minutes in 1% acetic acid followed by 1% AB (Carl Roth GmbH) for 30 minutes. Then, they were rinsed in 3% acetic acid and washed in deionized water (A. dest.) (two minutes). Additionally, cell nuclei were counterstained in nuclear fast red aluminum sulfate solution (Carl Roth GmbH) for five minutes.
Finally, all the slides were dehydrated using an ascending alcohol series and mounted with Entellan mounting medium (Merck KGaA). Images of the slides were taken using the Axioskop 40 light microscope and the Olympus digital camera XC30.
Microscopical scoring for OA
The stained sections were further analyzed by applying the Mankin score24,25 to them by one observer. The observers were blinded for the diagnosis (FNF, OA) of the cartilage donors. Both cartilage structure and chondrocyte distribution were scored, while safranin-O and AB staining allowed the grading according to the amounts of glycosaminoglycans detectable in the sample. Finally, tide-mark integrity was assessed for blood vessel penetrance.
Immunofluorescence labeling
Paraffin sections of five donors (two with OA, two with FNF, and one with OA and FNF) were immunolabeled for complement receptors (C5aR and C3aR) and CRPs (CD46, CD55, and CD59) using specific antibodies combined with a counterstaining of the cell nuclei by 4′,6-diamidino-2-phenylindole (DAPI). The sections were deparaffinized and fixed in 4% PFA before being rinsed for two minutes in tris-buffered saline (TBS; 0.05M Tris, 0.015M NaCl, pH 7.6). The sections were overlaid for 20 minutes with 0.1% pronase solution at 37 °C for demasking and rinsed two times with TBS (2 minutes). Ten, a 20-minute incubation period at room temperature (RT) with 100 μL protease-free blocking buffer (5% donkey serum [Merck Millipore] diluted in TBS) followed. Negative controls included omitting the primary antibody or using mouse IgG1 as primary antibody during the staining procedure. The primary antibodies (rabbit anti-human C3aR [1:30, Assay Biotechnology], mouse anti-human C5aR [1:100, GeneTex, Biozol], mouse anti-human CD46, CD59 [1:20 and 1:40, both AbD Serotec], and goat anti-human CD55 [1:40, R&D Systems]) were diluted in blocking buffer containing 0.1% Triton X-100 (Sigma-Aldrich) and incubated overnight at 4 °C in a humidified chamber. After rinsing three times with TBS, the labeled secondary antibody (donkey anti-rabbit or donkey anti-mouse-Alexa488 or -Alexa555, 10 mg/mL; Life Technologies Corporation) was diluted 1:200 in blocking buffer containing 0.1% Triton X-100 and 0.1 μg/μL DAPI and incubated for one hour at RT in a dark room. Labeled sections were rinsed with TBS three times and covered with Fluoromount G (Biozol Diagnostica). The fluorescence images were acquired using a confocal laser scanning microscope (SPEII; Wetzlar Leica).
Statistical analysis
Data were expressed as an average value with standard deviation or median and analyzed using GraphPad Prism 5. The ROUT test was performed to identify outliers. If applicable, the Kolmogorov-Smirnov test was used to analyze the data for the presence of a Gaussian distribution. In the presence of a Gaussian distribution, unpaired
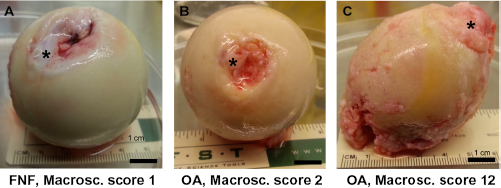
Exemplary macroscopical images of explanted femoral heads and scoring results. Depicted are femoral heads of three representative donors. (A) FNF, macroscopical score 1, (B) OA, macroscopical score 2, and (C) OA, macroscopical score 12.
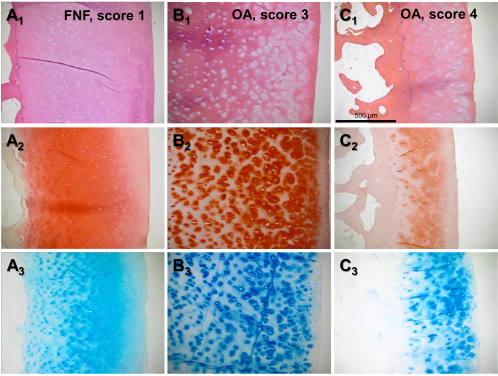
Exemplary histological images of cartilage samples. H&E (A1–C1), Safanin O (A2–C2), and AB (A3–C3) stained paraffin sections of femoral head articular cartilage of three representative donors are shown. (A) FNF, Mankin score 1, (B) OA, Mankin score 3, and (C) OA, Mankin score 4.

Macroscopical and Mankin scoring results of OA and FNF cartilage samples. (A) Macroscopical scoring results of FNF and OA samples. (B1) Correlation between all macroscopical and Mankin scoring data (FNF and OA) and separated for FNF (B2) and OA (B3).

Femoral head diameters with respect to OA and gender. (A) Femoral head diameters of FNF and OA donors. (B) Femoral head diameters of male and female donors.

Vitality, adherence, and cytoplasmic area of cultured femoral head chondrocytes isolated from FNF or OA samples. Images of cultured chondrocytes derived from femoral head cartilage prone to FNF (A1) or OA (A2). (B) Cell density in FNF and OA samples. (C) Isolated cell numbers per gram versus macroscopical scoring results. (D) Isolated cell numbers per gram versus Mankin scoring results. Images and calculations derived from chondrocytes cultured for 72 hours.
Results
Patients
Patients were sorted according to their age following the WHO classification as follows: less than 60 or 60 years (aging), between 61 and 75 years (old), between 76 and 90 years (older), and between 91 and 100 years (very old). Most of them were between 61 and 75 years (21, 56.76%) and females (20, 54.05%). The average age of the OA cartilage donors included was 63.68 years, whereas that of the FNF cartilage donors was 74 years. The body mass index (BMI) was significantly higher in OA compared with FNF patients (Table 3). The percentage of each comorbidity (diabetes, cardiovascular diseases, infection, and kidney disease) was generally higher in the FNF patients (Table 3).
Additional donor characteristics of FNF and OA samples.

The FNF donors were significantly older than the OA donors.
The FNF donors had a significantly lower BMI than the OA donors.
Grading of OA using the self-designed macroscopical scoring system and the Mankin score for histological scoring
A self-designed score system was used to determine the macroscopical grade of cartilage degradation (Table 2 and Fig. 1A–C). Using this scoring system, the same samples were evaluated by inexperienced versus experienced observers. The differences of the scoring results between both observer groups were not significant (data not shown). For histological evaluation of OA-associated changes (Fig. 2), the Mankin score was used. The macroscopical score was significantly higher in patients with hip OA compared with those suffering from FNF (Fig. 3A). The median of the macroscopical score for OA was 11, whereas that for FNF was 3.5 (
Femoral head diameters
When the femoral head diameters were measured using the X-ray images, the male femoral heads were, as expected, significantly larger compared with the female (
Chondrocyte morphology, adherence, and vitality
Cells were isolated enzymatically, and the percentage of adherent cells as well as cell morphology (overall cell area, length, and width) was determined after 72 hours of culturing (Fig. 5A1–2). No significant correlation could be detected between the cell density of cultured chondrocytes and the macroscopical score (
Characteristics of FNF- and OA-derived chondrocytes cultured for 72 hours.
Significant difference,
Detection of regulatory complement proteins in situ
C3aR receptor protein revealed only a weak immunoreactivity in the investigated samples. C5aR was expressed by the cells of the surface cartilage layer. The CRP CD46 could be found in two of the five donors analyzed. CD55 was synthesized in the samples of all investigated donors. Signals were not only confined to the cells but also observable in the whole lacunae. Nevertheless, it could not be found in the interterritorial ECM. Protein expression of CD59 was shown in all investigated donors with divergent distribution (Fig. 6).
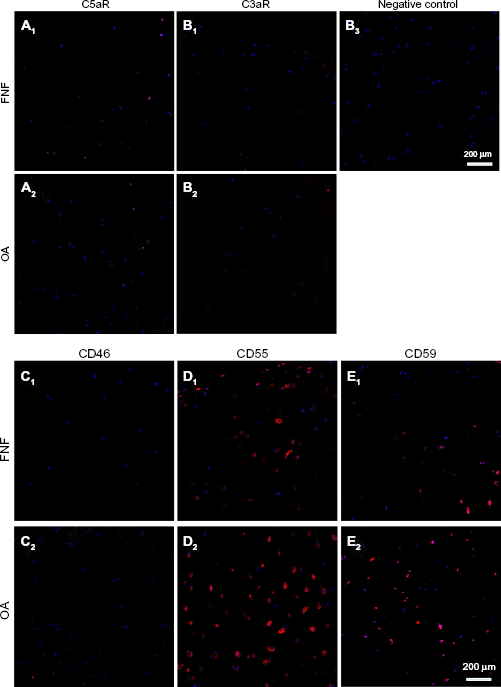
Expression of complement receptors (C5aR and C3aR) and CRPs (CD46, CD55, and CD59) in cartilage in situ. FNF and OA cartilage sections immunolabeled for (A1–2) C5aR, (B1–2) C3aR, (C1–2) CD46, (D1–2) CD55, and (E1–2) CD59.
Discussion
In this study, a simple in-house macroscopical score was established, and we approved it to distinguish FNF and OA cartilage samples. Macroscopical scoring results correlated significantly with the histological analysis. It is advisable to categorize the quality of cartilage used for chondrocyte isolation since chondrocyte phenotype differed between FNF or OA cartilage. The complement receptor C5aR and the weakly expressed receptor C3aR, as well as CRPs CD55 and CD59, were detected in the investigated FNF and OA cartilage samples. However, CD46 could only be found in two of the five investigated donors.
The macroscore was established for a rapid cartilage grading before cell isolation as simple as possible to allow even inexperienced laboratory staff to estimate OA in the recruited samples. Indeed, the results deduced from this easy-to-handle macroscopical score did not show major differences between experienced and inexperienced observers. This score could help to spare the time-consuming histology.
The total donor cohort analyzed macroscopically reflected a higher number of female individuals than male individuals as usually reported for OA. 26 On average, hip OA led to total joint replacement with 63.7 years, whereas the FNF (FNF) occurred around 10 years later with 74 years. A higher number of these FNF patients suffered simultaneously from other diseases such as diabetes, infections, kidney disease, and cardiovascular diseases than the OA patients. This fact might be age dependent. However, it was reported that women who have hip OA also attain a higher rate of comorbidities. 27 Furthermore, the BMI in the OA group was significantly higher. Comparison of the OA and FNF patient-derived cartilage samples indicated a highly significant difference between both groups concerning the macroscopical score (macroscopical scoring values of OA samples varied between 2 and 12 [median: 11]), and that of the FNF group in a similar range (also between 1 and 12), but the median was lower (median: 3.5). However, only samples with a scoring value lower than 9 were used for cell isolation, since samples with higher scores presented insufficient amounts of cartilage. From these samples, only the macroscopically barely changed cartilage was recruited. Also, the FNF group presented only three samples with a low macroscopical score (one score point), indicating macroscopically nearly unaffected cartilage. For this observation, age-associated and early OA changes might be responsible since the average age in the FNF group was around 74 years.
Histological scoring can be done using several scoring systems.28,29 The Mankin scoring system, which was selected in this study, is well-established one. 30 The Mankin and the recently recommended Osteoarthritis Research Society International (OARSI) cartilage histopathology assessment system revealed a high reliability. 31 The histological score and the macroscopical score correlated significantly. Many samples revealed focal OA due to incongruency of the femoral head and acetabulum. However, the Mankin score revealed differences between OA and FNF group (not significant). One has to consider the fact that the macroscopical score allows an assessment of the whole femoral head, whereas for histology, only one exemplary sample was selected, which represented an area of the best cartilage quality of the whole head. This cartilage area was selected to be representative for the cartilage subsequently harvested for cell isolation. Therefore, the histology was performed to get information concerning the chondrocytes isolated later from the same samples.
It has been reported that chondrocyte morphology changes in hip OA.
32
Holloway et al, detected a subpopulation of chondrocytes with multiple elongated cytoplasmic extensions.
32
We found the tendency of a larger cell area in OA samples compared with FNF cartilage, but the length and width of the cells did not significantly differ. The reason remains unclear. However, we evaluated only cell culture characteristics, but we did not measure the chondrocyte diameter in the histological sections. This was not necessary, since we wanted to elucidate chondrocyte differences in culture, which might influence future
OA is known to be associated with a higher rate of cell death
Complement has recently been implicated in OA pathogenesis.8,14,15 We previously observed gene expression and regulation of complement receptors and cell surface-associated regulatory proteins in cultured chondrocytes.
15
Now, we studied the expression of selected complement receptors and regulatory proteins
Limitations of this Study
This study is limited by the heterogenous donor collective and their high age, particularly in the FNF subgroup. So, age-related influences could not be excluded. For the determination of the cell parameters in culture, the OA group was rather small (
For histology, only one sample could be included from each donor, since the majority of cartilage was needed to be harvested for cell isolation. So, the best cartilage area of each femoral head was selected. This could be the reason why the Mankin scoring provided generally lower scoring numbers, suggesting higher cartilage quality, and hence, superior results than the macroscopical score. In this study, the macroscopical score is more predictive than the Mankin score, concerning cartilage quality. In this respect, sampling and time-consuming processing for histology could be omitted in future.
Conclusion
The self-designed scoring system is applicable for an easy grading of cartilage samples by inexperienced observers for experimental purposes. We are not expecting that our macroscopical scoring system will also find clinical application in arthroscopy of the hip joint. For our coworkers in the laboratory setting, who did not have XRs, etc., available, this score was accepted as helpful and practical. Experiments with human-derived chondrocytes are superior to using animal-derived samples due to interspecies differences.37,38 Therefore, the facts reported here for FNF- versus OA-derived chondrocytes, which are the main chondrocyte sources of human origin for research, help to assess their peculiarities and its potential influence on experiments. With regard to further characterize the extent of complement activation during OA progression, other complement factors such as C3, C4, and C5 and the split fragments such as C3a and C5a should also be included in future studies.
Author Contribution
Conceived and designed the experiments: JB, OG, GS-T, CM, MJ. Analyzed the data: JB, BK, TS, GS-T. Wrote the first draft of the manuscript: OG, GS-T. Contributed to the writing of the manuscript: ZH, SA. Agree with manuscript results and conclusions: JB, OG, BK, CM, MJ, ZH, SA, TS, GS-T. Jointly developed the structure and arguments for the paper: GS-T, BK, MJ. Made critical revisions and approved final version: JB, OG, MJ, ZH, SA, GS-T. All authors reviewed and approved the final manuscript.
Footnotes
Acknowledgments
The authors acknowledge the support of Marion Lemke. An abstract regarding this research was published as part of the conference proceedings of the 2016 Osteoarthritis Research Society International (OARSI) World Congress.
