Abstract
Introduction
A viral breakthrough occurs when a patient achieves a response while on interferon (IFN) therapy and then loses the response despite continued IFN therapy. The cause of viral breakthroughs is not well understood. We encountered three cases with viral breakthrough during treatment with pegylated interferon (PEG-IFN) and ribavirin (RBV).
Case presentation
The three cases were all late virological responders. They did not express anti-IFN alpha-2b antibodies after PEG-IFN and RBV therapy. We analyzed amino acid substitutions of core 70, core 91, and interferon sensitivity-determining region (ISDR), which significantly influence sustained virological response (SVR). Their amino acid substitutions of core 91 were mutant in two cases. Amino acid substitutions of ISDR were wild pattern in two cases. PEG-IFN adherence was above 80% in three cases, and RBV adherence was below 80% in two cases.
Conclusion
During PEG-IFN and RBV therapy, we should watch for viral breakthrough in late virological responders with mutant type of amino acid substitutions of core 91, wild pattern of amino acid substitution of ISDR, and decrease of RBV adherence. Viral breakthrough is an important problem in PEG-IFN and RBV therapy for chronic hepatitis C. Therefore, it should be investigated more thoroughly in more cases.
Keywords
Introduction
Infections with hepatitis C virus (HCV) have become a major cause of hepatocellular carcinoma. 1 Viral breakthrough is a recognized response pattern to interferon-based antiviral therapy in patients with chronic HCV infection. Current treatment algorithms result in rates of sustained virological response (SVR) in over 80% of patients infected with HCV genotype 2 or 3, suggesting that some of the primary challenges in the management of chronic hepatitis C (CHC) have now been resolved. In patients infected with HCV genotype 1, however, the standard combination treatment of 48 weeks of pegylated interferon (PEG-IFN) alpha and ribavirin (RBV) results in SVR rates of only 40%-50%, with higher rates following 48 weeks rather than with 24 weeks of treatment. 2 Previous studies from Europe and the United States have demonstrated that late virological responders (LVR) (HCV RNA-positive at 12 weeks and negative at 24 weeks after the start of treatment) show improved SVR rates when treatment is extended to 72 weeks, compared with the standard duration of therapy, largely as a result of reducing post-treatment relapse rates. Akuta et al reported that they compared the treatment efficacy of a 72-week regimen in 65 patients with that of a 48-week regimen in 130 patients, who were infected with HCV genotype 1b and treated with PEG-IFN/RBV in Japan. 3 SVR was achieved by 61.5% and 32.3% of patients in the 72- and 48-week groups, respectively, while non-virological response was noted in 9.2% and 29.2% of the respective groups. Multivariate analysis identified substitution of amino acid 70 and 91 (Arg70 and/or Leu91) and duration of treatment (72 weeks) as independent parameters that significantly influenced SVR. Amino acid mutations in the interferon sensitivity-determining region (ISDR) (amino acid position 237-276 of the nonstructural region 5A [NS5A] protein consisting of 447 amino acids) of HCV are related to increased interferon sensitivity and low viral load. 4 Kogure et al reported that adherence to PEG-IFN was found to influence the SVR rate as a treatment-related factor in their study. 5 SVR rates were low in patients who did not receive 80% or more of the intended dose of PEG-IFN. Adherence to RBV up to the 12th week did not significantly influence the SVR rate, but overall adherence to RBV significantly influenced the SVR rate. It was notable that adherence to PEG-IFN alpha-2b significantly influenced SVR in their study. In the patients who did not receive 80% or more of the intended dose by the 12th week, the SVR rate decreased markedly. Jorns reported that neutralizing anti-IFN antibodies were detected in the sera of 3 of 38 chronically HCV-infected patients who failed to respond to therapy with IFN-alpha but none in the sera of patients who cleared HCV after IFN therapy. 6 Some investigators have postulated that viral breakthroughs occur as a result of neutralizing antibodies to IFN.7,8
We encountered three cases with viral breakthrough during treatment with PEG-IFN and RBV. The three patients complied with the standard treatment with PEG-IFN and RBV. We analyzed each adherence to PEG-IFN and RBV, anti-IFN alpha-2b antibody, and amino acid mutations of core 70, core 91, and ISDR.
Case Presentations
Case 1
A 54-year-old male had no history of blood transfusion or surgery. He was treated with IFN alpha monotherapy for 6 months, but HCV-RNA relapsed after IFN therapy. The HCV genotype was Ib and the HCV-RNA level was 2010 KIU/ml before this treatment. He was treated with PEG-IFN alpha-2b 100 μg/week and RBV 800 mg/day. After 20 weeks, HCV-RNA was negative. After 52 weeks, HCV-RNA relapsed. During PEG-IFN and RBV therapy, ALT levels remained high. Adherence to PEG-IFN and RBV was 87% and 83%, respectively. Anti-IFN alpha-2b antibody was negative. Amino acid substitution of core 70 was mutant, core 91 was mutant, and number of ISDR was one (Fig. 1).
Clinical course of case 1.
Case 2
A 65-year-old female underwent surgery for uterine fibroids and had a history of blood transfusion at 44 years old. She received IFN monotherapy three times: IFN alpha (total of 624 MU), IFN alpha-2b (total of 810 MU), and IFN alpha-2b (total of 792 MU), but HCV-RNA relapsed. The HCV genotype was Ib, and the HCV-RNA level was 644 KIU/ml before this treatment. She was treated with PEG-IFN alpha-2b 80 μg/week and RBV 600 mg/day. After 14 weeks, HCV-RNA was negative. After 40 weeks, HCV-RNA relapsed. During treatment, ALT levels remained high. Adherence to PEG-IFN and RBV was 100% and 71.5%, respectively. Anti-IFN alpha-2b antibody was negative. Amino acid substitution of core 70 was wild, core 91 was wild, and ISDR was wild (Fig. 2).
Clinical course of case 2.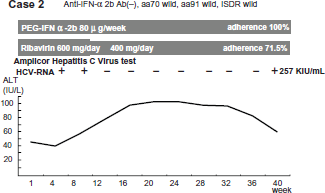
Case 3
A 60-year-old female had no history of blood transfusion, but she underwent an appendectomy at 10 years old. She received IFN monotherapy two times: IFN alpha (total of 480 MU) and IFN alpha-2b (total of 640 MU) but she relapsed. The HCV genotype was Ib, and the HCV-RNA level was 254 KIU/ml before this treatment. She was treated with PEG-IFN alpha-2b 80 μg/week and RBV 600 mg/day. After 18 weeks HCV-RNA was negative. After 69 weeks, HCV-RNA relapsed. During treatment, ALT levels remained at a high level. Adherence to PEG-IFN and RBV was 84.7% and 76%, respectively. Anti-IFN alpha-2b antibody was negative. Amino acid substitution of core 70 was wild, core 91 was mutant, and ISDR was wild (Fig. 3).
Clinical course of case 3.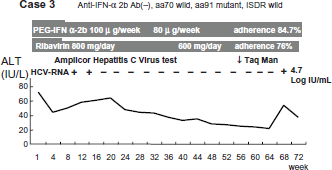
Discussion
A viral breakthrough occurs when a patient achieves a response while on IFN therapy and then loses the response despite continued IFN therapy. The cause of viral breakthroughs is not well understood. The viral breakthrough phenomenon might also be the result of the development of IFN resistance or the emergence of quasispecies that are less sensitive to IFN therapy. Vuillermoz reported that three hypotheses have been proposed to explain viral breakthrough during IFN treatment: i) the development of neutralizing anti-IFN antibodies, ii) the down-regulation of IFN receptors, and iii) the emergence of resistant viral strains. 9 The first hypothesis can be partially ruled out as neutralizing anti-IFN antibodies are not consistently found in association with viral breakthrough.10,11 Second, the down-regulation of IFN receptors during therapy, as observed in hepatitis B, has not been clearly correlated to treatment response. 12 The third hypothesis, which is the emergence of resistant strains during treatment, has been recently suggested. Indeed, a recent study showed that the evolution of hypervariable region 1 (HVR1) quasispecies during IFN monotherapy was different between sustained responders and patients who experienced a viral breakthrough. 13 In PEG-IFN and RBV combination therapy, there are still viral breakthroughs during therapy, but little is known with respect to viral breakthrough during PEG-IFN and RBV therapy. Vuillermoz analyzed HCV genome sequences and host humoral immune responses in nine patients who experienced a viral breakthrough during IFN alpha-2b and RBV combination therapy. The analysis of HCV genetic variability in patients with a viral breakthrough during IFN alpha-2b and RBV therapy showed a complex pattern of evolution of HCV quasispecies. The mean number of mutations in the ISDR was similar between viral breakthroughs and responders, but was greater for both of these groups as compared with non-responders. There was no common HCV genomic profile in breakthrough patients. Concerning the capsid and E2 proteins and the V3 region, the selection of strains with a higher fitness may play a role in the acquisition of resistance in some patients, but no selection of specific mutations was observed. 9 Zekuan suggested that viral breakthrough might be attributed to the selection of minor quasispecies variants at the baseline with or without additional mutations during antiviral therapy. Furthermore, the pattern for mutation clustering indicated potential mutation linkage among E2, NS2, and NS5a due to structural or functional relatedness in HCV replication. 14 Although we could not find any reports in the literature on the relation between vital breakthrough and substitution of amino acid 70 and 91, Akuta reported that the frequencies of substitution sites at aa70 and/or 91, which were a significant predictor of virological non-response based on multivariate analysis, were 95.0% (19/20), 62.5% (15/24), and 40.9% (9/22) in the three groups of absolute virological non-responders, relative virological non-responders, and virological responders, respectively. The proportion of substitution of amino acid 70 and 91 in absolute virological non-responders was significantly higher than that in relative virological non-responders and virological responders. 15
Case 1 was a late virological responder. He did not express anti-IFN alpha-2b antibodies after treatment. His amino acid substitutions of core 70 and core 91 were double mutant, and ISDR was also mutant. Adherences to both PEG-IFN and RBV were above 80%. Case 2 was a late virological responder, too. She did not express anti-IFN alpha-2b antibodies after treatment. Her amino acid substitutions of core 70 and core 91 were double wild, and ISDR was wild. Although adherence to PEG-IFN was above 80%, adherence to RBV was below 80%. Case 3 was also a late virological responder. She did not express anti-IFN alpha-2b antibodies after treatment. Her amino acid substitution of core 70 was wild, her amino acid substitution of core 91 was mutant, and her acid substitution of ISDR was wild. Although adherence to PEG-IFN was above 80%, adherence to RBV was below 80%.
Characteristics of the three patients.

Conclusions
During PEG-IFN and RBV combination therapy, we should watch for viral breakthrough in late virological responders with mutant type of amino acid substitutions of core 91, wild pattern of amino acid substitution of ISDR, and decrease in RBV adherence (below 80%). Viral breakthrough is very important in PEG-IFN and RBV therapy for CHC. Therefore, it should be investigated more thoroughly in more cases.
Footnotes
This manuscript has been read and approved by all authors. This paper is unique and is not under consideration by any other publication and has not been published elsewhere. The authors and peer reviewers of this paper report no conflicts of interest. The authors confirm that they have permission to reproduce any copyrighted material. Written consent was obtained from the patient or relative for publication of this study.
