Abstract
In this review, we analyze the effects of systemic lupus erythematosus and scleroderma on the gastrointestinal tract. There is a wide variation of gastrointestinal manifestations from these autoimmune disorders including but not limited to: oral ulcers, dysphagia, gastroesophageal reflux disease, abdominal pain, constipation, diarrhea, fecal incontinence, pseudo-obstruction, perforation and gastrointestinal bleeding. The purpose of this review is to discuss these manifestations, the appropriate diagnostic tests, and treatment.
Systemic Lupus Erythematosus
Systemic Lupus Erythematosus (SLE) is a multisystem disease characterized by antinuclear antibodies and complement deposition. The disease affects women more then men (especially African-American women) with a reported female/male ratio of 10:1. 1 Gastrointestinal (GI) complications are common in patients with active SLE. In 1895, William Osler was the first to describe how the GI complications of SLE can mimic any type of abdominal condition and overshadow the other organ complications associated with SLE. Gastrointestinal complications can lead to a variety of symptoms across a large portion of the GI tract. Up to 50% of patients will experience anorexia, nausea or vomiting.
Oral Cavity and SLE
Oral or nasopharyngeal ulceration, usually painless, observed by a physician is one of the eleven diagnostic criteria of SLE. Mucous membrane involvement occurs in 12%-45% of patients. The wide variation may be explained by demographic differences. One cross-sectional analysis reported that 46% of English patients with SLE had oral lesions compared to 15% of patients in Brazil and 11% of patients in Sweden. 2 Another study reported oral ulcerations at a prevalence of 26% in an Arab population of SLE patients, and noted a significant association with the presence of anti-smith antibodies (P = 0.033). 3 Causes of the mucosal ulcerations include the underlying disease activity, associated Sjogren's syndrome, intra-oral infections and complications associated with treatment.
The ulcers are generally classified as either discoid, erythematous or ulcerative. Discoid lesions, characterized by erythema, atrophy, and depigmentation, usually occur on the lips. Punched-out erosions/ulcers with surrounding erythema, irregularly shaped raised white plaques, and areas of erythema tend to occur on the hard or soft palate and the buccal mucosa. The discoid lesions are painful while the erythematous lesions (the majority) are painless. It has been reported by Jonsson et al that only those patients with active disease can have ulcerative and discoid lesions. However, they found no correlation between overall prevalence of oral lesions and SLE disease activity. 4
The treatment options for oral lesions include topical glucocorticoids, tacrolimus ointment, intralesional corticoids, and systemic antimalarial drugs. Most lesions respond to steroids in days to weeks.
Esophagus
Dysphagia occurs in 1.5%-13% of SLE patients and heartburn has been reported in up to 50%. These symptoms may be related to SLE or to complications associated with therapy such as pill esophagitis and oral candidiasis. The pathophysiology for the dysmotility is not known. Hypotensive LES and aperistalsis have been documented by manometry. The degree of manometric abnormality found in SLE appears to be less than that seen in mixed connective tissue disorder and scleroderma. Guiterez et al studied 14 SLE patients. Eight of them reported frequent esophageal symptoms. Three were found to have aperistalsis. 5 There have also been reports of dysphagia in SLE patients associated with epidermolysis bullosa acquisita, which results in severe esophageal mucosal disease. 6 Symptoms of dysphagia may be exacerbated by decreased saliva production similar to that seen in Sjogren's syndrome. Treatment of esophageal disease depends on the specific etiology. Hypomotility can be treated with metaclopramide. Immunomodulating agents have been used successfully to treat affected organ systems in SLE. However, there are no data that examine the response of the swallowing unit to that treatment. In fact, potent immunosuppressive regimens can induce dysphagia by causing opportunistic esophagitis from candidiasis, cytomegalovirus, and herpes simplex virus. Ischemia from underlying vasculitis can lead to esophageal ulceration. Perforation has also been reported; why they occurred is unclear. 25 GERD symptoms in SLE patients can be treated with routine GERD precautions and proton pump inhibitors.
Stomach and Duodenum
Complaints of dyspepsia have been reported in 4%-21% of SLE patients. 7 This may be related to the fact that the majority of SLE patients will require treatment with NSAIDs and/or corticosteroids. The link between NSAID use and peptic ulcer disease has been well established. Corticosteroids are also associated with dyspepsia and may produce a slightly increased risk of ulcers. The incidence of peptic ulcer disease associated with perforation has been reported as 5.8% and 7.6% in two separate studies of SLE patients presenting with abdominal pain. 1 However neither study commented on SLE disease activity or NSAID/corticosteroid use. The incidence of H. pylori infection in SLE patients with peptic ulcer disease is unclear. Helicobacter infection should be excluded in all SLE patients with peptic ulcer disease. Prophylactic therapy for peptic ulcer disease may be beneficial in SLE patients with dyspepsia, history of peptic ulcer disease, and on therapy with an NSAID and steroids. In addition to peptic ulcer disease, Gastric antral vascular ectasia (GAVE) has been reported in SLE patients.
Small and Large Bowel
There are several small and large intestinal manifestations of SLE including: Abdominal pain, bleeding, dysmotility, diarrhea (usually related to malabsorption or protein-losing enteropathy), pneumatosis cystoids, isolated rectal ulcers and steatorrhea.
Abdominal pain may be related to mesenteric vasculitis and infarction, acalculus cholecystitis, peptic ulcer disease and pancreatitis. Intestinal vasculitis has been reported to be 0.2% in a study by Drenkard et al of 540 SLE patients. 8 Other studies have shown an incidence rate of up to 53%. A recent chart review study of 175 patients by C-K Lee et al showed that vasculitis was the most common cause of acute abdominal pain in SLE patients (found in 45% of patients). They also showed that SLE disease activity index and laboratory data, except leucopenia, do not correlate with the occurrence of vasculitis. It is a disorder with life threatening complications (necrotic bowel, bowel perforation). The most common symptoms associated with intestinal vasculitis are abdominal pain, nausea and vomiting.
The diagnosis of vasculitis may be difficult to establish because of its insidious and intermittent behavior that is common prior to the acute event. Early nondiagnostic radiographic findings include thumbprinting, pseudoobstruction and segmental bowel dilatation. Abdominal CT and arteriography may be more helpful with diagnosis but a negative arteriogram does not exclude disease given vasculitis generally involves small arteries. The vasculitis causes transmural inflammation with fibrinoid necrosis. It is most commonly found in the terminal ileum and cecum where the blood supply is from the superior mesenteric artery. Endoscopically, the bowel may appear thickened and friable with ulceration and submucosal hemorrhages. Colonic manifestations include focal or diffuse ulcers, ischemic colitis, or colonic diverticula. In terms of treatment, there have been no randomized clinical trials. Patients are generally treated with bowel rest, broad spectrum antibiotics, steroids and cyclophosphamide. There have been case reports of successful treatment with intravenous methylprednisolone and cyclophosphamide. 1 Steroid responsive patients can be converted to oral prednisone and azathioprine. Patients who do not respond promptly to medical therapy should undergo laparoscopy.
Other, more infrequent causes for acute abdominal pain in SLE patients include pancreatitis and acalculus cholecystitis. A recent review article cited 77 case reports of acute idiopathic pancreatitis in the literature. 9 The median age was 27 and 88% were female. Most of the patients (84%) had active lupus at the time of pancreatitis. Mortality rate was reported to be higher than in non-SLE associated pancreatitis. Treatment with steroids or azathioprine was not significantly associated with increased mortality. In fact, treatment with steroids lowered the mortality by 67% compared to nontreated patients.
Protein-losing enteropathy (PLE) is another condition of the small bowel associated with SLE. It is characterized by significant hypoalbuminemia and diarrhea without proteinuria. The clinical manifestations are a result of villous atrophy with inflammatory infiltrates and submucosal edema. There have been approximately 20 case reports of PLE. Disease activity can be measured by monitoring fecal excretion of radiolabelled albumin. The exact cause of PLE is unknown. Treatment involves low dose steroids. Some patients may benefit from a gluten free diet and/or treatment of bacterial overgrowth.
Pneumatosis cystoides intestinalis is usually a benign condition. However, in patients with SLE, they may cause necrotizing enterocolitis and perforation. Vasculitis at the site of involvement is thought to be the cause for the perforation. Another condition seen in SLE patients, steatorrhea may occur secondary to villi blunting and immune complex deposition.
Scleroderma
Scleroderma disease severity scale.

Manifestations of SSc in the gastrointestinal tract by organ system with the pertinent procedure/radiologic study.
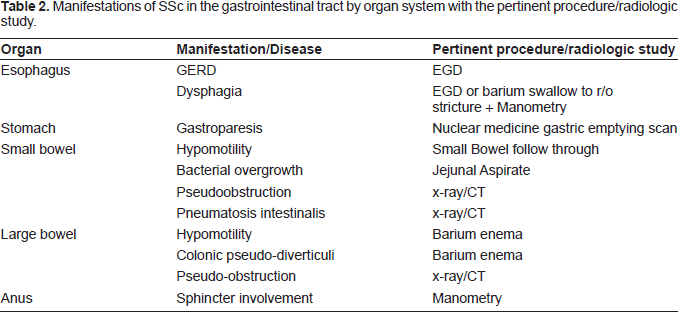
Esophagus
The esophagus is the most frequently involved gastrointestinal organ. It is only second to the skin as the most frequently involved organ in the body. Clinically, patients may report symptoms of dysphagia or gastroesophageal reflux disease (GERD). Other than symptoms of GERD, the incompetent lower esophageal sphincter can lead to esophagitis/stricture formation, diffuse esophageal spasm and a hypertonic upper esophageal sphincter. Symptoms of dysphagia are likely secondary to stricture formation from severe GERD and reduced peristalsis. Another complication of GERD is Barrett's esophagus. A series of patients studied by Katzka et al showed a prevalence of 37% for endocscopically documented Barrett's metaplasia in scleroderma patients. 12 Treatment of GERD in scleroderma patients is similar to the treatment of idiopathic GERD. 10 Behavioral modification and acid suppression are the cornerstones.
Prokinetic drugs may also be helpful. A study of metoclopromide therapy in 12 patients with GERD and scleroderma showed a modest improvement in GERD by increasing LES pressure and increasing gastric emptying. 13
Esophageal dysfunction in the form of abnormal motility studies has been reported in up to 90% of patients with scleroderma. 10 However, a review of 364 cases by Poirier et al showed that one fourth of asymptomatic patients had an abnormal esophogram. The diagnosis can be made based on clinical symptoms, barium swallow, and endoscopy. 11 However, the gold standard for diagnosis is esophageal manometry. The motility abnormalities were initially thought to result from infiltration and replacement of smooth muscle fibers by collagen. However, a pathologic review of cases by D'Angelo et al failed to show this abnormal collagen deposition. There have been observations made to suggest latent cholinergic nerve dysfunction. 14
Other esophageal manifestations of scleroderma include pill-induced esophagitis and infectious esophagitis. The most frequently implicated pills are non-steroidal anti-inflammatory drugs (NSAIDs), quinidine, and potassium chloride. Candida, Herpes Simplex and Cytomegalovirus esophagitis is usually seen only in patients treated with immunosuppressive therapy. However, colonization may occur in many patients with scleroderma.
Stomach
The stomach is not usually a primary source of symptoms in gastrointestinal scleroderma. However, dysmotility of the stomach has been documented in scleroderma patients in two independent studies.10,15–16 Specifically, gastirc motility studies have shown a decrease in the phase III migrating myoelectric conmplex.10,17 Severe involvement can lead to gastroparesis. The symptoms tend to be intermittent with long periods of remission. Treatment generally involves dietary modification, prokinetic drugs, and antiemetics.
Although dysmotility is the most frequent effect of scleroderma, bleeding is also a known complication. Severe bleeding has been shown to occur from mucosal telangiectasias. 18 Gastric antral vascular ectasia (GAVE) has also been shown to be a complication of scleroderma. 19 Clinical suspicion of GAVE should be high in patients with iron deficiency anemia. Treatment for GAVE involves laser photocoagulation or argon plasma coagulation.
Small Intestine
Small bowel manifestations of scleroderma have been reported to occur in as many as 50% of patients.10,20–22 Pathophysiology studies on the small bowel point towards an abnormal response to mechanical and humoral stimuli as the cause of the small bowel dysfuction. 23 Another study showed that abnormal postcibal antral and small bowel motility can be attributed to neuropathy and/or myopathy in systemic sclerosis. 17
The clinical presentation includes symptoms of chronic intestinal pseudo-obstruction, bacterial overgrowth, and malnutrition. These clinical manifestations are the result of reduced peristalsis with resulting bowel dilatation and stasis. Chronic intestinal pseudo-obstruction presents as recurrent signs and symptoms of obstruction in the absence of a true mechanical obstruction. Treatment of uncomplicated pseudo-obstruction generally involves bowel rest, decompression, nutritional support and prokinetic agents. Patients with small bowel bacterial overgrowth can present with symptoms of abdominal pain, bloating, flatulence, diarrhea and weight loss. In severe cases, nutrient malabsorption may occur. Although limited by several factors, the gold standard for diagnosis is the jejunal aspirate. Other diagnostic tests that may be used are 14 C xylose breath test, triolein breath test and the hydrogen breath test. The treatment of bacterial overgrowth is with antibiotic cycles to alter the intestinal flora, ideally in a way that leads to symptomatic improvement.
Large Intestine, Rectum, Anus
Colonic disease has been shown to occur in 10%-50% of scleroderma patients. The anorectum appears to be the most affected area. A comparative study of anorectal and esophageal manometry showed that anorectal motility is as frequently abnormal as esophageal motility. 24 Clinically, patients may experience constipation and fecal impaction and have rectal prolapse, megacolon, and diverticula. Other possible complications include stercoral ulcerations, colonic perforation and infarction. The pathology is thought to be collagen deposition in the mucosa and submucosa and atrophy of the muscularis externa. Atrophy of the muscular wall can lead to wide-mouthed diverticuli (true diverticuli).
Scleroderma tends to disrupt the normal cholinergic gastrocolonic response to eating. Normally, there is a spike in colonic activity within 40 minutes of eating. Scleroderma has been shown to reduce this postprandial spike in colonic activity. Early in the course of disease, this effect on the gastrocolonic response can be prevented with the administration of anticholinesterase drugs. This benefit decreases with advanced scleroderma because of significant smooth muscle atrophy.
In addition, disordered anorectal function is a key early finding in scleroderma and can result in fecal incontinence. Scleroderma can cause decreased internal anal sphincter resting pressure, reduced rectal compliance and decreased rectoanal inhibitory reflex.
Footnotes
Disclosure
The authors report no conflicts of interest.
