Abstract
Chronic idiopathic constipation (CC) and irritable bowel syndrome with predominant constipation (IBS-C) are the 2 most common conditions among functional gastrointestinal disorders. Despite current multiple therapeutic options, treatment remains challenging and dissatisfactory to many patients. Linaclotide is a novel therapeutic agent, which is a guanylate cyclase receptor agonist that stimulates water secretion from the intestinal epithelium by promoting chloride and bicarbonate efflux into the lumen through activation of the cystic fibrosis transmembrane conductance regulator. Clinical trials have demonstrated that linaclotide is effective, safe and well tolerated in patients with CC and IBS-C. This review article highlights the mechanism of action of linaclotide, reviews published literature based on a search of databases, including MEDLINE, EMBASE, and Cochrane Central Register of Controlled Trials (CENTRAL), up to February 2013, and compares its utility with other currently available agents.
Introduction
Chronic idiopathic constipation (CC) and irritable bowel syndrome with predominant constipation (IBS-C) are common conditions in North America and are often difficult to effectively treat. The prevalence of CC has been estimated to be approximately 14%, with higher rates in females, older patients and in patients of lower socioeconomic status. 1 The prevalence of IBS is approximately 11% with IBS-C comprising 22% to 35% of these individuals. 2 In addition, IBS is the most common functional gastrointestinal disorder worldwide. 3 These conditions have a negative impact on quality of life and a high resource demand on health care systems.1,4
Based on the Rome III diagnostic criteria, the presence of abdominal pain and discomfort and their association with the bowel movement is the defining feature that distinguishes IBS-C from CC. Current treatment options for the management of constipation, including diet and lifestyle modification, the use of fiber, laxatives, and more recently, the serotonin receptor agonist prucalopride and the chloride channel activator lubiprostone, do not directly ameliorate abdominal pain.5,6 Antispasmodics, tricyclic anti-depressants (TCAs) and selective serotonin reuptake inhibitors (SSRIs) have been shown to reduce abdominal pain and improve global IBS symptoms. 7 However, the anti-cholinergic property of these drugs can worsen constipation. Despite multiple treatment options, CC and IBS-C remain difficult to manage in some patients. A single agent that improves abdominal pain and discomfort as well as constipation in patients with IBS-C is not available. This remains an unmet need in the treatment of IBS-C.
Linaclotide is a guanylate cyclase C (GC-C) receptor agonist that acts locally in the gastrointestinal tract as a secretagogue. It has been found to improve colonic transit times and complete spontaneous bowel movements in patients with CC and IBS-C. In addition, it has also been shown to improve functional abdominal symptoms, such as pain, discomfort and bloating, which are major symptomatic complaints of patients with CC and IBS-C. Linaclotide represents a novel therapeutic modality for managing patients with these conditions, which are often difficult to treat. This review article highlights the molecular mechanisms, efficacy, and safety of linaclotide in the treatment of patients with CC and IBS-C.
Mechanism of Action
Linaclotide is a GC-C receptor agonist that shares its mechanism of action with the endogenous molecules guanylin and uroguanylin, and with bacterial heat stable enterotoxins. Guanylin and uroguanylin, produced by enterocytes in the duodenum and colon, are responsible for the regulation of water and electrolyte secretion in the gastrointestinal tract by binding GC-C on the luminal surface of epithelial cells. This activates the cyclic 3’,5′-monophosphate (cGMP) signaling pathway,
8
which in turn activates the cGMP-dependent protein kinase II (PKG II).9,10 PKG II activates the cystic fibrosis transmembrane conductance regulator (CFTR) that increases chloride and bicarbonate secretion from the epithelial cell
10
(Fig. 1). This subsequently promotes sodium excretion and water diffusion from the cell into the intestinal lumen, thus decreasing colonic transit time.
10
Heat stable enterotoxins produced by Escherichia coli act on the same pathway to cause diarrhea in an infected host.
11
In an in vitro study, linaclotide was found to inhibit the ability of bacterial heat stable enterotoxin to bind to GC-C, confirming that GC-C is the molecular target of linaclotide.
12
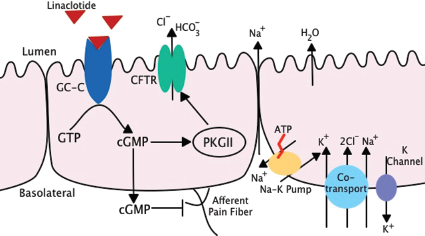
Linaclotide has also been shown to exhibit antinociceptive properties. This is an additional benefit in the treatment of IB S-C where visceral hyperalgesia is a major component of the pathophysiology of the condition. In 2 rodent models of non-inflammatory visceral pain (the acute partial restraint stress-induced colonic hypersensitivity model 13 and the acute water avoidance stress model 13 ), linaclotide significantly decreases colonic hypersensitivity as measured by a decrease in the number of colonic contractions detected by EMG in response to colorectal distension. A similar response was demonstrated in the trinitrobenzene sulfonic acid (TNBS) induced inflammatory rodent model of visceral hyperalgesia. 13 Using this model in wild type compared to GC-C receptor null mice, it was shown that linaclotide reduced colonic hypersensitivity in the wild type mice alone. This suggests that the anti-nociceptive property of linaclotide is mediated through the activation of the GC-C receptor. 13 Although the exact molecular mechanism of linaclotide's anti-nociceptive property has yet to be fully described, initial in vitro data suggest that extracellular cGMP (as produced through activation of GC-C) is able to reduce the sensitivity of colonic nociceptors to mechanical stimuli10,14,15 (Fig. 1).
Pharmacodynamics and Pharmacokinetics
Linaclotide binds to GC-C with high affinity in a pH-independent manner (Ki: 1.23-1.64 nM). 16 Linaclotide increases water secretion in surgically ligated rodent small intestine, particularly in the duodenum and jejunum. 16 In vitro studies demonstrated that the increase in cGMP stimulated by linaclotide occurred in a concentration dependent manner. The concentration of linaclotide to produce 50% of the maximal effect (EC50) was 8 to 10 fold more potent than either guanylin or uroguanylin with an EC50 of 99 nM. 16
Linaclotide is a 14 amino acid peptide that is homologous in structure to the bacterial heat stable enterotoxins. It contains 3 disulfide bonds that stabilize its molecular structure to resist degradation and enhance its ability to bind to the GC-C receptors. 17 Linaclotide acts locally within the intestine. In rodent studies, it has been shown that linaclotide is only minimally absorbed through the gastrointestinal tract with an oral bioavailability of only 0.1%. 16 In a clinical trial, the serum levels of linaclotide and its metabolite in patients who had received the drug were negligible. 18
In the intestinal lumen, linaclotide is modified by carboxypeptidase A that removes the carboxy terminal tyrosine residue to produce a 13 amino acid biologically active peptide with an increased protease resistance. 19 The half-life of the parent peptide is approximately 3 minutes while the half-life of the active metabolite is approximately 10 minutes within the intestine. 17 Reduction of the 3 disulfide bonds by the glutathione reductase system within the intestinal lumen is required for proteolytic degradation of linaclotide and its metabolite. These amino acids are absorbed by the intestinal epithelium. 19
Clinical Studies and Efficacy
Search strategy
A comprehensive literature search was conducted to identify all published human clinical studies. Abstract data were excluded and only completed studies that underwent the full, rigorous peer-review process were included. Databases were searched, including MEDLINE, and EMBASE, and Cochrane Central Register of Controlled Trials (CENTRAL), up to February 2013. Search terms, both free text and medical subject headings (MeSH), included “linaclotide” or “Linzess” or “guanylate cyclase” combined with “constipation” or “irritable bowel symptom” or “IBS” or “irritable colon”. Variations of the root word were also searched alone or in combination. A recursive search of the bibliographies of all relevant papers was also conducted. No restrictions were placed on the language of publication when searching the electronic databases.
Chronic idiopathic constipation
Summary of clinical studies of linaclotide in the treatment of chronic constipation.

In another randomized double-blind phase IIa study, 310 patients with CC were treated with 75, 150, 300 or 600 μg of linaclotide or placebo for 4 weeks. 21 The primary endpoint was an improvement in the weekly SBM rate. There was a significant increase in the weekly number of SBMs from baseline at all doses of linaclotide compared to placebo (Table 1). This study also demonstrated that linaclotide significantly improved bloating, abdominal discomfort, global measurements of constipation, treatment satisfaction, and quality of life (PAC-QOL) compared to placebo.
Two phase III double-blind, randomized, placebo controlled trials (RCTs) (trials 303 and 01) were performed to evaluate the efficacy and safety of 145 μg and 290 μg of linaclotide daily over a 12 week period in a total of 1276 patients with CC.22 In trial 303 (n = 642), 433 patients who received linaclotide were subsequently randomized to an additional 4 weeks with either the same dose of linaclotide or placebo, and those patients who received placebo (n = 209) were subsequently treated with 290 μg of linaclotide. In trials 303 and 01, patients who received 145 μg and 290 μg of linaclotide were more likely to achieve the primary endpoint (3 or more complete spontaneous bowel movements (CSBMs) per week and an increase of at least one CSBM for 9 of the 12 weeks treatment period) as compared with placebo (p < 0.001 for all treatment groups versus placebo, Table 1). The differences in treatment response between the 2 linaclotide groups were not significant (trial 303, p = 0.63; trial 01, p = 0.19). Secondary endpoints, including stool consistency, straining, abdominal discomfort, bloating, severity of constipation, relief of constipation, satisfaction with the treatment and continuation of the treatment, demonstrated statistically significant improvement in both trials at both doses compared to placebo.
Irritable bowel syndrome with predominant constipation
Summary of clinical studies of linaclotide in the treatment of irritable bowel syndrome with constipation.

Some patients reported more than 1 adverse events.
In a subsequent phase IIb study, 420 patients with IBS-C were randomized to receive 75 μg, 150 μg, 300 μg or 600 μg of linaclotide or placebo over 12 weeks 24 . There was a significant improvement in the primary endpoint, change in the number of weekly CSBMs compared to baseline, at all doses of linaclotide compared to placebo (Table 2, p < 0.01 for all doses). This study further demonstrated that all test doses of linaclotide improved stool consistency (p < 0.001), need to strain (p < 0.001) and abdominal pain within the first week of treatment (p < 0.05) compared to placebo. Furthermore, within the first week, there was an improvement in abdominal discomfort (at doses 150 μg and above), and bloating (at all doses except 150 μg). This study also demonstrated significant improvement at all doses of linaclotide in IBS and constipation severity, and in relief of IBS symptoms.
Two phase III RCTs have been published demonstrating that linaclotide improves abdominal pain and bowel function in patients with IBS-C. Rao et al randomized 800 patients to receive either 290 μg of linaclotide daily or placebo for 12 weeks. 25 This was followed by a randomized withdrawal period where patients who received linaclotide were again randomized to treatment or placebo and those who received placebo to 290 μg of linaclotide for 4 weeks. The primary endpoints were: 1) improvement by more than 30% in abdominal pain scores (referred to as abdominal pain) and an increase of at least 1 CSBM per week above baseline for at least 6 of 12 weeks of treatment (the FDA recommended endpoint for IBS-C trials); 2) at least a 30% improvement in abdominal pain for 9 of 12 weeks of treatment; 3) having at least 3 CSBMs per week with an improvement of 1 or more above baseline for at least 9 of 12 weeks; 4) and a combination of the last 2 end-points. The number needed to treat (NNT) to achieve the FDA recommended endpoint was 8 (Table 2; 33.6% in the linaclotide group, 21% in placebo, p < 0.0001). Linaclotide significantly improved abdominal pain (NNT = 13.8, p = 0.0262), and increased the number of subjects who achieved at least 3 CSBMs per week with an improvement of 1 or more above baseline for at least 9 of 12 weeks (NNT = 7.6, p < 0.0001) and the combined endpoint (NNT 14.2, p = 0.0004) compared to the placebo group. Linaclotide was found to be superior to placebo in all of the secondary endpoints, including an improvement in abdominal pain, abdominal discomfort, bloating, stool frequency and consistency, the need to strain, cramping, fullness, severity of IBS symptoms and constipation, the degree and adequacy of relief from IBS symptoms and patient satisfaction (p < 0.0001). It also illustrated that those who remained on linaclotide during the withdrawal period continued to demonstrate benefit from treatment, while those that were randomized to receive placebo during the same time period had a return of IBS-C symptoms.
Another phase III RC randomized 804 patients to receive 290 μg of linaclotide or placebo daily for a 26-week treatment period. 18 This study had the same primary and secondary endpoints as the trial outlined above by Rao et al. 25 It was found that 33.7% of treated patients achieved the FDA recommended end-point compared to 13.9% in the placebo treated group (p < 0.0001) with a NNT of 5.1 (Table 2). Abdominal pain improved in 38.9% of treated patients in 20 of 26 weeks compared to 19.6% in the placebo group (NNT = 5.2, p < 0.0001). Three or more CSBMs with an improvement of 1 or more above baseline was achieved in 18.1% of treated patients for at least 20 of 26 weeks compared to 5.0% in the placebo group (p < 0.0001). The combined endpoint was found in 12.7% of treated patients versus 3.0% in the placebo-group (p < 0.0001). As in the previous study, linaclotide was superior to placebo in all of the secondary endpoints at 26 weeks (p < 0.0001). A pooled analysis of the 2 phase III IBS-C RCT trials,18,25 which specially evaluated the European Medicines Agency (EMA) specified endpoints, demonstrated that linaclotide significantly improved abdominal pain/discomfort and the degree of relief in IBS symptoms compared with placebo over 12 and 26 weeks 26 (Table 2).
Tolerability and Safety
The most common adverse event reported in all clinical trials is the development of diarrhea (Tables 1 and 2). In all of the phase III clinical trials in patients with CC and IBS-C, there were no statistically significant differences seen for treatment emerging adverse events between the linaclotide group and the placebo, except in the Chey et al trial 18 in IBS-C patients (65.4% in linaclotide group vs 56.6% in the placebo group, p < 0.05). Subsequent post-hoc analyses combining the Rao and Chey trials did not show any significance. 26 The phase III trials in patients with CC showed that 16% of patients receiving linaclotide 145 μg and 14.2% of patients receiving linaclotide 290 μg developed diarrhea compared to 4.7% in the placebo control group. 22 In the IBS-C phase III trials, the incidence of diarrhea occurred in approximately 1-in-5 patients, with a number needed to harm (NNH) of 5.8-6.5. 25 Increase in flatulence (4.9% vs 1.5%, p = 0.0084), and abdominal pain (5.4% vs 2.5%, p = 0.0462) were also higher in the linaclotide treated group versus the placebo. 25 Patients required to discontinue the study drug due to diarrhea was seen in 4.5-5.7% in the linaclotide group compared to 0.2-0.3% in the placebo group. There was no significant difference between the groups in terms of serious adverse events. In all phase III trials, there was no difference between the treatment and placebo groups in terms of laboratory investigations, electrocardiogram changes or vital signs.18,22,25
Place of Linaclotide in Therapy of CC and IBS-C
The initial non-pharmacological treatment in patients with CC and IBS-C is lifestyle and diet modification. Although regular physical activity has not been consistently shown to improve stool frequency, it has been found to improve quality of life and the severity of constipation in patients with CC27,28 and the symptom severity scores of IBS patients. 29 Psyllium has been shown to be moderately effective in improving constipation but it does not improve overall symptoms of IBS. 30 Osmotic and stimulant laxatives are effective in improving stool frequency. A recent meta-analysis, including 5 double-blind RCTs using osmotic laxatives (1 used lactulose and 4 used polyethylene glycol) and 2 double-blind RCTs using stimulant laxatives (1 used bisacodyl and 1 used sodium picosulfate), showed that both osmotic and stimulant laxatives improve constipation with a NNT equal to 3. 31 However, none of these agents improve functional abdominal symptoms. Although the use of osmotic laxatives is recommended by the American College of Gastroenterology (ACG) 7 and the UK National Institute of Health and Clinical Excellence (NICE) in the treatment of constipation associated with IBS, NICE discourages the use of lactulose because it may exacerbate IBS symptoms. 32 The common side effects of laxatives, such as abdominal pain, bloating, flatulence and diarrhea, limit their uses.
Lubiprostone has been approved in the United States for the treatment of CC and IBS-C. It is a bicyclic fatty acid that activates the apical membrane chloride channel in the intestinal epithelium to stimulate intestinal fluid secretion. The NNT of lubiprostone to improve constipation was shown in a meta-analysis, including 3 RCTs, to be 4. 31 In an extension study, following 522 IBS-C patients up to 52 weeks, diarrhea and nausea were found in 11% of patients. 33
Serotonergic (5-HT4) receptor agonists have been shown to be effective in the treatment of patients with CC and IBS-C. Cisapride and tegaserod were 2 previously widely used prokinetics. Due to their cardio-toxicity, both cisapride and tegaserod have been withdrawn from the market. A new 5-HT4 agonist, prucalopride, has shown to be effective in the treatment of CC with a NNT of 6. 31 Based on pharmacokinetic and pharmacodynamics, as well as clinical trial data, including trials in the elderly with cardiac co-morbidities,34,35 prucalopride has been shown to be well tolerated and safe without any significant cardiovascular side effects. In addition, 3 double-blind, placebo controlled RCTs 36 38 have demonstrated that not only does prucalopride improve constipation, it also improves patients’ QoL and constipation symptoms, particularly abdominal discomfort, bloating and pain. 39 Nevertheless, currently, prucalopride has only been approved for the treatment of CC and not for IBS-C.
TCAs and SSRIs are recommended by the ACG to treat abdominal pain in patients with IBS. 7 A systematic review has shown that TCAs and SSRIs improve global IBS symptoms and abdominal pain. 40 However, a meta-analysis, including 5 RCTs of SSRIs, demonstrated that SSRIs do not significantly improve abdominal pain or IBS symptoms. 41 Furthermore, the anticholinergic side effects of TCAs and SSRIs limit their tolerability.
Linaclotide offers a novel therapeutic option for the treatment of CC and IBS-C. Its visceral hyperalgesia modulatory effect makes it an exciting single agent to be used in patients with IBS-C. Recently, linaclotide has been approved by the FDA to treat CC and IBS-C and by the EMA to treat IBS-C. In Germany, linaclotide (290 μg) is indicated for the symptomatic treatment of moderate to severe IBS-C in adults. In the United States, it is approved to use in adults for the treatment of IBS-C (290 μg) and CC (145 μg). There has been no clinical trial comparing linaclotide to lubiprostone or prucalopride in patients with CC or IBS-C. Its place in the pharmacological treatment algorithm in these functional GI disorders will depend on the local availability of these various agents, as well as the cost of linaclotide and coverages by public and private programs. In the treatment of CC, it is reasonable to consider linaclotide only after patients cannot be adequately treated by fiber supplementation and osmotic laxatives in the treatment pyramid. 5 In patients with IBS-C, particularly those predominantly suffering from abdominal discomfort and pain, it is sensible to consider using linaclotide earlier in the treatment algorithm, even before laxatives.
Conclusion
Linaclotide represents an exciting novel therapeutic option for the treatment of CC and IBS-C. Clinical trials demonstrated that linaclotide is well tolerated and improves bowel function in addition to offering symptomatic relief, which other therapies often fail to provide, in patients with CC and IBS-C. Now that linaclotide has been approved by the FDA and EMA, further studies (e.g. direct comparative RCTs to established therapies and cost-effective decision models) and consensus expert working groups to determine its place in treatment algorithms will help in further optimizing the management paradigm for these functional GI disorders.
Author Contributions
All three authors contributed to the first draft and jointly developed and approved the final manuscript.
Funding
Author(s) disclose no funding sources.
Competing interests
In the past 48 months, Dr. Louis Liu has served on the advisory board for Janssen Pharmaceuticals, Nycomed Canada, Takeda Canada Inc, Abbott Canada and Forrest Laboratory Canada, Inc. and on the speaker bureau for Janssen Pharmaceuticals. Dr. Yuhong Yuan and Colleen Parker disclose no potential conflicts of interest.
Footnotes
As a requirement of publication the authors have provided signed confirmation of their compliance with ethical and legal obligations including but not limited to compliance with ICMJE authorship and competing interests guidelines, that the article is neither under consideration for publication nor published elsewhere, of their compliance with legal and ethical guidelines concerning human and animal research participants (if applicable), and that permission has been obtained for reproduction of any copyrighted material. This article was subject to blind, independent, expert peer review. The reviewers reported no competing interests. Provenance: the authors were invited to submit this paper.
