Abstract
The link between signaling pathways and diseases suggests the importance of pathway analysis for drug discovery. This includes target identification and validation, compound mode of action and drug candidate optimization. Here, we propose to apply cell signaling pathway panel approaches for oncology drug discovery. The strategies and guiding principles of the pathway panel approach are discussed. 2 pathway analysis examples with related processes and technology platforms are illustrated to identify cancer drugs that target cancer growth and metastasis. Finally, we highlight potential challenges and opportunities presented by the pathway panel approach.
Major cell signaling pathways are conserved from yeast to humans due to their critical roles in development and adult homeostasis. In the last 30 years, pathway deregulation has been implicated in multiple disease areas, especially cancer. Loss of control of cell cycle, apoptosis, growth and survival signaling pathways such as MAPK or Wnt signaling have been identified as some of the hallmarks of cancer and metastasis.1,2 Therefore, therapies targeting those signaling pathways have been widely applied to treat cancer.
Over the last decade, applications of signaling pathway approaches have had a significant impact on drug discovery and development, 3 especially in the areas of tumorigenesis and metastasis. Targeting signaling pathways identified as cancer hallmarks enabled the discovery of multiple marketed drugs, such as Bcr/Abl kinase inhibitors, imatinibmesylate (Gleevec; Novartis) and dasatinib (Sprycel, Bristol Myers Squibb). These drugs have been demonstrated to inhibit a key signaling target or multiple targets. As a result, they modulate the aberrant signaling activities linked with diseases. Subsequently, great efforts have been undertaken in both academic labs and pharmaceutical industries to identify novel signaling components in tumorigenesis and metastasis pathways using siRNA and chemogenomic approaches.4–7 Therefore, during drug discovery processes, monitoring the outcomes of targeting cell signaling becomes essential since the readouts can be applied for target identification and validation, as well as lead identification and assessment. Furthermore, the optimization of drug candidates and deep dissection of mechanisms of action require cellular measurements of the signaling pathway activities. Recently, advancements in two major areas enabled the applications of pathway approaches to target cancer growth and metastasis. The first area is the advance of next-generation sequence mutation analysis for cancer cell lines and primary tumors, which reveals cancer pathway deregulation in specific cellular contexts. The second area is the development of high-throughput technology platforms for pathway signaling readouts, ranging from receptor activation to downstream transcriptional readouts.
It is well known that cancer cell lines contain mutations in oncogenes and tumor suppressors, which lead to deregulated critical signaling pathways involved in cell growth and survival, apoptosis and cell cycle regulations (http://sanger.ac.uk/genetics/CGP/cosmic/CellLines). Diverse cellular backgrounds with different signaling pathway deregulation mechanisms confer different drug sensitivities.8,9 Therefore, it is essential to examine signaling pathway readouts in a panel of cancer cell lines with aberrant signaling pathways instead of in a couple of cancer cell lines only. We initiated the efforts of monitoring cellular signaling in a panel of cells during our drug discovery process. Those cell lines have been selected for cancer drug screening, 10 since each of these cell lines has mutations linked to distinct deregulated oncogenic pathways, such as apoptosis and MAPK signaling pathways. For example, P53 mutant status might determine if a drug affecting apoptosis pathway will be effective or not, considering the link of P53 and apoptosis pathways. Also, APC mutation or beta-catenin mutation might show different responses to a drug targeting Wnt signaling pathway based on its mechanism of action. Therefore, it is important to have a whole panel of cell lines for signaling pathway readouts to obtain comprehensive readouts for signaling events. This would translate better into a clinical setting, where the tumors in patients are truly heterogeneous. Sensitive and resistant tumor types can be identified based on those results. Furthermore, this signaling pathway panel approach would facilitate patient stratification and identify mechanisms of patient drug resistance.
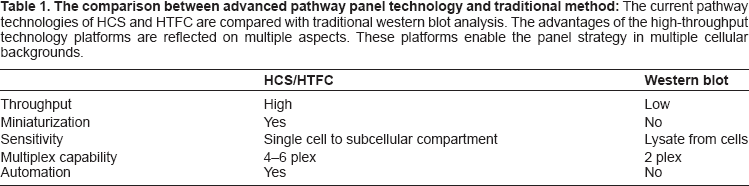
The comparison between advanced pathway panel technology and traditional method: The current pathway technologies of HCS and HTFC are compared with traditional western blot analysis. The advantages of the high-throughput technology platforms are reflected on multiple aspects. These platforms enable the panel strategy in multiple cellular backgrounds.
Apoptosis Pathway Panel
Apoptosis is the process of programmed cell death that serves critical functions for the development and homeostasis of multicellular organisms. Loss of apoptotic control could lead to tumorigenesis, and resistance to apoptosis is considered as a hallmark of cancer. Therefore, a myriad of efforts have been taken to target apoptosis for treatment of different types of cancers, as demonstrated by recent publications on prostate cancer, 14 Glioblastoma 15 and BCL-2–dependent hematological cancers, 16 among others.
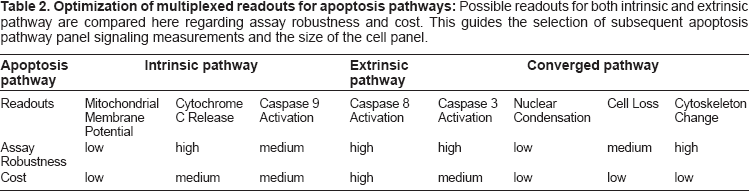
Optimization of multiplexed readouts for apoptosis pathways: Possible readouts for both intrinsic and extrinsic pathway are compared here regarding assay robustness and cost. This guides the selection of subsequent apoptosis pathway panel signaling measurements and the size of the cell panel.
With the assay platform to dissect the mechanisms of action for apoptosis in place, we selected a panel of tumor cells with different mutations linking to apoptosis deregulation such as P53 and PI3K. 18 This apoptosis panel approach was then applied to target identification and validation, followed by hit identification, confirmation as well as lead optimization. Further, this apoptosis pathway panel was used to dissect mechanisms of action for our potential cancer drugs and to demonstrate mechanisms of differentiation. As expected, diverse apoptosis activities and mechanisms were obtained with potential drug candidates in different cellular backgrounds, indicating the importance of a cellular panel approach, instead of performing drug discovery in only a limited number of cell lines.
Wnt Signaling Pathway Panel
Wnt signaling pathway plays critical roles in a wide spectrum of important biological processes, ranging from early development of an organism to adult homeostasis. Aberrant deregulation of Wnt signaling pathway leads to multiple diseases, especially cancer.19,20 It was reported in the early 1980s that MMTV-Wnt mice demonstrated tumorigenesis indicating the oncogenic nature of activating this signaling pathway. 21 More recently, Nguyen and colleagues have reported that the Wnt pathway might be important for lung adenocarcinoma metastasis to the brain and bone. 22 Therefore, targeting Wnt signaling pathway is not only beneficial to primary cancer treatment, but also may confer advantages in preventing metastasis.
The canonical Wnt signaling pathway consists of the following well studied key signaling events: Upon Wnt signaling, the ligand binds to its receptors, namely, the 7-transmembrane receptor Frizzled and the single-transmembrane receptor LRP6. The ligand-receptor complex initiates the signaling cascade by activating the downstream component Disheveled.23,24 This activation leads to the interaction between Disheveled and Axin. 25 Subsequently, β-catenin is dissociated from its destruction complex consisting of Axin, GSK3β and APC. Being released from the Axin complex, β-catenin is no longer phosphorylated by GSK3β, and thus accumulates in the cytoplasm. 26 As a result, β-catenin is stabilized and translocated into the nucleus, which marks the key signaling event of the canonical pathway. Subsequently, β-catenin binds to LEF/TCF like transcription factors in the nucleus and activates downstream target genes. 27 To monitor Wnt canonical pathway activation, high content assays for β-catenin nuclear translocation were established in different cellular backgrounds, and LEF/TCF reporter assays for signaling events at the transcriptional levels were developed. Once again, the pathway panel strategy is impactful by developing these readouts in a panel of cell lines which consist of not only cells responding to Wnt ligand signaling, but also cells with known mutations for APC or β-catenin, which leads to constitutively activated Wnt signaling. Wnt signaling pathway inhibitors or activators, such as naturally occurring soluble Wnt antagonists sFRP and DKK or GSK3β inhibitors, demonstrated expected activities in those cell lines in the presence or absence of Wnt signaling, respectively.
Pathway Panel Automation
A key enabler for pathway panel approaches is to leverage automation. We applied the automated cell culture system SelecT for cell panels relevant to each signaling pathway (Fig. 1). Not only are the cells maintained by this automated system, but they are also plated by the system into assay plates. Subsequently, these assay plates are loaded onto the assay robotic system, which consists of liquid handlers, incubators as well as ECHO nano-dispenser for compound addition (Fig. 2). Finally, readers such as Envision for luciferase readouts or ArrayScan for high content analysis can be integrated onto the assay robotic systems to fully automate the assay process. Furthermore, all the assays can be performed in 384-well format with automation systems that not only increase the capacity for compound evaluation, but also enable miniaturization to reduce the cost for each assay.
Automated cell culture system –-SelecT: Cell culture automation with capacity to process more than 60 cell lines and up to 175 flasks enabled the cell line panel approach for signaling readouts. Cells can also be plated into 384 assay plates using this technology platform. Automated high throughput assay robotic system for pathway readouts: Automation processes for reagent addition, compound dispense and assay detection enable signaling panel approaches for compound evaluation in multiple cell lines.

Conclusion
Applying cell-signaling pathway panel strategies is challenging since it is a novel approach. Automation processes need to be optimized in multiple cell lines, while best technological solutions need to be applied. For example, pathway readouts using high content analysis require a large amount of antibodies if examined in a panel of cell lines. The cost of those antibodies and the amount of information generated can be daunting. Solutions for further assay miniaturization or novel technological solutions, as well as new IT tools for pathway data analysis and viewing, become important aspects to be further addressed. Furthermore, pathway networking and signaling cross talk add another level of complexity to dissect signaling events using panel strategy. Leveraging the proper bioinformatic tools related to pathway analysis, as well as incorporating information from literature and other public domain sources, are essential to ensure the success of applying signaling pathway panel strategy in drug discovery and development.
Recently, translational approaches have attracted more attention, and pathway approaches can play an import role in translational research. Future directions of technology enhancement include breakthrough technology platforms such as multiplexing signals at the pathway level, cell arrays for large sample sizes, and patient stratified pathway analytics for personalized medicine. In addition, combinatorial approaches to target pathways are a new direction that attracts significant interest. Finally, pathway genomic and proteomic databases from cancer patients and pre-clinical animal models provide valuable resources for target selection and validation strategies. Today, this growing pathway knowledge is leading to opportunities to target oncogenic pathways directly. Gene expression signatures for pathway activation, such as the EGFR pathway and beta-catenin pathway, 28 have enabled both oncology drug discovery and clinical research to monitor those markers for targeted pathway modulation and to identify responders to anticancer drugs. Moving forward, one can envision expanded efforts in this area for both drug discovery and development.
Author Contributions
Both LS and LZ conceived and contributed to the writing of the manuscript.
Funding
This work is supported by Bristol Myers Squibb Company.
Competing Interests
Authors disclose no potential conflicts of interest.
Disclosures and Ethics
As a requirement of publication the authors have provided signed confirmation of their compliance with ethical and legal obligations including but not limited to compliance with ICMJE authorship and competing interests guidelines, that the article is neither under consideration for publication nor published elsewhere, of their compliance with legal and ethical guidelines concerning human and animal research participants (if applicable), and that permission has been obtained for reproduction of any copyrighted material. This article was subject to blind, independent, expert peer review. The reviewers reported no competing interests. Provenance: the authors were invited to submit this paper.
Footnotes
Acknowledgements
We thank members from LEMBC for their contributions to cancer pathway screening efforts under this panel strategy, as well as collaborations with automation and IT groups.
