Abstract
Generalized lipodystrophy (GL) is a rare inherited or acquired disease characterized by widespread loss of subcutaneous fat, leading to leptin deficiency, ectopic fat deposition, and severe metabolic abnormalities. Previous studies have shown the benefit of leptin replacement (metreleptin) in ameliorating metabolic complications, but little is known about the experience of metreleptin treatment outside of a research setting. We report on post-marketing clinical experience with metreleptin therapy in three patients with GL and marked hypoleptinemia, uncontrolled diabetes, and hypertriglyceridemia. After metreleptin treatment for 12–168 weeks, the mean glycated hemoglobin decreased from 10.9% to 5.8%, and serum triglycerides were normalized (a mean decline of 90%). These benefits were observed within weeks of starting therapy, were durable, and were accompanied by subjective improvements in quality of life, decreased need for concomitant medications, and no significant adverse effects. Metreleptin was safe and effective in normalizing certain severe metabolic abnormalities in the clinic setting.
Introduction
Generalized lipodystrophy (GL) is categorized as a rare congenital generalized lipodystrophy (CGL) or acquired generalized lipodystrophy (AGL) disease characterized by widespread loss of adipocytes, leading to leptin deficiency, ectopic fat deposition, and severe metabolic abnormalities (insulin resistance, diabetes, and/or hypertriglyceridemia).1,2 In women with insulin resistance and hypoleptinemia, polycystic ovarian syndrome, hyperandrogenism, and amenorrhea are very common. 3 Conventional glucose- and lipid-lowering medications do not address the underlying leptin deficiency and are often inadequate at addressing severe metabolic abnormalities. 1
Metreleptin is a recombinant human leptin analog that binds to and activates the leptin receptor, and it is the only US Food and Drug Administration-approved treatment for patients with CGL or AGL as an adjunct to diet and as replacement therapy to treat complications of leptin deficiency. 4 Metreleptin is not currently indicated for the treatment of partial lipodystrophy, liver disease, human immunodeficiency virus-related lipodystrophy, or diabetes and hypertriglyceridemia without evidence of concurrent CGL or AGL. The starting daily dose of metreleptin varies by weight and sex (Table 1), and daily dosing may be increased or decreased according to clinical response (changes in glycated hemoglobin [A1C], triglycerides, etc.). Per the US prescribing label, metreleptin should be administered once daily (QD) at the same time every day. In clinical trial experience, the most common adverse reactions (≥ 10% incidence) included headache, hypoglycemia, decreased weight, and abdominal pain.
Metreleptin dosing per the US Package Insert. 4

There is limited experience using metreleptin to treat patients with GL in the clinic setting. Herein, we present the metreleptin treatment experience in three patients with GL inadequately managed with conventional medications. Patients or their guardians gave consent for the publication of this report.
Case Report 1
Patient 1 was a 19-year-old female of Arabic descent and consanguineous parentage diagnosed with CGL at early infancy (serum triglycerides >2000 mg/dL at two months) Table 2. Prior to metreleptin, she presented with generalized subcutaneous fat loss, muscular appearance, acanthosis nigricans, elevated alanine aminotransferase (ALT; 47 U/L), hepatomegaly (confirmed later by magnetic resonance imaging scan: moderate hepatomegaly with fatty infiltration), irregular menstrual cycles (three to four per year), increased abdominal girth, and elevated A1C and triglycerides (11.8% and 3675 mg/dL, respectively; Table 1). These metabolic abnormalities were noted despite ongoing therapy with metformin 500 mg three times daily (TID), pioglitazone 30 mg QD, insulin detemir 40 U twice daily (BID), insulin aspart 40 U TID, fenofibrate 145 mg QD, niacin 500 mg QD, omega-3 fatty acids 2 g TID, and orlistat 120 mg TID for four years.
Baseline demographics, concomitant medications, and metabolic parameters prior to metreleptin treatment and at last measurement.

Most recent value was at age 12 and was decreased from 32.9 ng/mL at age 11.
Metreleptin was initiated at 5 mg QD subcutaneously by self-injection. A1C and triglycerides were markedly improved at four weeks from 11.8% to 9.7% and 3675 to 2118 mg/dL, respectively (Fig. 1). At 12 weeks, while maintained at the same metreleptin dose, A1C and triglycerides dropped further to normal levels (5.7% and 121 mg/dL, respectively), and slightly elevated ALT at baseline was within normal limits (23 U/L). The normalization of laboratory values occurred while the patient discontinued insulin aspart, pioglitazone, niacin, and fenofibrate (insulin detemir was decreased from 40 to 30 U BID, and the dosing frequency of omega-3 fatty acids was reduced to QD). Weight remained stable, hepatomegaly and abdominal girth were reduced, and the patient reported resumption of regular menstrual cycles for the last two months. No adverse events (AEs) were noted.
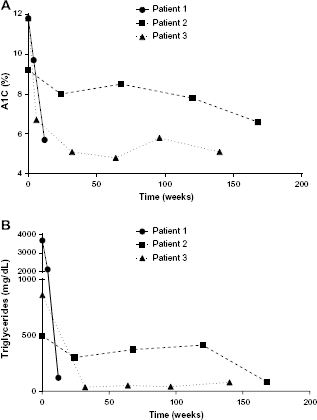
Changes in (
Case Report 2
Patient 2 was a 10-year-old female with CGL diagnosed from birth with a history of pancreatitis (four episodes requiring hospitalization), lytic bone lesions, and mesangial proliferative glomerulonephritis (Table 2). Prior to metreleptin treatment, she presented with generalized subcutaneous fat loss, acanthosis nigricans, elevated liver enzymes (ALT: 132 U/L; aspartate aminotransferase [AST]: 80 U/L), prominent abdomen due to hepatosplenomegaly (abdominal ultrasound demonstrated enlarged liver, homogeneous parenchyma, and decreased echogenicity), decreased energy, proteinuria (2.5 g/24 hours), and elevated A1C and triglycerides (9.2% and 497 mg/dL, respectively; Table 1). Her metabolic abnormalities were managed with metformin 500 mg/day, regular U100 insulin 1000 U/day, fenofibrate 200 mg/day, and enalapril 20 mg/day for 21 months.
Metreleptin was initiated at 4.2 mg/day while the patient was enrolled in research protocol 02-DK-0022 at the National Institutes of Health. At 24 weeks, A1C and triglycerides were reduced from 9.2% to 8.0% and 497 to 302 mg/dL, respectively (Fig. 1), and the patient's proteinuria was resolved (0 g/24 hours). As A1C was still elevated, the patient's metreleptin and metformin doses were increased in a stepwise manner over consecutive visits to 10 mg/day and 2000 mg/day, respectively. At week 168, the patient's metabolic parameters had improved (A1C: 6.6%; triglycerides: 84 mg/dL), and her total daily insulin requirement was reduced from 1000 to 75 U/day. ALT and AST were within normal limits (20 and 18 U/L, respectively). The patient's weight increased by 10%, and she self-reported improved energy levels, a flat abdomen, and that she completed her pubertal development with her first menses. No AEs were noted.
Case Report 3
Patient 3 was a 19-year-old male with AGL manifesting between eight and nine years of age with panniculitis in the abdomen, legs, and face, followed later by generalized loss of subcutaneous fat (Table 2). He presented with acanthosis nigricans, prominent abdomen, acromegaloid features, hyperphagia, asthenia, elevated liver enzymes (ALT: 171 U/L; AST: 81 U/L), hyperinsulinemia (fasting: 228 μU/mL), and elevated A1C and triglycerides (11.0% and 861 mg/dL, respectively). Abdominal ultrasound demonstrated an enlarged spleen and enlarged liver with smooth contour, hyperechoic texture, and no focal hepatic lesions. Metformin 500 mg/day was initiated and titrated later to 2000 mg/day. High insulin levels persisted (≤300 μU/mL) in parallel with worsening acanthosis, and he was started on regular U500 insulin 120 U/day (equivalent of regular U100 insulin 600 U/day). The patient was on this treatment regimen for seven years, and his last A1C measurement before metreleptin treatment was 11.8%. He was also managed with fenofibrate 200 mg/day, vitamin K 5 mg/month (due to vitamin K deficiency related to steatohepatitis), ranitidine 150 mg/day, ursodiol 300 mg/day, and enalapril 5 mg/day.
Metreleptin was initiated at 3.5 mg/day. At six weeks, the patient's A1C was reduced from 11.8% to 6.7% (Fig. 1). At 32 weeks, A1C was further reduced to 5.1% and triglycerides were reduced to 36 mg/dL. At 96 weeks, his A1C increased to 5.8%. It was noted that the patient gained 6 kg of weight during this time, so the metreleptin dose was increased to 4 mg/day. As of the last measurement (week 140), A1C was 5.1% and triglycerides were 80 mg/dL. During treatment with metreleptin, the patient was able to discontinue the use of insulin and fenofibrate, remaining only on ursodiol 300 mg/day and enalapril 10 mg/day. ALT levels were reduced from 171 to 120 U/L, while AST remained stable (from 81 to 79 U/L). Follow-up hepatic ultrasonography indicated increased spleen size with homogeneous structure and liver with rounded edges, enlarged, homogeneous-increased echogenicity compatible with steatosis, no focal lesions, and dilated intra- and extrahepatic bile ducts. The patient's weight increased from 61 to 67 kg with improvements in lean muscle mass observed. His appetite normalized allowing him to adhere to the recommended low-glucose diet for diabetes control, and his acanthosis nigricans was noted to have improved while on metreleptin. No AEs were noted, and the patient continues to remain on the same daily dose of metreleptin (4 mg/day).
Discussion
All three patients with GL had significant metabolic abnormalities that were inadequately addressed with conventional glucose- and lipid-lowering therapies. Metreleptin was effective at normalizing very elevated A1C and triglycerides, with this response occurring as early as four to six weeks in Patients 1 and 3. Metreleptin dose was individualized to match changes in A1C and triglycerides, and concomitant medications were adjusted to avoid hypoglycemia. In parallel with improved metabolic parameters, all patients were able to either discontinue or decrease their daily doses of insulin – an important point as patients with lipodystrophy sometimes require very high doses of insulin to maintain glycemic control. Only Patient 2 required an increase in metformin dose. Patients 1 and 3 were able to discontinue oral lipid-lowering medications. High pill burden in conditions requiring long-term treatment has been associated with decreased adherence and quality of life (QoL). 5 The success of metreleptin therapy is dependent on good adherence to metreleptin and concomitant medications besides dietary modifications and other lifestyle changes. 6 Patients with lipodystrophy who are nonadherent to metreleptin and/or concomitant medications often experience relapses to previous metabolic derangements. 6 8 Metreleptin does not cure the underlying disorder of lipodystrophy but addresses the associated leptin deficiency; thus, most patients may require lifetime therapy.
Although difficult to quantify, patients with lipodystrophy may have decreased QoL 9 owing to the metabolic abnormalities that they experience, particularly hyperphagia. Compliance with diet may be difficult because of hunger related to the leptin deficiency, implying that hypothalamic satiety is also inhibited. During metreleptin treatment, Patients 1 and 3 reported marked reductions in hunger, and Patients 2 and 3 reported improved energy levels. While similar subjective findings have been reported in at least one other cohort of patients with GL, 10 hyperphagia response to metreleptin has not been quantified in any previous clinical trials to date. Cosmetic disfigurement due to loss of adipose tissue and leptin deficiency can be very discouraging. In this case series, Patients 1 and 2 reported a less prominent abdomen or decreased girth during metreleptin treatment. The same patients also reported more regular menstrual cycles (Patient 1) or the achievement of first menses (Patient 2). Acanthosis nigricans was observed to have improved in Patients 2 and 3. Metreleptin is not approved for the treatment of hyperphagia or any other QoL parameters.
In addition to improved A1C and triglycerides, elevations of liver enzyme were numerically improved or normalized in all patients, with decreased size of hepatomegaly observed in Patient 1. At least one study group has described significant improvements in liver pathology from baseline with metreleptin treatment11,12; however, improved liver enzymes do not always correlate with improved liver-related end points. 13 Patient 2 had proteinuria at baseline that resolved after metreleptin treatment. Metreleptin has been shown in a prior cohort of patients with GL to reduce urinary protein excretion in 11 of 15 patients treated for four months. 14
In the patients with GL presented here, metreleptin was effective at improving certain metabolic abnormalities in the clinic setting with no notable AEs. Subjective QoL improvements (increased energy, normalization of appetite/menstrual function, improved appearance, etc.) are equally important to the patient treatment experience, as are improvements in laboratory parameters.
Author Contributions
Provided care for Patient 1: VS. Provided care for Patients 2 and 3: CM, MLM, EA. All authors participated in manuscript development. They also reviewed and approved the final version prior to submission.
Footnotes
Acknowledgments
Robert Schupp, PharmD, CMPP, of inScience Communications, Springer Healthcare (Philadelphia, PA, USA), provided medical writing support funded by Aegerion Pharmaceuticals, Inc.
CM would like to acknowledge Dr. Phillip Gorden and Elaine Cochran of the National Institutes of Health who helped to provide care for Patients 2 and 3 in this case series and for their collaboration.
