Abstract
A 46-year-old Japanese female expatriate living in Jakarta presented with intermittent fever lasting for a month. Although she was considered at low risk of Fasciola spp. infection because she lived in an upper-class residential area of the city, the patient presented with eosinophilia after consuming organic raw vegetables; in addition, contrast-enhanced computed tomography detected microabscesses in a tractlike pattern in the liver. These findings led to an early diagnosis of fascioliasis, which was successfully treated without sequelae. In any patient with a history of consuming raw vegetables, fascioliasis should be suspected regardless of where the patient has lived.
Introduction
Fascioliasis is a worldwide zoonosis caused by Fasciola spp. and is often neglected despite its common occurrence in endemic areas.1,2 If detected at an early stage, fascioliasis can be treated successfully; however, the disease in the advanced stages can cause liver fibrosis, hepatic cirrhosis, and sometimes death.3,4 Therefore, early detection and treatment are important. However, owing to its nonspecific symptoms and observational findings, human fascioliasis is difficult to diagnose.5–7 Some clinicians have limited experience with human fascioliasis in nonendemic countries. And this situation leads to misdiagnosis or delayed treatment. We herein report a case of fascioliasis in a traveler from Jakarta, Indonesia, who was diagnosed at an early stage based on a detailed history of the patient, eosinophilia, and the characteristic findings on computed tomography (CT), resulting in successful treatment without any sequelae. Informed consent was obtained from the patient.
Case Report
A 46-year-old Japanese woman without a medical history expatriated to Jakarta, Indonesia, for 5 years. She lived in an upper-class residential area, in a clean environment. Around the time she returned to Japan, 1.5 months prior to her visit to our clinic, the patient started experiencing a dry cough, which subsided after several days. One month prior to her visit, she developed a fever of 40 °C that persisted for several days accompanied by malaise, followed by fever and malaise every week. Two days prior to her visit, the patient experienced fever and epigastric pain. The patient had no history of consumption of raw seafood; however, the patient had been eating organic raw vegetables on a daily basis, including watercress.
At admission, her body temperature was 35 °C, vital signs were stable, and the liver was enlarged and tender. She had eosinophilia (eosinophil count, 3,465 cells/μL) and mild hepatic dysfunction (aspartate transaminase, 37 U/L; alanine transaminase, 60 U/L; γ-glutamyl transpeptidase, 70 U/L; and alkaline phosphatase, 398 U/L). Hyperbilirubinemia was absent. Three of the stool specimens were negative, the latter for ova, cysts, and parasites, examined under a microscope. Enhanced CT showed multiple microabscesses arranged in a tractlike pattern with ring enhancement in the right lobe of the liver (Fig. 1). Endoscopic retrograde cholangiopancreatography did not show worms in the bile duct, and no ova were detected in the bile examined under a microscope. The diagnosis of fascioliasis was confirmed by demonstration of specific antibodies to Fasciola sp. using the Ouchterlony double-diffusion test (Fig. 2). 8

Enhanced abdominal CT findings in a 46-year-old woman with fascioliasis on admission. The white arrow shows multiple nodular cysts with tubular branching.

The specific serological examination for fascioliasis using the Ouchterlony double-diffusion test. The black arrow shows a strong precipitin band between the crude antigen of Fasciola sp. and the patient's serum. It means the patient has a large amount of antibody for Fasciola sp. However there is no precipitin band between the crude antigen of Fasciola sp. and the control serum (the white arrow).
For treatment, 500 mg (10 mg/kg) triclabendazole (Egaten™, Novartis Pharma AG, Switzerland) was orally administered two times at 12-h intervals. After the initiation of triclabendazole administration, the fever and malaise resolved immediately. Her eosinophil count decreased to 1,966/μL in 1 month, 624/μL in 2 months, and 273/μL in 3 months, reaching the normal range. Contrast-enhanced CT performed a year later showed almost complete disappearance of the multiple nodular lesions (Fig. 3).
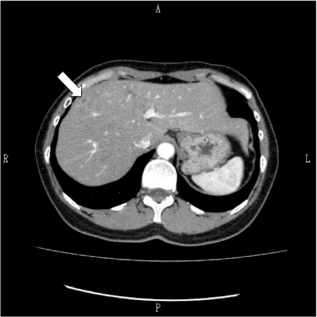
Follow-up enhanced abdominal CT findings of a 46-year-old woman with fascioliasis after 1 year of treatment. The multiple nodular cysts observed at admission are almost no longer visible (the white arrow).
Discussion
Fascioliasis is a common trematodiasis distributed worldwide mainly in human-endemic areas and animal-endemic areas. 9 However, in several nonendemic countries such as Japan, most clinicians lack experience and knowledge about human fascioliasis. Therefore, the disease can sometimes be excluded from differential diagnosis and overlooked.6,7
Adult Fasciola spp., the causative agent for fascioliasis, inhabit the bile duct of ruminants such as cattle and sheep, which then pass their ova in stool. The eggs become embryonated in fresh water. The eggs release miracidium. The miracidium develops and invades a suitable snail (Galba/Fossaria group, genus Radix and Pseudosuccinea) as an intermediate host. In the snail, the miracidium develop to the sporocysts, rediae, then cercaria. The cercaria are released from the snails and encyst as metacercaria on aquatic plants such as watercress.3,5 Humans can accidentally become infected as the final host by eating raw aquatic plants or drinking water contaminated with metacercaria. 1 Therefore, the incidence of fascioliasis is considered scarce in urban areas but concentrated in rural areas, where these conditions are likely to be met. 4
The present patient had lived in a clean environment in an urban area of Jakarta. Thus, the risk of becoming infected with Fasciola spp. would have been considered low. However, she had routinely consumed organic raw vegetables grown at an organic farm that also raised cattle. Therefore, contaminated organic raw vegetables were considered the source of infection.
When ingested by humans, immature larvae invade the abdominal cavity via the small-intestinal wall 2 hours after ingestion, grow into juveniles, reach the liver within 6 days after excystment, and migrate into the liver through the hepatic capsule, causing the formation of linear cystic lesions. 1 In the liver, juveniles migrate for 5–6 weeks. This is considered the invasive or acute phase1,10 and is distinctive on CT, 11 as in the present case (Fig. 1). During this phase, the patient might experience right hypochondrial pain, fever, nausea, vomiting, and cough, and eosinophilia is common. 1 Because the patient in the present case exhibited all these characteristics, she was considered to be in the invasive or acute phase. The biliary phase follows next, when juveniles migrate into the bile duct where they become sexually mature and deposit eggs at least 6 months from the beginning of the invasive or acute phase.1,10 It is possible to detect the eggs in both stool and bile. Chronic-stage fascioliasis may be asymptomatic but can develop into irreversible liver fibrosis or liver cirrhosis as a result of recurrent cholangitis caused by worm-induced biliary obstruction. 3 Adult worms can also cause irreversible lesions by invading various organs other than the liver, such as the gastrointestinal tract, abdominal wall, lung, heart, brain, and eye (ectopic fascioliasis). 3
Because of the effects in the more advanced biliary phase, it is important to detect and treat fascioliasis early. While the presence of worms or ova provides a direct diagnosis of fascioliasis, a patient in the invasive or acute phase does not have worms in the bile duct or ova in the bile. Therefore, even if worm or ova is negative, this result could not rule out the possibility of fascioliasis. If the clinician does not have knowledge of the lifecycle of fascioliasis, diagnosis is difficult during the invasive or acute phase, which can consequently lead to a misdiagnosis or delayed diagnosis. 5 Although fascioliasis during the invasive or acute phase can only be definitively diagnosed via serological testing, contrast-enhanced CT can sometimes significantly help, as in the present case. However, clinicians or radiologists who have limited experience with human fascioliasis often misdiagnose lesions observed on contract-enhanced CT as a malignant tumor, causing the patient to undergo liver biopsy or hepatectomy.6,7 Therefore, it is very important that clinicians and radiologists outside endemic regions be aware of the typical contrast-enhanced CT findings of fascioliasis. 6
Triclabendazole is the first-line treatment for fascioliasis, 1 with a cure rate of 78%–100% at doses of 10 mg/kg. 12 Unlike other helminth infections, treatment with praziquantel is not effective. Triclabendazole resistance concerned livestock in animal-endemic areas. 1 Fortunately, the symptoms of our patient disappeared rapidly and eosinophilia and CT findings were improved, and there was no triclabendazole in our case.
The present case with fascioliasis was diagnosed and treated at an early stage of infection based on a detailed patient history, eosinophilia, and typical CT findings. Fascioliasis is no longer a disease limited to human-endemic areas or animal-endemic areas. It might be diagnosed in any areas across the world. Therefore, all clinicians should acquire knowledge about fascioliasis. Fascioliasis should be considered in the differential diagnosis of any patient who consumes raw vegetables and presents with gastrointestinal symptoms and eosinophilia, regardless of the patient's residence and living situation. The characteristic contrast-enhanced CT findings may provide a clue to the potential presence of fascioliasis, and treatment should be initiated immediately after a serological diagnosis.
Author Contributions
Conceived and designed the experiments: MN, TS. Contributed to the acquisition of the clinical data: MN, NT, TK, TK, YF, YK, HM. Analyzed the data: NT, TK, TK, YF, YK, HM. Wrote the first draft of the manuscript: MN. Contributed to the writing of the manuscript: TS, YM. Agreed with manuscript results and conclusions: MN, TS, HG, YM, NT, TK, TK, YF, YK, HM. Jointly developed the structure and arguments for the paper: MN, TS. Made critical revisions and approved final version: MN, TS, HG, YM, NT, TK, TK, YF, YK, HM. All authors reviewed and approved of the final manuscript.
