Abstract
Introduction
Sertoli-Leydig cell tumors (SLCTs) are rare tumors mostly occurring in young women. Here we report an unusual case of a SLCT with simultaneous occurrence of endometrioid adenocarcinoma of the endometrium in a woman in menopause.
Case Report
A 67-year-old woman presented with progressive signs of virilization. Blood tests showed increased levels of testosterone, delta-4-androstenedione, and dehydroepiandrosterone (DHEA). DHEA-sulphate, 17β-estradiol, estrone, and sex-hormone binding globulin serum levels were within the normal range. Magnetic resonance imaging revealed a solid mass of 2.7 × 2.9 cm in the right ovary set against the background of the uterus. The patient underwent bilateral salpingo-oophoretomy with hysterectomy. The mass in the right ovary was a differentiated SLCT. Incidentally, the endometrium revealed an endometrioid adenocacinoma. Following surgical treatment the plasma androgens dropped to normal levels, and signs and symptoms of virilization improved.
Conclusion
SLCT should be suspected in postmenopausal women who present rapid progressive androgen excess symptoms with hyperandrogenemia.
Introduction
Sertoli-Leydig cell tumors (SLCTs) are uncommon neoplasms, accounting for 1% of all sex cord-stromal tumors (0.1%–0.5% of all primary ovarian neoplasms). 1 Some of these tumors are well differentiated, produce steroid hormones, and may be suspected in patients with estrogen-excess symptoms (precocious puberty, abnormal uterine bleeding, and endometrial hyperplasia or endometrioid carcinoma) or androgen-excess signs (hirsutism, virilism, acne, hyperseborrea, and alopecia) associated with the presence of an ovarian mass. Benign, well-differentiated SLCTs often occur in young women with an average age of 25 years; less than 10% of the patients are over 50 years of age.1,2 They account for 10% of all SLCTs and are often associated with either congenital anomalies of internal genitalia or with testicular feminization syndrome.3,4 Microscopically, the tumors show uniform solid or tubular structures lined by Sertoli-type cells. Tubules may contain eosinophilic secretion. The intervening stroma contains a variable number of Leydig cells that tend to be packed in ribbons or nests between the tubules. Mitoses are rare. The neoplasm grading is an important predictive factor related to both prognosis and post-surgical follow-up of the patient. Here we report a rare case of simultaneous occurrence of SLCTs with endometrial carcinoma in a postmenopausal woman.
Clinical Case
A 67-year-old female was evaluated because of a progressive increase of hair growth of upper lip, chin, and linea alba, deepening of voice, increase in libido, mood instability, increment of muscle mass, and onset of frontal alopecia during the last six months. A transvaginal ultrasonography performed the previous year showed no abnormalities in her reproductive tract. She had two pregnancies, and menopause took place at the age of fifty. Her medical history included a total thyroidectomy for goiter and cholecystectomy in 2006, irritable bowel syndrome, and severe osteopenia treated with calcium (1000 mg daily) and ibandronate (150 mg once a month). Body weight was stable (body mass index, 30.4 kg/m2) 5 and the blood pressure was normal (130/70 mmHg). On physical examination, a clitoromegaly, increased hair growth (20 on the Ferriman-Gallwey scale), 6 and marked frontal alopecia were confirmed.7,8 The blood tests showed normal values of hemoglobin, haematocrit, and eritrocyte number and no abnormalities in lipid profile, serum electrolytes, and glucose level. Serum alpha fetoprotein (AFP), CA15-3, CA 125, CA 19.9, CEA, hepatic enzymes, and alkaline phosphatase were all within the normal range. Hormonal profile 9 revealed increased total serum levels of testosterone (T) (4.33 ng/mL, normal range in women, 0.15–0.7), delta-4-androstenedione (3.5 ng/mL, normal range, 0.5–1.5), and DHEA (6.2 ng/mL, normal range, 0.7–3.1). The following were within the normal range: 17β-estradiol (14.6 pg/mL, normal range for menopause, <18 to 110), estrone (28 pg/mL, normal range for menopause, 7–40) follicle-stimulating hormone (82.96 mIU/mL, normal range for menopause, 2.58–150.53), luteinizing hormone (28.09 mIU/mL, normal range for menopause, 1.78–92.10), DHT (38.4 pg/mL, normal range, 35–185), sex hormone binding globulin (27.5 nmol/l, normal range, 20–90), ACTH (20 pg/mL, normal range, 5–60), cortisol (10.2 µg/dL, normal range, 7–25) and prolactin (12 ng/mL, normal range, 2–29). A 2 mg overnight dexamethasone suppression test was performed. T, androstenedione and DHEA plasma levels remained unvaried. Transvaginal ultrasonography showed a solid, ovoid, sharp margins well-vascularized mass. The abdomen contrast magnetic resonance imaging (MRI) confirmed a solid and heterogeneous mass of 2.7 × 2.9 cm in the right ovary, characterized by a hypo-intense core set against the background of the uterus with regular margins. The left ovary was normal. The patient underwent hysterectomy with bilateral salpingo-oophorectomy. The pathology study confirmed the presence of an encapsulated yellow solid lobulated tumor of 3.5 cm, of hard-parenchymatous consistency on the right ovary. Furthermore, a polipoid neoplasm of the endometrium (1 cm size) was found in the surgical specimen of the uterus. Microscopical examination of the neoplasm located in the right ovary revealed a proliferation of tubular structures lined by one layer of cuboidal epithelial cells with Sertoli cell appearance, amphophilic vacuolated clear cytoplasm, oval basal nuclei, and scant nucleoli (Fig. 1A). The intervening stroma contained nests of variable numbers of large monomorphic cuboidal cells with eosinophilic cytoplasm and central located nuclei with the aspect of Leydig cells (Fig. 1A). Mitotic figures, vascular invasion, and necrosis were not observed. Immunohistochemistry showed strong positivity of the neoplasm for inhibin and pancytokeratine (Fig. 1B and C) and negativity for vimentine, placental alkaline phosphatase, synaptophysin, cytokeratine 20, and alpha-fetoprotein; less than 5% of tumor cells were positively stained for the proliferation marker Ki67/ MIB-1. The final diagnosis was SLCT of the right ovary pT1a/FIGO IA. Concerning the neoplasm found in the endometrium, the diagnosis was endometrioid adenocarcinoma (G2 pT1B/FIGO IA, FIGO 2009 staging) (Fig. 1D). Two weeks after surgery, the patient reported a gradual and a progressive regression of alopecia and a recovery in voice tone. The treatment with adjuvant chemotherapy or radiation was not performed, and the patient underwent follow-up checkup every three months. Twelve months postoperatively, the patient was in good health, alopecia (Fig. 2A and B) and hirsutism (Fig. 2C and D) had substantially improved, and total serum T levels (0.8 ng/mL), delta-4- androstenedione (0.8 ng/mL), and DHEA (1.5 ng/mL) were within the normal range. MRI with and without contrast of the upper and lower abdomen did not show pelvic or distant pathological processes. Mammography and pelvic and breast ultrasound were normal. A written consent was obtained from the patient to reproduce information and photographs appearing in this work.
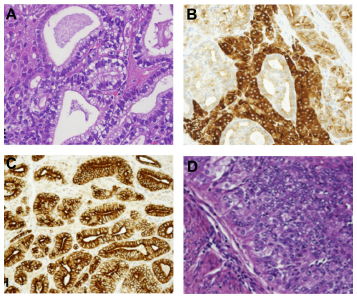
Microscopy and immunohistochemistry of the SLCT of the right ovary (

Effect of salpingo-oophoretomy with hysterectomy on alopecia and hair growth on linea alba. (
Discussion
Here we report a case of SLCT associated with endometrial carcinoma in a postmenopausal woman with hyperandrogenism. During the reproductive age, the differential diagnosis of hyperandrogenemia includes polycystic ovary syndrome (PCOS), ovarian and adrenal androgen-secreting tumors, ovarian and adrenal steroidogenic enzyme deficiencies, as well as other endocrine disorders. In a postmenopausal woman, the main reason for a fast-developing hirsutism is cancer. The most common ovarian cancers related to hyperandrogenism are ovarian sex-cord stromal tumors, an extremely rare cause of virilization, comprising 5% to 8% of all primary ovarian neoplasm's. 10 Rarely, SLCTs may be associated with hyperestrinism. 11
An important aspect of our case report was the concomitant presence of SLCT and endometrioid adenocarcinoma of endometrium. Endometrial carcinomas are hormone-dependent tumors associated with high circulating levels of estrogens. The paradox consists in the evidence that endometrial carcinomas are a disease of aging with over 80% of cases occurring during menopause, a time during which estrogen secretion is waning. 12 However, despite the decline of the ovarian function that follows menopause, estrogen synthesis continues during the postmenopausal years through increases of estrone formation from androgens of adrenal and ovarian origin. 12 The reaction that takes place in the adipose tissue and enhances with body weight and advanced age may produce high concentrations of estrogens. Despite the high levels of androgens, we did not find high levels of estrogens in our patient. However, a role of hyperandrogenemia as a risk condition for endometrial cancer development must be considered. Androgens are involved in many regulatory processes in the endometrial epithelia. 13 The androgen receptor has been found both in normal endometrium, endometrial hyperplasia, and endometrial adenocarcinoma, and DHT plays an important role in the regulation of androgen action in endometrial cancer and normal human endometrium. 14 Furthermore, a local endometrial conversion of androgens to estrogens due to the high endometrial aromatase P450 expression 15 cannot be excluded.
An alternative and intriguing explanation for the concomitant development of SLCT and uterine tumor in our patient could be provided by mutations of the STK11/LKB1 gene at chromosome 19p13.3. Germline STK11 gene mutation is the underlying genetic alteration responsible for most cases of Peutz-Jegher's syndrome (PJS),16,17 a rare autosomal dominant disorder characterized by mucocutaneous pigmentation, hamartomatous polyposis, and predisposition to benign and malignant tumors of the gastrointestinal tract, breast, ovary, uterus, and testis. 18 Women with PJS have an increased incidence of SLCT and adenocarcinoma of the uterine cervix, 19 but STK11/LKB1 gene mutations may drive human endometrial carcinogenesis as well.20,21 Since allelic losses at chromosome region 19p13.3 occur in at least a subset of sporadic cases of SLCT, 19 this has generated substantial interest in evaluating sporadic cancers of the same type as those observed in PJS patients for mutations in this gene. Thus, a potential role of a mutation in the STK11/ LKB1 gene in our patient cannot be ruled out.
In conclusion, SLCTs are rare functionally active ovarian neoplasms, which must be suspected in post-menopausal women who present rapid progressive androgen excess symptoms and virilization with hyper-androgenemia. The concomitant presence of endometrial carcinoma despite the normal circulating levels of estrogens suggests a direct androgen action on the endometrium. Accordingly, endometrial cancer risk among postmenopausal women was positively associated with increasing levels of testosterone. 22 However, a local conversion of excess androgens to estrogens as potential stimulants of endometrium proliferative activity cannot be ruled out. To the best of our knowledge, this is the first report 23 describing the hormonal status in a postmenopausal woman affected by this rare condition.
Author Contributions
Conceived and designed the experiments: PDG, AF, LG, CM. Analysed the data: PDG, LC, GV, LG, EC, LG, CM. Wrote the first draft of the manuscript: SM, AF, LG, CM. Contributed to the writing of the manuscript: LG, CM. Agree with manuscript results and conclusions: PDG, LC, GV, LG, EC, SU, SM, CA, AF, LG, CM. Jointly developed the structure and arguments for the paper: LG, CM. Made critical revisions and approved final version: LG, CM. All authors reviewed and approved of the final manuscript.
Funding
Author(s) disclose no funding sources.
Competing Interests
Author(s) disclose no potential conflicts of interest.
Disclosures and Ethics
As a requirement of publication author(s) have provided to the publisher signed confirmation of compliance with legal and ethical obligations including but not limited to the following: authorship and contributorship, conflicts of interest, privacy and confidentiality and (where applicable) protection of human and animal research subjects. The authors have read and confirmed their agreement with the ICMJE authorship and conflict of interest criteria. The authors have also confirmed that this article is unique and not under consideration or published in any other publication, and that they have permission from rights holders to reproduce any copyrighted material. Any disclosures are made in this section. The external blind peer reviewers report no conflicts of interest.
