Abstract
Evaluation of airway inflammation is important in achieving adequate dosing of inhaled corticosteroids (ICS) for treating bronchial asthma. However, there is no evaluation tool that can be used in clinical settings. We examined biomarkers that can precisely reflect airway inflammation when ICS are decreased in stable asthmatic patients. This was a 12-week, single-arm, open-label clinical study performed at a single university hospital. Twenty-five patients (6 male and 19 female) with stable asthma were included in this study. We investigated whether the levels of nitrite and nitrate in exhaled breath condensate (EBC) increase after ICS reduction. We also investigated whether blood eosinophils, serum immunoglobulin E (IgE), high-sensitivity C reactive protein (hs-CRP), interleukin (IL)-13, IL-17, and periostin are different before and after ICS reduction. Peak expiratory flow (PEF), pulmonary function tests, asthma control test (ACT), and asthma quality of life questionnaire (AQLQ) were also examined. We considered an unscheduled hospital visit due to asthmatic symptoms and decline in average PEF over one week by more than 10% to indicate disease instability, and compared patients with stable and unstable disease for analysis. Unstable status was detected in 5 patients. Age, sex, asthma duration, ACT and AQLQ scores, and the level of serum IgE did not differ between stable and unstable groups. In the unstable group, the total concentration of nitrite and nitrate at the last visit was 9.84 (6.65–11.24) μM. Surprisingly, this was similar to the concentration at the first visit (5.58 (2.94–17.29) μM). Serum periostin before ICS reduction (141.9 [107.7–147.7] pg/mL) was higher in the unstable group than in the stable group (91.5 [78.75–103.5] pg/mL). The unstable group had a higher peripheral blood eosinophil count and wider diurnal variation of PEF at the first visit compared to the stable group. Higher eosinophils in peripheral blood and wider diurnal variation of PEF were predictive markers for unstable disease after ICS reduction. Serum periostin is another candidate for the predictive marker.
Introduction
Bronchial asthma is a chronic disorder characterized by airway hyperresponsiveness. 1 Th2 cytokines and eosinophilic inflammation of the airway play important roles in the pathogenesis of bronchial asthma.2–4 The effectiveness of ICS for asthma control is widely recognized; inhaled corticosteroids (ICS) are now used as first-line therapy for airway eosinophilic inflammation and hyperresponsiveness in asthmatic patients. 5 However, adequate dosing with ICS according to disease activity is important for achieving and maintaining ideal disease control in asthmatic patients. Eosinophilic infiltration in bronchial biopsy specimens, the number of eosinophils in induced sputum, and airway hyperresponsiveness to nonspecific stimuli are often used as indicators of airway inflammation in an investigational setting. However, it is difficult to repeatedly use such procedures to evaluate actual asthmatic activity in a clinical setting. Several more convenient methods have been proposed to assess the activity of bronchial asthma. However, the efficacy and accuracy of these methods remain unknown.
Nitric oxide has been suggested as a sensitive marker of airway inflammation. The fractional concentration of exhaled nitric oxide (FeNO) has been shown to correlate with eosinophilic inflammation and disease severity.6,7 However, patients with allergic rhinitis also have higher FeNO levels regardless of asthma control. 8 These findings indicate that FeNO levels are influenced by both upper airway events and asthmatic inflammation.
Collection of exhaled breath condensate (EBC) is another possible technique to assess asthmatic activity in a clinical setting. Several markers in EBC have been investigated to evaluate airway inflammation.7,9 Matsunaga et al reported that cytokines, including regulated on activation normal T cell expressed and secreted (RANTES), tumor necrosis factor (TNF)-α, and transforming growth factor (TGF)-β, in EBC can be used as inflammatory markers of asthma. 10 The stable end products of nitric oxides, nitrite and nitrate, in airway surface liquid can also be measured in EBC.11,12 These have been observed to increase in asthmatic patients.13,14 Evaluation of end products of NO in collected EBC has some advantages over analysis of FeNO. First, nitrite and nitrate in the liquid phase are much more stable than NO in the gas phase. Second, EBC, collected during 10 to 15 minutes of restful breathing, appears to reflect the condition of the lower airway more precisely than FeNO analysis, which only uses one breath.
This study was conducted to identify promising tools for evaluating asthmatic activity that can be used frequently in a clinical setting. Hence, we measured nitrite and nitrate in EBC at the first visit and 12 weeks after dose reduction of ICS in a prospective study. We also compared respiratory and hematological parameters and their interval change between patients with worsened disease and those with good asthma control.
Methods
Study objectives
The primary endpoint of this study was to investigate whether the levels of nitrate and nitrite in EBC increase 12 weeks after the reduction of ICS. Secondary end-points were to examine whether other possible markers for asthma activity reflect the effect of ICS reduction, and to determine which marker can predict worsening of asthma control. Markers tested included peak expiratory flow (PEF), reversibility of forced expiratory volume (FEV1), asthma control test (ACT), and asthma quality of life questionnaire (AQLQ). Peripheral blood eosinophil counts and serum levels of IgE, high-sensitivity C-reactive protein (hs-CRP), interleukin-13 (IL-13), IL-17, and periostin were also tested.
Subjects
Patients with stable bronchial asthma on ICS therapy and more than 20 years of age were recruited. All patients met the following criteria: well controlled asthmatics, less than 20% diurnal variation in PEF, no disease exacerbation, and no treatment with systemic corticosteroids in the previous 3 months. Exclusion criteria were patients with a smoking history of more than 5 pack-years, chronic obstructive pulmonary disease, and administration of systemic corticosteroids or omalizumab. All patients provided written informed consent, and the study was approved by the medical ethics committee of Saga University. Recruitment was performed from December 2011 to May 2012.
Study design
This was a 12-week, single-arm, open-label clinical study performed at a single university hospital. The study design was as shown in Figure 1, with patients undergoing five clinic visits at 4-week intervals. At visit 0, consent was obtained and the study visits were explained. The dose of ICS was reduced according to Global Initiative for Asthma (GINA) guidelines at visit 1. For example, in patients receiving a high dose of ICS, the dose was reduced to a medium dose. Other drugs were continued at the same dose and no new drugs were added. At visits 2–4, we assessed the status of the patient's disease by assuming that an unscheduled hospital visit due to asthmatic symptoms or a decline in the average PEF for the week by more than 10% indicated unstable asthma.

Study design.
Pulmonary function test
Pulmonary function testing before and after inhalation of a bronchodilator (procaterol) was performed using spirometry (H101, Chest MI, Inc., Tokyo, Japan) according to American Thoracic Society (ATS) guidelines. Patients kept diaries of their PEF during the study. Patients measured their PEF every morning and evening. The mean morning and evening PEFs for the week were calculated separately at each visit.
Assessment of asthma control and asthma-related QOL
Asthma control and asthma-related QOL were assessed via a self-reported questionnaire. Asthma control was assessed by the ACT, while asthma-related QOL was assessed by the AQLQ. 15 Permission for usage of the Japanese version of AQLQ was obtained from Astra-Zeneca (London, UK).
EBC collection
EBC was collected using an R-tube™ (Respiratory Research, Inc., Austin, TX, USA). Patients were told to breathe through the mouthpiece of the R-tube for 15 minutes while maintaining eupnea. Collected samples were immediately frozen at −80 °C and thawed for a total of one freeze-thaw cycle before use.
Measurement of nitrite and nitrate
The concentrations of nitrite and nitrate in EBC were measured using a fluorometric assay kit based on the Griess reaction (Dojin Chemical Laboratories. Kumamoto, Japan). After duplicated samples were incubated with Griess reagent, the intensity of fluorescence was measured at an excitation wavelength of 360 nm and emission wavelength of 450 nm.
ELISA for cytokines
Serum samples were frozen at −80 °C after collection and thawed immediately for a total of one freeze-thaw cycle before use. Serum IL-13 levels were measured using a human enzyme-linked immunosorbent assay (ELISA) kit (R&D Systems, Inc., Minneapolis, MN, USA), serum IL-17 A levels were measured with a human high sensitivity ELISA kit (eBioscience, Inc., San Diego, CA, USA) and serum CRP levels were measured using a high sensitivity human ELISA kit (Immunology Consultants Laboratory, Inc., Newberg, OR, USA).
Measurement of serum periostin
Serum periostin concentration was measured by ELISA as previously described. 16 Periostin concentrations were calculated by simultaneously measuring recombinant periostin proteins.
Statistical analysis
Data are presented as the median, with ranges from 25th percentile to 75th percentile in parenthesis. Differences between unpaired samples were analyzed using the Mann-Whitney U test. Differences between paired groups were analyzed using the Wilcoxon signed-rank test. Since the sample sizes for statistical comparisons were relatively small, power (1 – β) was calculated using a post hoc analysis with the G3 computer program. All tests were two-sided with a 10% level of significance. Statistical calculations and data plotting were carried out with SPSS software (SPSS, Inc., Chicago, IL, USA).
Results
Patient characteristics
Twenty-five patients (6 male and 19 female) were enrolled in this study. The median age of patients was 64 (51–71) years and duration of asthma was 16 (6–28) years. Five patients were judged to have unstable disease after reduction of ICS. One patient had asthma exacerbation after an upper respiratory infection, which required an unscheduled hospital visit. The other four were found to have a fall in mean PEF at visit 4. When baseline characteristics of unstable patients were compared with those of stable patients, there were no significant differences in gender, age, onset of asthma, duration of asthma, and usage of long acting
Baseline characteristics of the patients.
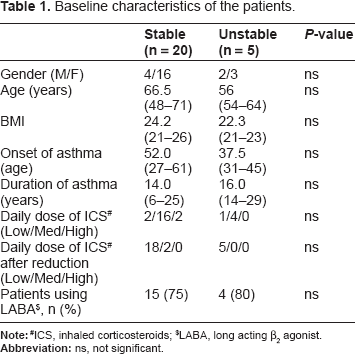
ICS, inhaled corticosteroids;
LABA, long acting β2 agonist.
The effect of ICS reduction on symptoms and physiological parameters
As shown in Table 2, ACT scores decreased significantly in the unstable group after ICS reduction, with a score of 25 (24–25) at the first visit and 22 (21–24) at the last visit. In the stable group, in contrast, the score at the last visit was comparable to the score at the first visit (24 (22–24) and 24 (23–24), respectively). AQLQ total score and symptom score worsened significantly in the unstable group after ICS reduction, while the scores remained unchanged in the stable group.
Asthma control test (ACT) and asthma quality of life questionnaire (AQLQ) scores before and after ICS reduction.

As expected, the weekly averages of morning PEF at the last visit were significantly lower than those before ICS reduction (297 (258–320) and 344 (310–360), respectively) in the unstable group (Table 3). Conversely, the weekly evening PEFs at the last visit were slightly higher than those at the first visit (331 (262–350) and 321 (275–410), respectively). In the stable group, in contrast, the weekly averages of morning and evening PEFs did not change throughout the study period. Although daily PEF variation at baseline was significantly higher in the unstable than in the stable group, the amount of variation did not increase after ICS reduction in both groups.
Pulmonary function tests and peak expiratory flow (PEF) before and after reduction of ICS.
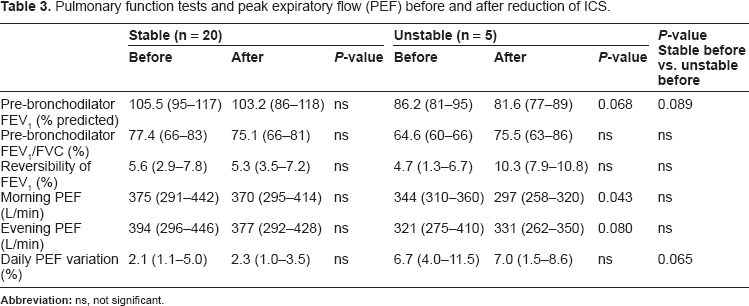
In the unstable group, FEV1 before bronchodilator inhalation was 86.2 (81.3–94.6)% predicted at the first visit, decreasing to 81.6 (77.1–88.9)% predicted at the last visit. FEV1 before bronchodilator inhalation did not change in the stable group, with values of 105.5 (94.9–117.4)% predicted and 103.2 (86.0–117.5)% predicted at the first and last visits, respectively. FEV1/FVC did not change after ICS reduction in both groups. Reversibility of FEV1, as an indicator of the airway's response to bronchodilators, was 4.7 (1.3–6.7)% at the first visit and 10.3 (7.9–10.8)% at the last visit in the unstable group, while the interval change in the reversibility did not reach significant levels. Reversibility of FEV1 at the first and last visits was comparable in the stable group.
The effect of ICS reduction on nitrite and nitrate levels in EBC
We hypothesized that an increase in EBC nitrite and nitrate levels reflected increased airway inflammation after ICS reduction. Hence, we compared the concentrations of nitrite and nitrate in EBC after ICS reduction with those at the first visit separately in the stable and unstable groups. Data are presented as the median, with ranges from 25th percentile to 75th percentile in parenthesis in Figures 2–4. In the stable group, nitrite concentrations at the last visit (1.78 (0.49–3.15) μM) were comparable with those at the first visit (1.84 (1.03–3.85) μM) (Fig. 2). Nitrate, and thus the total nitrite and nitrate, concentrations at the last visit (0.69 (0.51–1.23) μM and 2.29 (1.14–4.33) μM, respectively) were lower than those at the first visit (1.27 (0.96–2.48) μM and 3.74 (2.53–5.81) μM, respectively). In the unstable group, the concentrations of nitrite, nitrate, and the total of nitrite and nitrate at the last visit were 4.49 (3.28–5.37) μM, 1.58 (1.57–6.75) μM, and 9.84 (6.65–11.24) μM, respectively. Unexpectedly, these values did not differ from the concentrations at the first visit (7.87 (2.30–10.73) μM, 2.59 (1.33–4.53) μM, and 5.58 (2.94–17.29) μM, respectively).
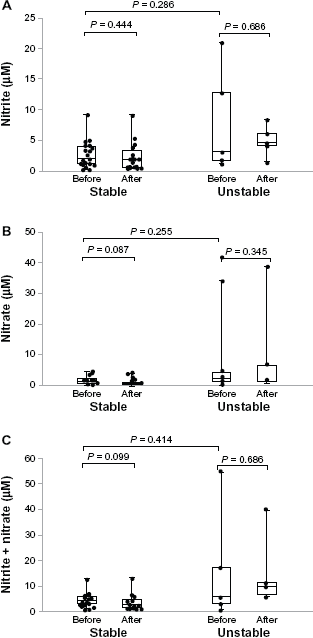
Nitrite and nitrate levels in exhaled breath condensate (EBC).

Peripheral blood eosinophils.

Serum hs-CRP and periostin levels.
The effect of ICS reduction on biomarkers
Peripheral blood eosinophil levels at the first and last visit are shown in Figure 3. Both the percentage as well as the absolute number of eosinophils did not change after ICS reduction in both groups. Serum IL-13 was undetectable in all patients even after ICS reduction. Serum IL-17 was measured in 10 of 25 patients at the first visit, while no significant change was noted after ICS reduction in both groups (data not shown). The level of hs-CRP at the last visit was similar to that at the first visit in both groups (Fig. 4A). Serum periostin levels, measured using ELISA, were 91.5 (78.8–103.5) pg/mL at the first visit and 138.6 (119.3–144.5) pg/mL at the last visit in the unstable group, and 141.9 (107.7–147.7) pg/mL at the first visit and 93.0 (85.2–119.5) at the last visit in the stable group. The interval change in serum periostin level was not significant in both groups.
The difference between unstable and stable groups before ICS reduction
We also investigated markers that can predict worsening of asthma control before ICS reduction. Compared to the stable group, lower pre-bronchodilator FEV1 and higher daily PEF variation were noted in the unstable group. Peripheral blood eosinophils were increased in the unstable group. There was no difference in the concentration of nitrite and nitrate in EBC between both groups. Interestingly, serum periostin levels were higher in the unstable than in the stable group before ICS reduction.
Discussion
ICS are commonly used as first-line treatment for bronchial asthma.17–19 In a previous clinical study, asthmatic patients with well-controlled disease for more than 3 months were able to step down their ICS dose according to GINA guidelines. 20 PEF and ACT are frequently used to evaluate the adequacy of control of bronchial asthma.21,22 Further, although quantification of sputum eosinophils is a powerful method for assessing airway inflammation, 23 the process of inducing sputum is invasive for some patients. In fact, using standard methods, sputum production was induced in only 62% of asthmatic patients in a previous study. 24 Therefore, more sensitive and convenient markers, particularly biomarkers which reflect asthmatic activity, have been sought in the last decades. The primary endpoint of this study was to investigate whether the levels of nitrate and nitrite in EBC increase after the reduction of ICS. In this study, 5 of 25 patients (20%) had worse control after reduction of ICS. Unexpectedly, nitrite and nitrate in EBC failed to reflect the increased disease activity after ICS reduction in the unstable group, as some patients in the unstable group had rather high levels of these products even before ICS reduction. We also examined whether other possible markers for asthma activity reflect the effect of ICS reduction. As was observed for nitrogen oxides, peripheral blood eosinophils and serum periostin levels were elevated in the unstable group even before ICS reduction. Serum IL-13 and IL-17 were undetectable in many patients. Hs-CRP could be detected, although it did not reflect asthmatic activity. Although all patients met the criteria of ICS dose step-down, five patients had worse ACT, AQLQ, and FEV1 at final evaluation. These parameters were comparable between the two groups at the first visit. Our findings suggest that ACT, AQLQ, and pulmonary function tests are insufficient for precisely evaluating asthmatic activity and to predict worsening after ICS dose step-down. The total amount of nitrite and nitrate in EBC did not differ between the stable and unstable groups before ICS reduction. This was mainly due to the wide variation in the unstable group. Interestingly, both patients with the highest nitrate and nitrite levels were included in the unstable group, suggesting that increased levels of nitric oxides predict unstable asthma. Previous studies showed that wide daily PEF variation, 21 airway hyperresponsiveness (AHR), 24 and FEV1 25 indicate poor control after reduction of ICS. Our data also showed that wide PEF variation and low FEV1 were associated with unstable status. This suggests that parameters of pulmonary function continue to be useful in a clinical setting.
Periostin has been identified as an IL-4/IL-13- induced matricellular protein, 26 which plays an important role in the development of bones, teeth, and the heart. It is reportedly associated with subepithelial fibrosis in bronchial asthma. 27 Its relationship with atopic dermatitis, 16 pulmonary fibrosis,28,29 and bone marrow fibrosis 30 has also been reported. Recently, Corren et al reported that serum periostin is useful for predicting the efficacy of anti-IL-13 antibody (lebrikizumab) treatment in asthmatic patients. 31 When lebrikizumab treatment was administered to severe asthmatic patients, significant improvement in lung function was observed in patients with increased serum periostin levels. This study revealed the significant role of periostin in asthmatic inflammation. Our findings that higher serum periostin levels before ICS dose step-down were associated with poor control after reduction of ICS further substantiates this observation. This is the first report suggesting that serum periostin can be used as a predictive marker for the efficacy reducing ICS dose.
Conclusion
In conclusion, higher eosinophil counts in peripheral blood and wider diurnal variation of PEF were predictive markers for unstable disease after ICS reduction in stable asthmatic patients. Serum periostin is another candidate for predictive markers. However, this study was performed on a small group of patients at a single university hospital. To ensure the validity of our findings, further prospective, multicenter studies involving a large number of patients should be performed.
Author Contributions
The experimental design and carried out the experimental work: GK, KT. The data analysis and drafted the manuscript: GK. Conributed to the writing of the manuscript: KT, SH. Participated in the discussion of the experiments: KK. Agree with manuscript results and conclusion: KI. Provided overall leadership to the experimental design, data analysis: SH, SK. All authors reviewed and approved of the final manuscript.
Funding
Author(s) disclose no funding sources.
Competing Interests
SH reports grants and personal fees from Glaxo Smith Klein KK, Boehringer Ingelheim, Asteras Pharmaceutical and Astra Zeneca, and personal fees from Pfizer, outside the submitted work. Other authors disclose no potential conflicts of interest.
Disclosures and Ethics
As a requirement of publication the authors have provided signed confirmation of their compliance with ethical and legal obligations including but not limited to compliance with ICMJE authorship and competing interests guidelines, that the article is neither under consideration for publication nor published elsewhere, of their compliance with legal and ethical guidelines concerning human and animal research participants (if applicable), and that permission has been obtained for reproduction of any copyrighted material. This article was subject to blind, independent, expert peer review. The reviewers reported no competing interests.
Footnotes
Acknowledgements
We thank Dr. Edmund J. Miller (Feinstein Institute for Medical Research, NY, USA) for critical review of this manuscript. We also thank Chidori Tanaka (Saga University, Saga, Japan) and Junya Ono (Shino-Test Corporation, Sagamihara, Japan) for technical assistance with this study.
