Abstract
Erythropoietin (EPO) increases the number of circulating erythrocytes and muscle oxygenation. The recombinant forms of EPO have indiscriminately been used by athletes, mainly in endurance sports to increase their erythrocytes concentration, thus generating a better delivery of oxygen to the muscle tissue. The administration of recombinant human erythropoietin (rHuEPO) except for therapeutic use was prohibited by the International Olympic Committee (IOC) and its unauthorized use considered as doping. In the last few years, a number of studies using parameters indicative of accelerated erythropoiesis have investigated a number of indirect methods for the detection of rHuEPO abuse. No single indirect marker has been found that can satisfactorily demonstrated rHuEPO misuse. Soluble transferrin receptor (sTfR) is a new marker of iron status and erythropoietic activity. It has been included in multivariable blood testing models for the detection of performance enhancing EPO abuse in sports. Indirect markers of altered erythropoiesis give reliable evidence of current or discontinued rHuEPO usage. This review describes the physical, biological and pharmacokinetic properties of endogenous EPO and its recombinant form. It also discusses the available strategies for the detection of rHuEPO abuse in sports, involving the use of sTfR concentration directly or in mathematical multivariate models.
Introduction
Extensive research has shown that soluble transferrin receptor (sTfR) is of great value in diagnosing iron deficiency (ID).1,2 Serum sTfR is also a diagnostic tool for determining erythropoietic activity. The increased concentration of sTfR in patients with ID reflects the hyperplasia of erythroid precursors and is a reliable indicator of iron deficiency anaemia (IDA) and conditions with enhanced erythropoietic activity. 3 Disorders such as autoimmune haemolytic anaemia, sickle cell anaemia, hereditary spherocytosis, β-thalassemia, and polycythemia vera have been reported to increase the number of circulating receptors, 4 whereas idiopathic haemochromatosis has been associated with decreased values. 5 In contrast with ferritin levels, sTfR levels are not affected by inflammatory reactions or other diseases and can therefore be used for diagnosing ID even under such conditions. 6
The monitoring of erythropoiesis and body iron status in several clinical conditions is currently the leading application of the measurement of sTfR.7,8 Recently, the measurement of sTfR has been proposed as a reliable tool in the prediction of the haematological response in individuals subjected to therapy with recombinant human erythropoietin (rHuEPO).7,8 Soluble transferrin receptor has gained considerable importance in drug testing. International Sporting Federations perform random blood tests before major competitions for the indirect detection of rHuEPO use. The purpose of this review is to summarize current scientific knowledge of the use of sTfR in detecting erythropoietin (EPO) abuse in sports. This article also examines: (i) the structure of the transferrin receptor, (ii) the chemical and pharmacological properties of endogenous EPO and its recombinant forms, (iii) the effect of physical exercise and doses of rHuEPO on sTfR levels, and (iv) the usefulness of blood testing multivariate models consisting of sTfR in the detection of rHuEPO.
Soluble Transferrin Receptor (sTfR)
Iron plays a vital role in the maturation, growth, and division of cells. The extracellular transport and dynamic storage of iron within the body are accomplished by its binding to a specific carrier protein, transferrin. 9 Transferrin has a high affinity for iron at neutral pH and iron requires a specific membrane receptor. This is the transferrin receptor (TfR) which binds diferric transferrin, and the receptor-transferrin complex is internalized into an endosome. Iron is released into the cytosol when the pH of the internal vesicles is reduced to approximately 5.5. After recycling to the cell surface, the apotransferrin dissociates, and the receptor is free to repeat the process. 10
The TfR is encoded by a gene (TFRC) located on chromosome 3, and consists of two identical 760-amino acid glycoprotein subunits each of molecular mass 95 kDa. 11 The functional receptor is a disulfide-linked dimer of the two subunits. 12 Each subunit has a 61-amino acid N-terminal cytoplasmic domain, a transmembrane region and a large extracellular domain. The TfR is shed from cells by proteolytic cleavage at Arg100- Leu101, 13,14 just external to the plasma membrane and just after the two inter-chain disulfide bonds. The product circulates in the blood as soluble TfR (sTfR), a 74 kDa monomer bound to transferrin. Therefore, serum sTfR is a truncated form of the intact receptor lacking the cytoplasmic and transmembrane domains. 13 The level of circulating sTfR is proportional to the total amount of cell-associated TfR. 15
Kohgo et al 16 in 1986 reported the detection of transferrin receptors in plasma by immunoassay and since then numerous articles have been published on the physiological and diagnostic significance of circulating sTfR. 12 Plasma concentration reflects the number of cellular receptors and in patients with adequate iron stores, the number of nucleated red cells in the bone marrow. Because TfR expression is upregulated when a cell needs more iron and because sTfR is proportional to total TfR, concentrations of sTfR are increased in plasma or serum of an iron-deficient subject.
Erythropoietin
Increasing oxygen delivery to tissues is important to optimize muscular activity and improve athletic performance, particularly in endurance. Several methods have been shown to increase oxygen delivery to tissues including altitude and hypoxic rooms, blood transfusion, and treatment with EPO. Erythropoietin represents for some athletes the ultimate tool to gain an edge over their peers in competition. It is a 165 amino acid (304 kDa) glycoprotein hormone produced mainly in the kidney, in the liver (< 10%) and, in very minute quantities in the brain. 17 Erythropoietin circulates in a pool of isoforms that differ in glycosylation and biological activity. It is the main regulator of erythropoiesis and the physiological stimulus for EPO production is tissue hypoxia, which, in the most instances, is directly related to the number of circulating erythrocytes. Thus, EPO and erythropoiesis are part of a negative feedback cycle that keeps tissue oxygen delivery within a narrow range by controlling the number of erythrocytes circulating in the blood. 18
In a normal individual, any loss of erythrocytes due to bleeding or haemolysis, decreases the delivery of oxygen to the tissues. When this tissue hypoxia is sensed by cells in the kidney and liver capable of producing EPO, they produce and secrete EPO into the plasma. Erythropoietin is carried to the bone marrow, where it binds to specific cell surface receptors on its target cells–-the colony forming unit-erythroid (CFU-E), pro-erythroblasts, and basophilic erythro-blasts. 19 The binding of EPO by these cells increases their ability to survive and reach the reticulocyte stage and thereby contribute to the population of circulating erythrocytes. The increased number of circulating erythrocytes in turn delivers more oxygen to the tissues. This increased oxygen delivery is sensed by the EPO producing cells, which then reduce EPO production so that the normal steady state number of erythrocytes is restored.
Depending on the sports modality, EPO administration to elite athletes may lead to an artificial performance improvement, decreasing, for instance, their time marks. So, in order to maintain health of the individual and sports ethics, the International Olympic Committee (IOC) and other International Sporting Federations considered the non-therapeutic use of EPO and its analogue drugs as blood doping cases. The use of EPO was banned in sports by the IOC in 1987.20
Recombinant Human EPO (rHuEPO)
Availability of this class of compounds in the market dramatically increased in the 1980s, with the advent of recombinant deoxyribonucleic acid (DNA) technology. The synthetic form of EPO, recombinant human EPO (rHuEPO), was genetically engineered for therapeutic use in 1987 and marketing began in 1988. Since then, it has been successfully used in medical care, improving quality of life of patients in constant need of blood transfusions, or with anaemia due to chronic renal failure. Several types of rHuEPO are commercially available including: Epoetin alpha (Eprex, Janssen-Cilag), Epoetin beta (Neorecormon, Roche), and Darbepoetin alpha (Nespo, Dompè). 21 The recombinant forms of EPO have indiscriminately been used by athletes, mainly in endurance sports, to increase the erythrocyte concentration, thereby generating a better delivery of oxygen to the muscle tissue. It may have been used as early as 1988 in the Calgary Winter Olympics Skiing events.
Recombinant EPO is a glycoprotein of 304 kDa, has a short half-life, although its effects in the blood only become evident 3–5 days after administration. These effects subsist for one to several weeks in cases of prolonged treatment. If treatment is stopped a few days before competition, the athlete thus benefits from its effects without risk of it being detected. After intravenous administration, maximum serum rHuEPO concentration peaks are reached within minutes, whereas time to reach maximum peak values from subcutaneous administration may range from 5 to 24 hours after administration, or a little more. 22 Clinical trials carried out in mid 1990s to assess intravenous and subcutaneous route of administration did not show differences in the effectiveness of treatment or in blood pressure when the two routes were compared. 23
Immediately after rHuEPO became available as an erythropoiesis-stimulating drug it was imputed to be abused by athletes in aerobic sports. Recombinant human EPO doping by healthy athletes who do not need any treatment is not acceptable. Fatal cases connected to doping occurred in early 1990s, when professional Dutch cyclists competing in Europe died at rest, some of them while sleeping, due to unexplainable cardiac arrest. 24 These sportsmen are among the more than 20 cases of death of cyclists in Europe reported between 1987 and 1991, when rHuEPO was first made available in that continent. 25 Evidences on the use of rHuEPO were found after a blood check of Italian professional cyclists that showed serum iron overload comparable to those of genetic haemochromatose patients. The levels of ferritin in these cyclists were abnormally high, a clear sign of intravenous iron and rHuEPO administration. 24 In 1989, the IOC Medical Commission introduced the new doping class of peptide hormones and analogues, which includes rHuEPO, human chorionic gonadotrophin and related compounds, adrenocorticotrophic hormone, human growth hormone, and all the releasing factors of these hormones which are required for their synthesis. 26
In 1998, discovering rHuEPO ampoules with cyclists of a number of teams participating in the “Tour de France” was probably the most striking and publicized rHuEPO doping in recent sports events. At that time, boxes with many ampoules of the drug were found in trucks of the main participating teams, and even in the competitors’ lodgings. The discovery reflected endemic use of this hormone among elite athletes. Over the 21 days of the competition, drug busts, inquiries and arrests became part of the competitions. French newspapers called the debacle “Tour de Farce.” Some athletes, ailing from the illicit use of rHuEPO, admitted using the hormone. 27 A cyclist with severe headache, nausea, vomiting, and photophobia two months before a competition, led to a diagnosis of idiopathic intracranial hypertension, non-responsive to standard treatment. Medical examination and laboratory tests suggested the use of rHuEPO, confirmed by the athlete upon questioning. 28 In spite of all the cases above, liberation of the drug has been advocated by some groups, arguing that because rHuEPO is impossible to be detected, it is best to allow its use. The IOC, however, included this recombinant hormone in the list of forbidden drugs from 1990, after the Calgary Winter Games and the Seoul Olympic Games, where its use was evidenced. Prohibition was on a moral ground, due to lack of technology to fully detect and differentiate rHuEPO from endogenous EPO.
The main risks of erythrocytosis include heart failure, myocardial infarction, seizures, peripheral thromboembolic events and pulmonary embolism. The risks are elevated during competition when the blood viscosity increases further due to intensified perspiration and the shift of fluid from the intravascular into the interstitial space. 29 The fact that the above mentioned cases of death of cyclists suspected of rHuEPO doping did not occur during exercise but during periods of physical inactivity does not militate against the detrimental effect of erythrocytosis. Blood flow in the microcirculation will slow down during physical inactivity thereby favoring the development of thrombi. The risk of EPO to promote tumour growth has been considered, 30 although supporting clinical evidence is missing.
Soluble Transferrin Receptor and Exercise
Strenuous exercise modifies haematological parameters. Red cell count, haemocrit and haemoglobin levels change in endurance runners during marathon, while mean cell volume and mean cell haemoglobin were negligibly affected. 31 It has been suggested that the high levels of EPO may induce an increase in red cell mass after a marathon run although the regulation of EPO synthesis in athletes is far from being under-stood. 32 Erythropoietin levels rise also in well-trained individuals even after short high-intensity exercise that induces hypoxaemia. 33 Furthermore an increase in reticulocytes with high RNA content supports early erythropoietic changes. 34 These erythropoietic changes may be due to several mechanisms such as intravascular haemolysis due to mechanical trauma, high body temperature, dehydration, haemoconcentration and oxidative stress. 35
It is known that red blood cell mass and therefore erythropoietic activity is increased in endurance trained athletes. 36 Numerous reports have described a poor iron status in female endurance athletes. However, the traditionally applied indicators of iron status (haemoglobin, ferritin, transferrin) may not truly reflect iron status. Poor iron status was found in female endurance athletes before a triathlon and sTfR increased slightly after the race. 37 However, when the values were corrected for haemoconcentration no change was found. As sTfR reflects the overall erythropoietic activity, this variable should be higher in athletes than in untrained or moderately trained subjects. In recent years many investigators have observed a decrease in haemoglobin concentration, as well as high prevalence of iron deficiency in elite athletes, particularly among endurance runners and female athletes. The increase in iron requirements due to intensive prolonged exercise, the increase in iron loss particularly by sweating, and an often inadequate iron supply from dietary intake especially in females, explain the appearance of an iron deficiency that, when it is not corrected, leads to anaemia. 38
Only a few studies on the impact of physical exercise on sTfR have been conducted. Despite the importance of sTfR in the diagnosis of iron deficiency, to our knowledge, only a few studies have tested the hypothesis that sTfR levels remain stable after acute exercise 39 or after a brief training period. 40 Malczewska et al 40 assessed the effect of physical exercise on the changes in the concentration of ferritin and sTfR in plasma of ten female elite judoists in course of 10 consecutive days of training camp exercise. They found that physical exercise had no effect on serum sTfR concentration and is a more stable indicator of iron status under high intensity training since, unlike ferritin, it does not respond to workload on the preceding day. Moreover, the intravascular haemolysis observed in athletes does not affect the sTfR levels in plasma. 40 In a similar study, Schumacher et al 39 found no significant changes in ferritin or sTfR of untrained individuals after a treadmill running protocol (70% maximum oxygen consumption rate) for 45 minutes. However, ferritin (but not sTfR) increased slightly (by 6%) in trained individuals subjected to the same protocol. 39 Furthermore, in a study with similar exercise protocol, serum ferritin and sTfR levels remain stable up to 24 hours after acute prolonged exercise of moderate intensity. This renders both parameters reliable indices of iron status for amateur endurance athletes. 41 Weight training increased strength and fat-free mass and decreased levels of percent body fat. Haemoglobin concentration declined after 12 weeks of training in young male and female college students performing weight-training exercise. No significant changes were observed for haematocrit, total iron binding capacity (TIBC), transferrin saturation (TS) and sTfR. 42
There are a few studies which have showed that sTfR increased after exercise. Rahmani-Nia et al reported that iron levels decreased significantly in experienced female taekwondoist after incremental treadmill running and 30 minutes running test at constant speed at 50% of HR max. 43 Soluble transferrin receptor increased after both exercise protocols however increases were significant after incremental treadmill running only. 43 The haematological parameters and possible modifications in elite rugby players throughout a competitive season were assessed by Banfi et al. 44 Serum sTfR increased after training and during the competition period while haemoglobin and haemocrit levels decreased. The authors concluded that reticulocytes and sTfR are the most sensitive parameters for studying iron metabolism of the athletes. In the investigation of the prevalence of iron deficiency and anaemia in adolescents undergoing strenuous physical training, Huerta et al found that 19% of the study subjects had mild anemia at recruitment and depletion of iron stores was observed among 18%. These changes were not accompanied by a significant increase in sTfR. 45
Recombinant EPO and Soluble Transferrin Receptor
A number of studies have investigated indirect screening methods to detect use of Epoetin or Darbepoetin by athletes (rHuEPO abuse) by means of parameters indicative of accelerated erythropoiesis. Parameters or markers of altered erythropoiesis proved to be closely related to rHuEPO administration include haematocrit (Hct), percentage reticulocytes, reticulocyte haematocrit (RetHct), and haemoglobin content (CHr), percentage hypochromic red cells, percentage of macrocytes, sTfR and serum EPO.46,47
In 1997, the International Union of Cyclism (IUC) implemented random blood tests before each competition. The test involved measuring the level of haematocrit, the proportion of blood volume that is occupied by red blood cells. The haematocrit is a physiological variable closely related to performance enhancement on exercise. The IUC established that male and female athletes with haematocrit more than 50% and 47% respectively could not compete, on grounds that this would be a “hazardous competition” for them. 48 There were controversies on these limits, as dehydration status, the hour of the day sample is taken, and the body position of the athlete are limiting factors, in addition to the existence of a small proportion of elite athletes who present a haematocrit higher than the established limits without using rHuEPO.24,49 Subcutaneous administration of rHuEPO has been found to increase haematocrit, time of exhaustion from exercise, maximum oxygen intake, and systolic blood pressure. 48 The side effects described for rHuEPO are more marked when excessive hormone doses raise haematocrit to values between 50 and 55%, which occurs with endurance sports, due to natural dehydration. 50 Subsequently, the IUC adopted the maximum level of serum haemoglobin of 17 g/dL as an additional criterion for allowing athletes to compete. 51 The International Ski Federation also implemented banning procedures for athletes, and adopted the limit of 18.5 g/dL for serum haemoglobin. 24
Due to limitations in sample collection and problems related to the reliability in associating haematocrit values, other serum components were assessed as possible indirect markers of EPO activity. 52 Reticulocytosis and macrocytosis are observed in the peripheral blood, reflecting an enhanced marrow activity, both after secretion of endogenous EPO and rHuEPO administration. Reticulocytes and macrocytes have higher potential as parameters to indicate substance abuse, as their response in terms of serum concentration is quite proportional to the amount of serum rHuEPO. 53 However, testing the quantity of reticulocytes did not yield satisfactory results due to the vast discrepancy between the reference values of their concentrations. 54
The level of sTfR can be elevated in cases of iron deficiency, increased demand for iron during stimulated erythopoiesis and administration of rHuEPO. 55 The results of a trial involving healthy subjects demonstrated that regular rHuEPO injections significantly increased the sTfR concentration. 56 Furthermore, administration of high rHuEPO doses results in increased sTfR concentration, at most, 5 times above basal level. The mRNA that presents for this receptor increases to about 40 times above the basal value which suggests that in the near future erythroid genetic markers may become available to detect rHuEPO abusive use in sports.57,58 However, although sTfR was also regarded as a potential rHuEPO marker, research showed that it can act as a drug detector only in the case of administration of large doses of rHuEPO without iron supplements. 54
Another indirect test, developed in 1996 for the detection of rHuEPO abuse, was based on the determination of the sTfR/ferritin ratio. 59 Ferritin was used as a denominator mainly to prevent variations in hydration level. This ratio, however, cannot be used as a marker, alone as additional iron supplementation tends to balance this ratio by increasing ferritin levels.60,61 Iron supplementation, a common practice among athletes (especially intravenous iron injections), 62 will result in the modification of the sTfR/ferritin ratio. The sTfR/ferritin ratio is masked by iron supplementation and is also affected by possible exercise induced haemoconcentration, the sTfR/total protein. 63 An increase in concentration of hypochromic erythrocytes is seen, and in this case they are the best markers when diagnosed immediately after rHuEPO administration. 49 Serum EPO is also measured, but because the EPO levels fluctuate over time and among individuals, measuring EPO concentration alone may not be useful to detect doping. Thus a the lack of sensitivity of some of the secondary blood markers as well as the lack of specificity of others encouraged some scientists to put them together in a multiple marker mathematical model to discriminate rHuEPO abusers from healthy sports people. The reference values for these parameters were found to vary by gender, ethnicity, and altitude. 64
The Effect of Doses of rHuEPO on sTfR
Beside clinical applications, the measurement of sTfR, either alone or combined with other biochemical markers, was proposed as the best predictor of response to treatment with rHuEPO. 65 In agreement with clinical studies, sTfR has been recently proved as the most reliable marker for doping control, as its measurement indicated the consumption of rHuEPO with the highest probability. 66 In a study by Yang et al six male volunteers (age 19 to 23) were subcutaneously administered rHuEPO at a dosage of 30 IU/kg body weight, three times a week for 4 weeks. 67 The serum concentrations of sTfR and EPO were found significantly increased following the multiple injections of rHuEPO. An increased haematocrit with concomitant changes in sTfR and sTfR/ferritin values seems to supply a valuable diagnostic tool that avoids false-positive doping and allows the identification of Epoetin abusers. 67 In another study, Audran et al investigated the effects of repeated subcutaneous injection of rHuEPO (50 IU/kg) in athletes and found that significant changes in reticulocytes, haemoglobin concentration, haematocrit, sTfR, and sTfR/serum proteins were observed during and after rHuEPO treatment (Table 1). 68 The increased values of sTfR and sTfR/serum proteins, respectively, above 10 μg/mL and 153, indicated the probable intake of rHuEPO. 68
Effect of recombinant human erythropoietin (rHuEPO) on soluble transferrin receptor (sTfR).
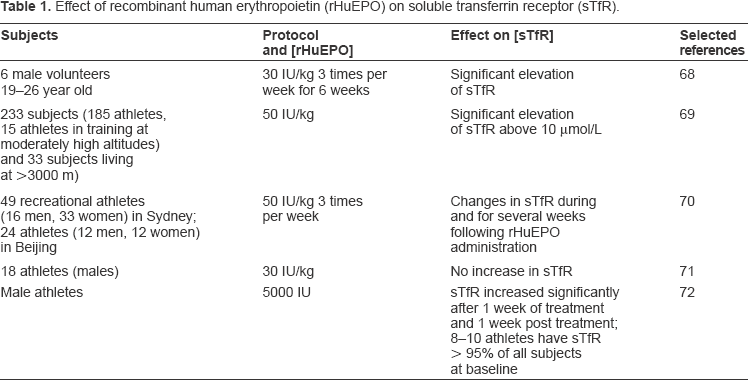
Recreational athletes resident in Sydney, Australia or Beijing, China were randomly assigned to rHuEPO or placebo groups prior to a 25 day administration phase. Injections of rHuEPO (or saline) were administered double-blind at a dose of 50 IU/kg three times per week, with oral iron (105 mg) or placebo supplements taken daily by all subjects (Table 1). 69 These markers were disturbed both during and for several weeks following rHuEPO administration. Ethnicity did not influence the markers identified as being able to detect athletes who abuse rHuEPO. This work establishes an indirect blood test which offers a useful means of detecting and deterring rHuEPO abuse. 69 However, a significant decrease in circulating ferritin and an increase in sTfR were not found in athletes receiving low (30 IU/kg) doses of rHuEPO. 70 Thus, an increase in the sTfR/ferritin ratio cannot be used as an indicator of rHuEPO abuse, at least when the hormone is administered at low concentrations (Table 1). 70
Birkeland et al assessed the possibility of using serum levels of sTfR as a marker of doping with rHuEPO by performing a double blind, placebo-controlled study with the administration of 5000 IU of rHuEPO or placebo three times weekly for four weeks to 20 healthy male athletes (Table 1). 71 The authors found that serum levels of sTfR may be used as an indirect marker of supra-normal erythropoiesis up to 1 wk after the administration of rHuEPO. The effects on endurance performance outlast the increase in sTfR. The data shows that under real conditions (iron supplementation and low doses of EPO) the sTfR/ferritin relation is not a useful indication of EPO doping. 71
Nissen et al demonstrated whether changes in the charge pattern of urinary human erythropoietin (u-HuEPO) from well-trained athletes before, during and after controlled administration of rHuEPO could be related to altered levels of haemoglobin, haematocrit, sTfR and maximal oxygen uptake. 72 The charge of the u-HuEPO variants shifted from an acidic to a more basic pattern after initiating rHuEPO administration. This shift appeared together with increased levels of sTfR, and appeared before increased levels of haemoglobin, haematocrit, and maximum oxygen uptake O2. Until three days after the last injection, the isoelectric focusing (IEF) profiles were similar to the charge profile of rHuEPO. Thereafter the levels of sTfR decreased and the charge profiles of the HuEPO variants gradually became more acidic. 72 In contrast, the levels of haemoglobin and haematocrit remained elevated for an extended period of time. A significant correlation was found between the relative amount of basic u-HuEPO variants and the relative levels of sTfR, demonstrating that the relative levels of sTfR may be used as a marker to select urinary samples for further analysis of rHuEPO by IEF in routine doping control. 72
Models that are Indirect Markers of rHuEPO
Potential abuse of rHuEPO can best be detected by measurement of haematopoietic parameters. Following a double blind study with regular rHuEPO injections, the Australian Institute of Sport, together with the Australian Sports Drug Testing Laboratory, designed an anti-doping test using five secondary blood markers such as the haematocrit level, the reticulocyte haematocrit, serum sTfR and EPO concentrations, and the percentage macrocytic cells. Two models were developed based on the behaviour of each of the five parameters during and after controlled treatments with rHuEPO and placebo.73,74 The ON-model, indicative of accelerated erythropoiesis, was developed to discriminate between current rHuEPO users and placebo subjects using the component variables: haematocrit, reticulocyte haematocrit, percent macrocytes, serum EPO concentration, and serum sTfR. It was applied during or shortly after rHuEPO treatment.75,76 The OFF-model, which relies on indicators of depressed erythropoiesis (abnormally low reticulocyte haematocrit and EPO) juxtaposed with high haematocrit, was developed to identify subjects who had recently discontinued rHuEPO administration and was used weeks after termination of treatment.75,76 In addition, because of the marked acceleration in erythropoiesis during the early stages of rHuEPO use, and the decrease in erythropoiesis after cessation of use, there is a resultant variation in ON- and OFF-model scores as a function of time. The models 73 are as follows:
For the “ON” model: ON score = haemoglobin + 9.74 ln (EPO);
OR haemoglobin + 6.62 ln (EPO) + 19.4 ln (sTfR).
For the “OFF” model: OFF score = haemoglobin –60 (reticulocyte percentage)1/2;
OR haemoglobin – 50 (reticulocyte percentage) 1/2 –7 ln (EPO)
In August 2000, the IOC Medical Commission approved the ON-model to be used during the 2000 Olympic Games in Sydney. As the direct method capable of discriminating endogenous EPO from rHuEPO had already been published in Spring 2000, 77 the ON-model was used only as a screening test to determine which urine samples had to be collected to perform the urinary test. In addition, if these parameters were unusual, isoelectric focusing (IEF) of urine samples was then employed to provide proof of rHuEPO abuse.78,79 The combination of the blood parameters with urine IEF was approved by the IOC in 2001. 78 This test resulted in the forfeiture of medals won by three cross-country skiers in the Salt Lake City Winter Olympics in 2002. 80 In June 2003, the Executive Committee of the World Anti-Doping Agency (WADA) approved the use of only urine tests for detecting the presence of rHuEPO in doping control. On the other hand, indirect EPO tests offer the advantage of detecting other related kinds of blood doping. The direct EPO test only detects the use of Epoetin-alfa, Epoetin-beta, and Darbepoetin. However, Dynepo/Erythropoietin-delta, which is produced in cultured human cells 81 has no distinct glycosylation pattern that enables its detection by the direct EPO test.
Use of Models to Detect rHuEPO
The models incorporating combinations of the variables reticulocyte haematocrit, serum EPO, sTfR, Hct and % macrocytes were analyzed by logistic regression. 73 One model (ON-model) repeatedly identified 94%–100% of rHuEPO-treated athletes during the last 2 weeks of the rHuEPO administration phase. One false positive was recorded from a possible 189 cases. The other model (OFF-model) incorporating reticulocyte haematocrit, serum EPO, and haematocrit, was applied during the wash-out phase and, during the period of 12–21 days after the last rHuEPO injection, it repeatedly identified 67%–72% of recent users, with no false positives. 73 The study demonstrated that multiple indirect markers used simultaneously were potentially effective for identifying current or recent users of rHuEPO. 73 In another study in 2000 by Parisotto et al it was confirmed that rHuEPO administration causes a predictable and reproducible haematological response both during and for several weeks following administration. 69
In a latter study Sharpe et al undertook a large-scale, international blood profiling study of elite athletes with the aim of developing 95% reference ranges. The authors also quantified the effects of gender, ethnicity, exercise, sporting discipline, altitude as well as biological and analyzer variation on the haematologic variables and detection models. 64 The models used in this study 69 were:
ON-model score = 3.721 Hct + 30.45 RetHct + 0.1871 loge(EPO) + 0.1269 loge(sTfR) + 0.1115 loge (% macrocytes + 0.1)
OFF-model score = 6.149 Hct - 92.87 RetHct –0.1463 loge (EPO)
The study 64 identified a small number of athletes with ON-model scores that were considerably in excess of the expected range. For OFF-model scores which were the maximum upper limit of the 95% reference ranges were calculated to be 2.24 for males and 2.08 for females. Males injected with rHuEPO recorded OFF-model scores in excess of 2.50, whilst 14 of the 15 females recorded scores in excess of 2.10. Furthermore, none of the haematologic abnormalities led to elevated OFF-model scores. 64 The researchers concluded that changes in model scores (together with monitoring of urine samples for the presence of rHuEPO) provide a promising approach to detection of rHuEPO abuse, because they are less sensitive to subject characteristics and less variable than the raw model scores. In addition they suggested that these models may, by themselves, be extremely useful tools for identifying athletes who have recently ceased using rHuEPO. However the stability of the model scores (particularly ON-model) for an individual over time has sufficient potential for highlighting rHuEPO use to warrant further research. Specifically, when applied in unison with the IEF technique for detecting rHuEPO in urine, this could leverage the efficacy of out-of-competition testing for rHuEPO where both blood and urine samples are collected. 64
Some of the limitations of these models are associated with the dependency of some of the parameters on cellular volume, potentially affected by blood storage and transportation problems. Consequently a second generation of models with higher robustness that were simpler to apply were proposed by Gore and colleagues. 82 They were labeled with the letters h, e, r, and s according to the parameters [Hb], [EPO],% ret and [sTfR], respectively used in each case.
The new equations 82 are as follows:
ON he = [Hb] + 9.74 [EPO], ON hes = [Hb] + 6.62 ln[EPO] + 19.4 ln[sTfR]
Gore et al found that the ability of the new models to detect rHuEPO administration was generally higher than previous models, particularly during phases when low doses of rHuEPO were used and after injection had ceased. 82
The indirect methods reported above can be further improved and researchers are currently conducting several studies worldwide. Clearly, the first objective is to abolish false positives as much as possible. The refined approach, however, should be part of global strategy for preventing and detecting blood doping. Such a strategy must include definition of the individual haematologic profile, the so-called haematologic passport. 83 The haematologic passport CONI and FIGC (Italian Soccer Federation) have recently launched a campaign called Io non rischio la salute! [I take care of my health!]. This involves regular haematologic investigations (2–3 times a year) including blood counts, reticulocyte count, serum ferritin and sTfR measurements. 24 These determinations must be performed by laboratories participating in quality control programmes to keep analytical errors as low as possible. Sequential evaluation of the above parameters allows definition of the individual haematological profile. Comparing an athlete's individual haematologic values against his or her own historical baseline rather than a population derived threshold value enhances the potency of indirect testing. An athlete's true baseline value can be estimated with just one prior blood test. Eliminating intra-individual variability by comparing new results against an historical baseline increased the ability to detect blood doping. 84 In addition longitudinal monitoring of athletes’ blood profiles can help detect blood doping. 84
Indirect EPO detection methods require blood sampling, which is a practical disadvantage in doping control. To obtain optimal test efficiency, attention has to be paid to the pre-analytical phase.85,86 Data on the effect of altitude training on blood model scores to detect EPO abuse are inconclusive and needs further investigation. 87
Conclusion
Blood doping is an illicit practice aimed at increasing oxygen tissue delivery and aerobic performance, mainly but not exclusively in endurance sports. The availability of rHuEPO has increased the risk of its illegal use in sports, its detection being a difficult challenge. Soluble transferrin receptor has been included in multivariable blood testing models (ON and OFF) for the detection of performance enhancing EPO abuse in sport. In recent years new models (ON-he and ON-hes) to detect rHuEPO administration were generally higher than previous models, particularly during phases when low doses of rHuEPO were used and after injection had ceased. However, procedures should be incorporated for elevated model score caused by genetic health and environmental circumstances. Indirect EPO detection methods require blood sampling, which is a practical disadvantage in doping control. These models are associated with the dependency of some of the parameters on cellular volume, potentially affected by blood storage and transportation problems. The limitation of sTfR is the absence of an internationally recognized reference standard of measurement and comparable units, which makes comparison of results from different assay kits difficult. However, the imminent introduction of fully automated assays and the calibration against natural plasma represents substantial steps toward harmonization of procedures and units of measurement. Direct methods attempt to identify slight differences between recombinant EPO, which is produced through genetic engineering, and human EPO. A more direct method of urine analysis (IEF, double blotting, and chemiluminescent detection) based on the charge differences between rHuEPO and endogenous EPO, related to their carbohydrate composition, provides proof of rHuEPO use. Furthermore, this approach permits the detection of darbepoetin, a direct analogue of EPO also known as NESP (new erythropoiesis stimulating protein).
The use of extensive out-of-competition testing and analysis within accredited laboratory systems is critical to any testing programme. An alternative approach involves sequential evaluation of haematologic parameters to determine an individual's haematologic performance, i.e. athlete's haematologic profile. Combining haematologic passport and use of models might be an effective strategy to detect current blood doping and deter future EPO abuse.
Disclosures
The authors report no conflicts of interest.
