Abstract
Purpose
To evaluate cardiac doses in breast cancer patients with stage II/III treated with 4-field radiotherapy based on computed tomography (CT) dose planning.
Methods and Materials
Based on archived CT images, whole heart and cardiac chamber radiation doses were analyzed in 216 (111 left-sided and 105 right-sided) mastectomized or lumpectomized breast cancer patients treated at a single institution, the Norwegian Radium Hospital, between 2000–2002. Individual dose volume histograms for the whole heart and for the four cardiac chambers were obtained, and mean, median and maximum doses to these structures were calculated. The dose (Gy) delivered to the 5% of the volume of each cardiac structure (D5%), and the volume percentage of each structure receiving ≥ 25 Gy (V25Gy) were reported. Normal tissue complication probability (NTCP) calculations were used to estimate the risk for ischemic heart disease (IHD).
Results
Cohort-based medians of the whole heart mean dose (Dmean) for left- and right-sided tumors were 3.2 Gy and 1.3 Gy, respectively, with similar ventricular but lower atrial values. The atrial doses did not differ according to laterality of the breast tumor. In 13 patients with left-sided cancer, 5% of the heart volume was exposed to >25 Gy. The NTCP estimates were generelly low, with a maximum of 2.8%.
Conclusions
During adjuvant CT-based locoregional radiotherapy of women with breast cancer, the cardiac radiation doses are, at the group level, below recommended threshold values (D5% < 25 Gy), though individual patients with left-sided disease may exceed these limits.
Introduction
A number of large randomized trials have shown a small but statistically significant risk of cardiac mortality associated with locoregional irradiation of breast cancer.1–3 In the Early Breast Cancer Trialists Collaborative Group meta-analysis, radiotherapy (RT) compared with no RT was associated with excess mortality (rate ratio 1.27) from heart disease. 4 In many of the included trials, older techniques were applied such as Cobalt-60 (Co-60) or orthovoltage radiation, and treatment was based on manual planning. Other studies have shown that modern techniques and dose planning recommendations result in much smaller cardiac radiation doses.5–7 However, even with modern techniques, a subgroup of patients with left-sided breast cancer might receive cardiotoxic doses.6,8–10 Further, considerable variation in the cardiac doses according to the use of different RT treatment regimens has been pointed out. 11 On the other hand, Correa et al 12 have concluded that cardiac toxicity could be avoided by use of proper field arrangements and choice of beam energy. The application of CT-based dose planning, breath-hold techniques and intensity-modulated RT thus represents an important step to reduce long-term cardiac risk in breast cancer survivors.
Dose-response relationships based on cardiac mortality data have been produced using a radiobiological model. 8 Model-based estimates predict that V25 < 10% (V25: heart volume receieving 25 Gy or more) is associated with a < 1% probability of cardiac mortality 15 years after radiotherapy 13 The Norwegian Breast Cancer Group (NBCG) recommends that V25 < 5% (http://www.nbcg.no), and The National Danish Breast Cancer Group states that no part of the left anterior descending artery (LAD) should be irradiated with 20 Gy or more, with V40 < 5% and V20 < 10% for the heart (http://www.dbcg.dk).
To our knowledge, no detailed dosimetric data from a large routinely treated patient cohort are available in which the radiation exposure to the cardiac chambers using modern radiotherapy techniques for a large cohort of real-life patients has been documented. Furthermore, most commonly radiation-induced cardiotoxicity is described as abnormal function of the left ventricle, as demonstrated by echocardiography 14 In recent years, reduced diastolic function 14 has gained increasing attention as a parameter of early cardiotoxicity, and assessment of atrial radiation doses has become a goal of future research. Such data should be analyzed together with long-term atrial and ventricular morbidity in breast cancer survivors.
We plan to investigate the relationship between cardiac doses applied during adjuvant irradiation of breast cancer stage II/III and cardiotoxicity in 10-year breast cancer survivors. As a first step, we report here total ventricular as well as atrial cardiac doses for future estimation of radiation-induced complication probabilities among the patients in question. The aim of the current descriptive study was to measure radiation doses to all 4 cardiac chambers based on archived CT images established when planning adjuvant radiotherapy in breast cancer patients.
Material and Methods
Patients
Patient demographics and treatment related characteristics for 216 included breast cancer patients.

Adjuvant radiotherapy to the thoracic wall and lymph nodes regions+ boost to the lumpectomy area.
The study was approved by the ethical committee of the South Health Region South in Norway.
4-Field Radiation Technique
All women were treated with 4-field RT where the target volume included the breast (after lumpectomy) or the chest wall (after mastectomy), the ipsilateral supraclavicular and infraclavicular fossa, the ipsilateral internal mammary nodes (IMNs), and the ipsilateral axillary nodes. Respiratory gating was not used. The RT planning was based on noncontrast enhanced transverse CT scans covering the region from the 6th cervical vertebra to the middle part of the abdomen, the CT-slice thickness being 1 cm. Wing board with headrest was employed. Treatment planning and dose calculation were performed using the Helax-TMS (Version 6.0 or higher) system applying a pencil beam algorithm. The voxel size of the dose calculation matrix was 0.5 × 0.5 × 1.0 cm3.
The beam arrangement consisted of four half-beams with two tangential beams covering the caudal part of the target volume, one anterior-posterior field (0°) and one oblique field, 110° to 115°, covering the cranial part of the target volume (Figs. 1 and 2). Field 1 and 2 were intended to treat the ipsilateral supraclavicular and infraclavicular, axillary, and internal mammary lymph nodes (IMNs) and the cranial part of the breast/breast wall, while tangential fields 3 and 4 treated the caudal part of the breast/breast wall. The beam angles, apertures, weights, and dynamic wedges were optimized by standard (forward) planning. In the longitudinal direction, the isocentre was placed immediately caudal of the IMNs, which typically was 2 to 3 cm caudal of the carina. The photon beam energy was mainly 6 MV using a Siemens or Varian linear accelerator. The dose plans were normalized to the mean dose to the CTV. The breast/chest wall received a total dose of 50 Gy, and the regional lymph nodes, 46 to 50 Gy. Thirty-six lumpectomized women, all younger than 50 years of age, received a boost of 10 or 16 Gy to the tumor bed (9–12 MeV) using a circular field with a diameter of 5 to 9 cm. Mastectomized women with stage III breast cancer and patients with stage II breast cancer with tumor-positive resection margins had bolus covering the whole breast wall; otherwise, a bolus of 6 cm width covering only the mastecomy scar was applied. The impact of boost on cardiac radiation doses was not investigated in this study.
The 4-field arrangement used in CT-RT shown schematically. Radiation fields covering the caudal part of the breast/breast wall (fields 3 and 4) are shown here.
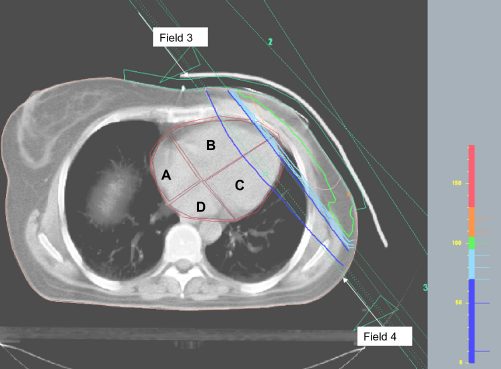
Cardiac Volume Definition
During the routine dose planning procedure, a clinical oncologist had delineated the heart on the CT images, but for the purpose of this study, cardiac delineation was redone by a radiologist using the archived CT images. With applied CT scan technique, the coronary arteries were not consistently visible; hence, coronary arteries were not contoured. The cranial limit of the heart included the right atrium and excluded the pulmonary trunk and coronary arteries. The caudal contour of the heart was the caudal myocardial border. The pericardium was included in the heart volume; the inferior caval vein was excluded. The right and left heart atria and ventricles were contoured separately. The atrial septum was well discernible on the available scans as it had lower attenuation than the surrounding blood and myocardium. Cardiac motion and lack of contrast made it difficult to reliably identify the valves and the interventricular septum. To have reproducible results, we defined these borders from certain key structures that were reliably identified on the available scans:
The atrioventricular groove The interventricular groove/apex cordis An area of fibrous/fatty tissue where the atrial septum meets the atrioventricular plane, anatomically corresponding to the trigonum fibrosum dextrum The pulmonary artery bifurcation.
The border between atria and the ventricles were defined as straight lines drawn between A and C. The border between the ventricles was a straight line between B and C. The cranial border of the right ventricle was defined as the third CT-slice below D (Fig. 2).
Dose Calculations
For each patient, dose volume histograms (DVHs) for the whole heart and each of the four delineated cardiac chambers were obtained from the treatment planning module supplemented by the individual mean, median, and maximum doses. Further, the individual dose (Gy) delivered to 5% of the volume of each specific cardiac structure (D5%) was assessed as well as the volume percentage of the respective structure receiving 25 Gy or more (V25Gt).
Normal Tissue Complication Probability
The normal tissue complication probability (NTCP), in terms of excess cardiac mortality at 15 years, was estimated using the relative seriality model. 8 This model employs the DVH for the heart and accounts for the serial and parallell architecture of tissue subunits and for the binomial nature of cell kill. The dose at 50% response, D50, and the maximal relative slope, γ, were used to describe the dose-response curve. The parameters used were D50 = 52.3 Gy and γ = 1.28. 8 A fractionation sensitivity (α/β-ratio) of 3 Gy was assumed. Individual NTCP estimates were obtained for all patients based on their DVH.
Statistics
As a rule, cohort-based medians along with their ranges were established for the continous demographic and dosimetric parameters. Differences between variables in patients with right- and left-sided tumors were evaluated by the Mann–Whitney test. However, the dose-volume histograms display mean values and one standard deviation. A P value < 0.05 was considered to be statistically significant.
Results
Compared to women with right-sided tumors, those with left-sided tumors had significantly higher radiation doses to the whole heart and the ventricles (Fig. 3 and Table 2), whereas the doses to the atria did not differ according to laterality of the tumor. Significant differences also emerged for D5% for the whole heart and for the left and right ventricle but not for the atria. There was a considerable interpatient variability in cardiac doses. Greater variations of the cardiac parameters were observed for patients with left-than with right-sided tumors, with exception of the right atrium. Cohort-based medians of the whole cardiac mean dose (Dmean) were 3.2 Gy and 1.3 Gy for patients with left- and right-sided breast cancer, respectively. In none of the patients with right-sided tumors, but in 13 (12%) of those with left-sided cancer, 5% of the heart volume was exposed to >25 Gy.
Mean dose-volume histograms displaying whole heart, ventricular and atrial radiation doses, in 111 patients with right-sided (red) and 105 patients with left-sided (black) tumors. Whole heart and cardiac chamber radiation dose, together with the normal tisuue complication probabilities (NTCP) based on the dose volume histogram for the whole heart. P < 0.01 comparing left-sided with right-sided tumors; †13 patients, all left-sided tumors, received D5% > 25Gy with a range of [28.3–44.1Gy].

Figure 3 provides cohort-based cumulative DVHs for the whole heart and the 4 cardiac chambers. For whole heart and ventricular doses between 5 to 45 Gy, significant differences were observed comparing left-sided with right-sided tumors. No statistical differences for the dosimetric results were observed for the atria with exception of the 5 Gy point of the right atrium.
The estimated normal tissue complication probability for the whole heart was moderately higher for the patients with left-sided compared with those with right-sided disease (Table 2). Furthermore, the NTCP estimates are very low for the whole population included in this study, with a maximum probability of 2.8%.
Discussion
Cohort-based mean radiation doses to the ventricles did not differ significantly from the whole heart mean doses, but both parameters were related to the laterality of the breast cancer. No side-related differences emerged between the very low atrial doses. Further, the NTCP estimates were very low for the whole population, independent of laterality.
With the recognition of the beneficial effect of adjuvant radiotherapy of breast cancer, research has increasingly dealt with long-term radiation-related adverse effects such as cardiotoxicity. Many studies have shown that tumor laterality has the greatest impact on radiation doses to the heart8,9,12,13 with additional impact of the radiotherapy technique.12,16–19 Not surprisingly, there has been a gradual decrease of the cardiac doses along with technical improvements during the last two decades with the introduction of CT-based radiotherapy planning, IMRT, and respiratory gating.8,12,20 The possibility of establishing individual CT-based DVHs has also lead to official guidelines as to doses to the whole heart or its parts. In most of our patients, the NBCG's recommendation that not more than 5% of the heart's volume should receive 25 Gy has been met but with considerable interpatient variability. Lower cardiac doses may be achieved when the breast wall is irradiated by electron beams.8,21 However, breast wall irradiation with electron beams may not provide a uniform target dose distribution. Reduction in mean cardiac dose can also be achieved by breath holding techniques, such as deep-inspiratory breath-hold introduced during the last decade. 12 Taking into account the differences in dose estimation procedures and different patient numbers, our presented figures are comparable to those for modern radiotherapy techniques published by Taylor et al 8 regarding whole cardiac mean doses (0.9–14 Gy for left-sided disease and 0.4–5 Gy for rightsided disease).
Pierce et al 21 noted that the volume of heart receiving greater than 30 Gy (V30) was significantly higher with Co60 treatment than other linear accelerator-based methods. Based on a study of 28 patients, Fuller et al 22 recorded that when using orthovoltage radiotherapy approximately 87% of the mean cardiac volume received a BED (biologically effective dose) of 5 Gy or more in case of left-sided tumors. This share was reduced to 41% after introduction of megavoltage radiotherapy. The target volume in Fuller et al's patients 22 did not comprise the IMNs. Several studies have demonstrated a higher risk of cardiac toxicity when the internal mammary nodes were included in the treatment fields as a result of using wide tangents.2,21–23 Janjan et al 23 showed that electron beam irradiation of the IMNs reduced the total dose to the anterior wall of the left ventricle with 30% compared with photon irradiation. Furthermore, in another study, the use of electrons in the treatment of IMNs and chest wall showed no impact on the risk for ischemic heart disease when patients with irradiated IMNs were compared with those without. 3 In the EORTC trial 22922/10925, 24 the survival and cardiac toxicity were investigated in approximately 4000 patients three years after elective irradiation of the internal mammary and medial supraclavicular nodes. The authors reported no increased cardiac toxicity related to the irradiation of these structures. However, it was suggested that a follow-up of at least 10 years was needed to determine whether cardiac toxicity is increased after radiotherapy. Thus, the indication to irradiate the IMNs and the optimal radiation technique remain controversial. 23
Using the 4-field high-voltage photon technique, our median values for V25Gy (0.0% for both left-sided and right-sided tumors) are far below those presented by Fuller et al, 22 Gyenes et al, 6 and Taylor et al. 11 However, the great variability in our values should be emphasized, as some individuals with left-sided tumors still received rather high doses as a result of variation in internal anatomical differences, with approximately 10% of these patients exceeding a D5% of 25 Gy. Most probably, interpatient variations of the individual breast and thoracic anatomy explain the wide range of cardiac doses found in our study. Due to the heart being located mainly in the left thoracic half, these influences are predominantly recognized for left-sided tumors.
The left anterior descending artery (LAD) is one of the most critical structures in relation to subsequent radiation-related cardiac infarction. In Fuller et al's series, 22 the radiation dose to LAD remained high even after the introduction of megavoltage radiotherapy, whereas the doses for the other coronary arteries were reduced. We have not calculated the individual LAD's radiation dose, as the radiologist considered it impossible to delineate the LAD without the use of contrast agents. According to Taylor et al, 11 the RT doses to LAD was 3 to 4 times higher than the mean cardiac dose. However, due to differences between the authors’ older radiation technique and ours, we refrained from using these figures in our cohort.
Our study has several limitations. In the current work, we have not taken into account the contribution to heart dose of the electron boost treatment for a subgroup of 36 patients. In this case, no dose plan data was available, and the old dose planning system (Helax-TMS) does not provide reliable dose estimates from electron beams. However, patients currently receiving electron boosts at our clinic with Monte Carlo–based treatment plan calculations (using Oncentra) have typically been D5% = 0.2 Gy for a boost dose of 16 Gy. Admittedly, this dose contribution may be somewhat higher in patients with medially located tumors receiving electrons. Thus, the contribution to the heart dose for the electron beam is expected to be minute for the patients in question. Furthermore, respiratory gating was not used, which currently appears to be the most attractive technique for reducing cardiac doses to patients with left-sided disease. However, causes of cardiotoxic events from standard techniques have to be explored, and the results may aid decision making when introducing new techniques in the clinic. Finally, our results are based on CT images without the use of contrast and 1 cm slice thickness, limiting the demonstration of individual details. Thus, our figures should be viewed as the best achievable approximation of cardiac radiation exposure in a large retrospectively assessed series of breast cancer survivors. The main strength of our study is the large sample size of consecutive patients treated in real-life, whose cardiac conditions will be surveyed by a future clinical survey of 10-year breast cancer survivors to quantify potential cardiac risks caused by a defined cardiac dose. This clinical survey will also provide important feedback to the currently used NTCP model, which estimated that the excess cardiac mortality rate in the current cohort probably is low.
In summary, in the majority of included patients with stage II/III breast cancer whole heart D5% was below 25 Gy as recommended by the national guidelines. However, women with left-sided breast cancer still represent a risk group. Our next step will be to perform cardiological examinations in these 10-year survivors relating the individual cardiac radiation exposure to the clinical findings.
Author Contributions
Conceived and designed the experiments: SDF, SJ. Analysed the data: SJ, EM. Wrote the first draft of the manuscript: SJ. Contributed to the writing of the manuscript: SJ, EM, SDF, KF, KT, TDD. Agree with manuscript results and conclusions: SDF, GB, SJ, EM, KF, KT, TDD. Jointly developed the structure and arguments for the paper: SJ, SDF. Made critical revisions and approved final version: SDF, EM, SJ. All authors reviewed and approved of the final manuscript.
Funding
Author(s) disclose no funding sources.
Competing Interests
Author(s) disclose no potential conflicts of interest.
Disclosures and Ethics
As a requirement of publication author(s) have provided to the publisher signed confirmation of compliance with legal and ethical obligations including but not limited to the following: authorship and contributorship, conflicts of interest, privacy and confidentiality and (where applicable) protection of human and animal research subjects. The authors have read and confirmed their agreement with the ICMJE authorship and conflict of interest criteria. The authors have also confirmed that this article is unique and not under consideration or published in any other publication, and that they have permission from rights holders to reproduce any copyrighted material. Any disclosures are made in this section. The external blind peer reviewers report no conflicts of interest.
