Abstract
After ~30 years of widespread usage, immunohistochemistry (IHC) has become a standard method of diagnosis for surgical pathology. Because of the plethora of diagnoses and often subtle nature of diagnostic criteria, IHC finds particular utility in soft tissue tumors. The use of progressively small amounts of tissue for diagnosis highlights the importance of this method. The sensitivity and crispness of IHC stains have progressively improved with the advent of new techniques. Traditionally, IHC detects cell-typic markers that characterize cell phenotypes, such as chromogranin for neuroectodermal tissue, myogenin for skeletal muscle, and cytokeratin for epithelium. However, the advent of genetic discoveries have led to IHC testing for detection of fusion gene products or overexpressed oncogenes associated with deletions and mutations. Proliferation-based markers such as Ki-67 can also be used for prognosis and grading, but more standardization is needed. Development of monoclonal antibody-based pharmaceuticals, such as imatinib or crizotinib, holds the promise of tailored anticancer therapy. IHC thus has assumed importance not only for diagnosis but also for guidance of personalized medicine.
Keywords
Historical Use of Ancillary Testing
Ancillary testing of surgical pathology specimens began soon after the birth of histopathology. A variety of histochemical stains became available, and these were supplemented by a variety of techniques designed to show microbial organisms or specialized cells. Aniline dyes were used to replace the naturally occurring dyes that were first used for microscopy. 1 James Homer Wright used his eponymic stain to demonstrate spirochetes in syphilitic aortas, 2 and the technique was later modified to facilitate the visualization of hematopoietic cells. In 1873, Camillo Golgi began using techniques to demonstrate neuronal tissues. 3 With soft tissue tumors, periodic acid–Schiff (PAS) stains were routinely used until the 1990s to detect the abundant glycogen associated with Ewing sarcoma or the crystals associated with alveolar soft part sarcoma (Fig. 1). Phosphotungstic acid–hematoxylin staining was used to highlight the cross-striations associated with myogenic differentiation in rhabdomyosarcomas. This search for methods to delineate cytologic detail and characteristic tissue structure eventually led to immunohistochemistry (IHC), which added immunological techniques to chemically based stains. 4
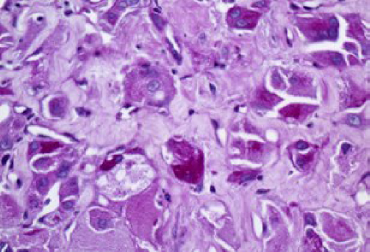
PAS stain of alveolar soft part sarcoma. The purple color highlights intracytoplasmic crystals characteristic of this lesion.
Electron microscopy (EM) was first invented as a technique in the 1940s 5 for observing subcellular detail in cells. When it became apparent that fine cellular detail of tumors could be discerned, 6 then EM became adapted for surgical pathology in general and tumor diagnosis in particular. The appearance, morphology, and topical distribution of submicroscopic organelles correlated with specific tissue types, which in turn were the basis of tumor diagnosis and classification. 7 In this manner, features such as myosin and actin filament bundles and Z-bands were searched for with rhabdomyosarcomas, and neurosecretory granules were detected in peripheral neuroectodermal tumors. Some structures, such as the crystalline organelles (Fig. 2) seen in alveolar soft part sarcoma, 8 became characteristic features of a diagnosis, even if determination of the cell type proved elusive. This method continues to be used as an ancillary technique for tumor diagnosis, but it has lost some of its luster due to the lower cost, widespread availability, and relative ease of IHC.
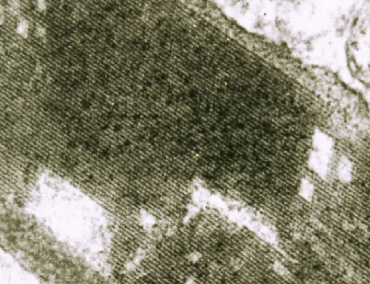
EM of alveolar soft part sarcoma. The tumor cell contains a cytoplasmic crystal that is composed of linear arrays of electron-dense material.
IHC began as an immunofluorescent assay used to detect microbial antigens 4 or the presence of self-directed antibodies such as the fluorescent antinuclear antibody. 9 It initially required fluorescence microscopy, a test performed in the dark and lacking cellular detail. However, it became a popular laboratory test, particularly in microbiology and immunology laboratories, and it remains so till today. Testing can be a direct fluorescent assay, in which a fluorescein molecule is tagged to a primary antibody, such as mouse antirespiratory syncytial virus. 10 Sensitivity is increased by use of indirect testing, in which a fluorescein-tagged secondary antibody, such as goat anti-mouse IgG, is tagged to the primary antibody. 11 This process allows more tagged molecules per target and was used with the initial studies on MyoD IHC (Fig. 3); however, frozen sections were required. With the peroxidase–antiperoxidase (PAP) method, a third antibody, directed against peroxidase, is attached to make a sandwich tag that yields a brown reaction product when diaminobenzidine (DAB) is added as a final substrate. 12
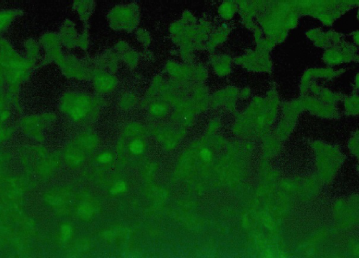
Indirect immunofluorescent stain of MyoD in alveolar soft part sarcoma. With this technique, the fluorescein dye molecule is attached to a secondary antibody directed against the primary one, ie, goat anti-rabbit immunoglobulin, which in turn binds to the primary rabbit anti-myoD polyclonal antiserum. This globoid pattern of positivity highlights a major problem with immunofluorescence as compared with PAP stains: the lack of cellular definition. Because of the configuration, this staining pattern was originally interpreted as nuclear, but in fact, it resulted from MyoD-positive cytoplasmic globules.
A great advance came to IHC with the invention of an avidin-biotin binding method. 13 In this technique, a biotin molecule is attached to the secondary antibody rather than a tertiary antibody. Avidin is then applied with attached peroxidase, which can subsequently react with a peroxidase-sensitive chromogen such as DAB. The subsequent peroxidation of this molecule leads to the colorization of the binding complex. The strong molecular bind between avidin and biotin makes it resistant to stringent precolorization washing and substantially reduces the degree of background staining. This technique has been further enhanced by the use of polymers that bind even tighter to the antibody complex. 14
In surgical pathology, the PAP technique was originally used in ancillary diagnosis of hematological neoplasms, primarily to demonstrate monoclonality. 15 Primary antibodies against lambda and kappa (Fig. 4) light chains were applied to sections of suspected lymphomas. Reactive proliferations showed the presence of both immunoglobulin types, whereas neoplastic diseases showed a predominance of one light chain. This was subsequently translated into investigations on a host of clusters of differentiation (CD) markers. 16 Some, such as lymphocytic common antigen (subsequently renamed CD45 [Fig. 5]), stained all hematopoietic cells, whereas others, such as CD3 or CD20, defined a subset of lymphocytes or other cells.
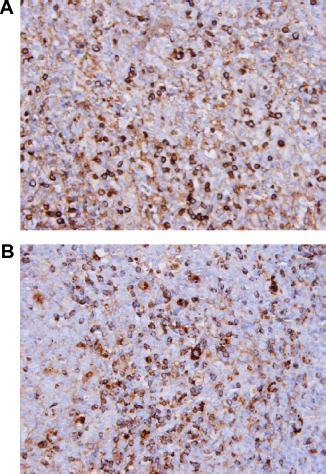
Lambda (A) and kappa (B) immunohistochemical stains of lymphoproliferative lesion. This stain highlights the presence of B-cells with cytoplasmic lambda and kappa light chains. The lack of a predominant lambda or kappa population is consistent with a polyclonal proliferation.
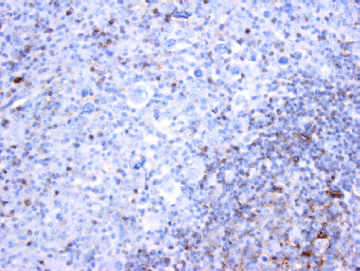
CD45 stain of syncytial variant of Hodgkin lymphoma. The reactive lymphocytes show membranous staining, whereas the neoplastic cells are negative, typical of classic Hodgkin lymphoma.
Cell-Typic Markers
Further use of IHC came with the concept of cell-specific markers (Table 1). This was most developed by Lazarides, 17 when he characterized intermediate filaments. Intermediate filaments (or microfilaments) were first described using EM (Fig. 6) and were found to differ in their molecular and antigenic structure among cell types. Cytokeratins were found in epithelial cells, vimentin in mesenchymal cells, neurofilaments in neural cells, desmin in muscle cells, and glial fibrillary acidic protein (GFAP) in glial cells. Osborn and Weber 18 further refined this concept to show that derivative neoplasms arising from these cell types retained the intermediate filaments characteristic of their parent cells. In this manner, carcinomas expressed cytokeratin (Fig. 7), sarcomas expressed vimentin, neural tumors expressed neurofilaments, myogenic tumors expressed desmin (Fig. 8), and gliomas expressed GFAP. This was a major step forward in ancillary diagnosis but quickly became plagued with exceptions, such as leiomyosarcomas expressing cytokeratin. 19 Nevertheless, this basic concept remains operative till today, and in soft tissue pathology, markers, such as cytokeratin, are used in the diagnosis of epithelioid sarcoma, and desmin is used to confirm rhabdomyosarcomas. Another concept was the use of enzymatic isotypes, similar to the use of muscle-specific serum creatine kinase and lactate dehydrogenase to diagnose myocardial infarction. Neuron-specific enolase (NSE; Fig. 9) was an early marker of this type, which was used for the diagnosis of neural and neuroendocrine neoplasms and also found to be relatively nonspecific. 20 Cytoplasmic proteins such as myoglobin (Fig. 10) 21 and chromogranin 20 were also used to define myogenic and neuroendocrine tumors, respectively. IHC for specific hormones became a component of the workup for endocrine tumors, for example, insulin for beta cell tumors 22 and prolactin for pituitary tumors. 23
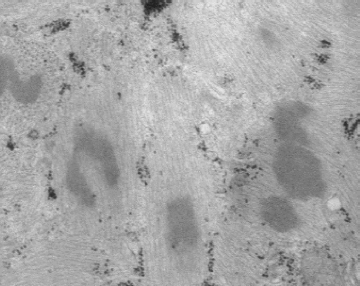
EM showing intermediate filament bundles in muscle fiber of nemaline myopathy. Some forms of this disease are characterized by disorganized clusters of intermediate filaments.
Avidin-biotin immunostain of high molecular weight cytokeratin (CK7) in liver. The CK7 highlights bile ductules and illustrates bile duct proliferation.
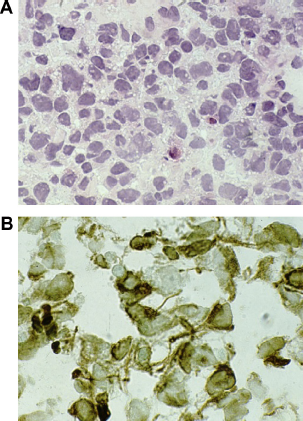
Frozen section of rhabdomyosarcoma with routine hematoxylin–eosin stain (A) and desmin PAP immunohistochemical stain (B). The desmin indicates the myogenic nature of a primitive small round cell neoplasm that cannot be diagnosed by routine stains only.
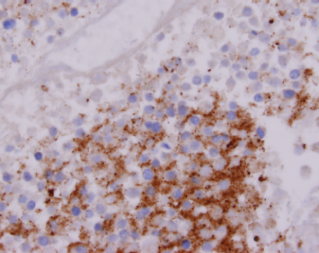
Immunohistochemical stain of NSE in a Ewing sarcoma. NSE is an isozyme of enolase that is primarily expressed in neuronal and neuroendocrine cells. In spite of the name, this neuroectodermal cell marker is no longer considered specific for diagnosis.
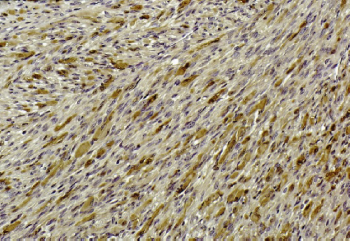
Myoglobin immunohistochemical stain of rhabdomyoma, a benign tumor of skeletal muscle. This demonstrates the presence of differentiated myogenic cells, or rhabdomyoblasts, with abundant cytoplasmic myoglobin, indicated by the dark brown staining.
Selected cell-typic markers commonly used in IHC.*
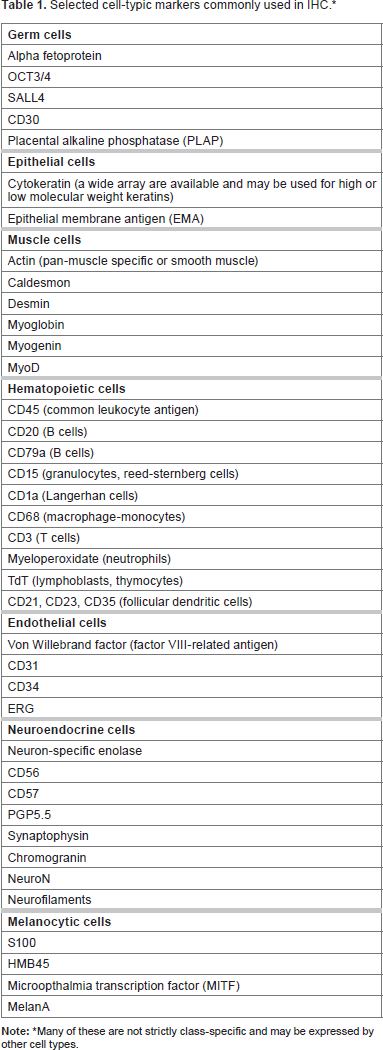
Many of these are not strictly class-specific and may be expressed by other cell types.
Initially, most IHC was directed against cytoplasmic antigens. However, research in the 1980s revealed a set of cell-typic transcription factors, proteins binding to DNA upstream of the transcription site and capable of initiating production of RNA encoding cell-specific proteins. One of the classic proteins was MyoD, a helix–loop–helix protein that bound to upstream regions of genes encoding muscle-specific factors such as myoglobin and desmin. 24 Forced expression of MyoD converted fibroblasts and epithelial cells into muscle cells. Expression of these proteins thus preceded and initiated expression of cytoplasmic proteins and offered a means of detecting the earliest phases of cell differentiation. Because it is a myogenic protein, MyoD (Fig. 11) was tested as an ancillary marker for rhabdomyosarcoma diagnosis, and it was found to be more specific and sensitive than cytoplasmic proteins such as myoglobin. 25 A family of myogenic transcription factors was then found and included myogenin and Myf-5. Similar work produced transcription factors of other tissue types, including neuroD, a marker of neuroendocrine cells, 26 and WT1, a marker of nephrogenesis. 27 WT1 was so named because of its relationship to Wilms’ tumor; constitutional deletions were found in a group of patients prone to have these neoplasms, and a larger group of tumors were found to have acquired mutations. Of interest, both WT1 28 and myoD 29 genes are found in chromosome 11p, a region noted for the presence of CpG islands. 30 CpG islands may contain methylated cytosine, a molecule responsible for parental imprinting by which maternally or paternally derived genes switch off expression of derivative RNA. 31 Abnormalities in imprinting were found to be common in embryonal neoplasms, tumors that look similar to embryonal tissues. 32 Both rhabdomyosarcoma and Wilms’ tumors belong to this group of lesions.
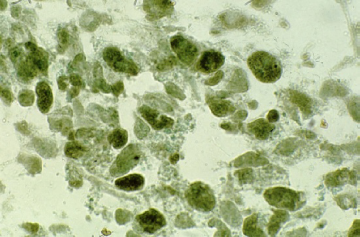
MyoD of rhabdomyosarcoma, PAP stain using monoclonal anti-MyoD (5.1A clone). In comparison with Figure 4, note that this stain clearly shows nuclear expression of the antigen. MyoD is a myogenic transcription factor that binds to DNA in the promoter region of muscle-associated proteins such as desmin and creatine kinase.
Proliferation Markers
DNA replication involves a host of cell cycle-related molecules, often defined by their molecular weight on a polyacrylamide gel. These are used to define cell proliferation and malignancy. In this manner, IHC for p53, p21, and p16 was introduced. 33 Ki-67 (also known as MIB1, after the clone name) is a similar marker that can be used to detect cells that have cycle activity, whether in G1, S, G2, or M phase. 34 Interphase, quiescent cells are not stained (Fig. 12). This marker has been used extensively as a correlate for malignancy, particularly in central nervous system (CNS) neoplasia. 35 However, problems arise because of interobserver variability and lack of definition of involved cells. 36 In the latter case, inflammatory cells cannot easily be distinguished at times from tumor cells, yet immune stimulation causes them to enter the cell cycle and thus be potentially counted. Observer variability lessens the value of these tests because of lack of precision as a diagnostic test. Automated counting does not solve this problem, as an observer versed in histopathology is still needed for more discriminate evaluation. Use of morphometric methods and fixed images has been recommended as the best way to solve this conundrum. 36 Mitosis counting has long been a province of standard histopathology, but can be difficult in some cases. New IHC stains such as phosphohistone 3 (PHH3) offer a method to enhance mitosis counting and possibly introduce automation, but again observer discrimination is required. 37
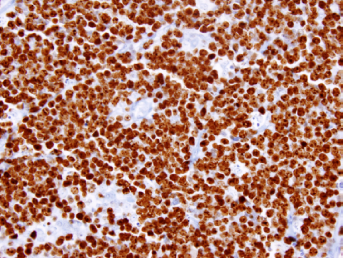
Ki-67 immunostain of Burkitt lymphoma. Burkitt lymphoma is a highly proliferative B-cell neoplasm driven by a gene fusion between the immunoglobulin heavy gene and the protooncogene Myc. As a result, every tumor cell is the cell cycle, resulting in 100% nuclear positivity for Ki-67. Compare the tumor cells with the nonreactive macrophages and endothelial cells, which stain with the blue counterstain.
Although it is not directly involved in cell proliferation, the SMARCB1 gene generates a molecule important for chromatic maintenance and lacking in certain high-grade, aggressive visceral and soft tissue tumors. 38 This protein, INI1, was first used to characterize rhabdoid tumors, lesions predominately affecting kidney, brain, soft tissue, and diverse viscera. Rhabdoid tumors were initially characterized as a phenotype lacking a definitive marker and showing polytypic expression of a variety of proteins. However, exploration in the chromosome 22q deletions that repeatedly occurred, particularly in CNS tumors (known as atypical teratoid/rhabdoid tumors), revealed a single gene locus, SMARCB1, that was lost in lesions arising at all sites, primarily in aggressive infantile neoplasms. Patients may show constitutional deletion of this gene, whose protein product INI1 is highly conserved because of its key role in chromatin maintenance. Discovery of INI1 led to a definitive diagnostic tool for IHC, as expression is completely lost in rhabdoid tumor and present in most other lesions (Fig. 13). However, subsequent studies have revealed that a number of neoplasms, all sometimes containing rhabdoid-like cells, may show similar changes. 39 Primary among them is epithelioid sarcoma, which shows similar genetic and phenotypic features, particularly in the high-grade proximal variant.
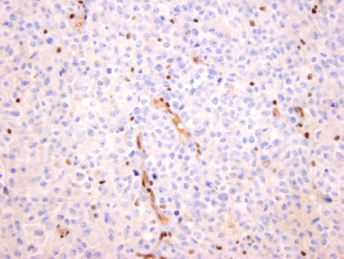
Immunohistochemical stain of INI1 in malignant rhabdoid tumor. This lesion is characterized by loss of the SMARCB1 gene, which encodes INI1 protein. This is a highly conserved gene that is critical to chromatin maintenance. Note the positive staining endothelial cells and leukocytes, which contrast with the INI1-negative rhabdoid tumor cells.
Fusion Proteins
The field of tumor biology was revolutionized in the 1980s and 1990s by the discovery of fusion genes resulting from reciprocal translocations characteristic of specific diagnoses (Table 2). These findings have become part of our standard diagnostic armamentarium, and some have transcended histopathology as a means of final diagnosis and treatment stratification. 40 Although fluorescence in situ hybridization (FISH) offers a molecular means to verify the presence of gene rearrangement and/or fusion, this technique is not strictly IHC, because it is not dependent of an antibody probe for a signal. However, the gene fusions may initiate downstream expression of characteristic cell proteins, and sometime they cause overexpression of one of the proteins forming part of the transcribed fusion molecule. Gene expression array panels have given us a means of querying the tumor genome to identify unique protein expression, 41 but as with all markers, subsequent testing often yields discovery of decreased specificity.
Fusion gene product markers or surrogates detected by IHC staining.**
Surrogate markers that may be discovered by expression arrays or other techniques that target cells with selected fusions. These are not detecting the actual fusion molecule but rather proteins that are overexpressed as a result of it.
WT1 is positive in desmoplastic small round cell tumors only by using IHC antibodies that detect the 5‘-portion of the WT1 protein. The 3’ end of WT1 in the EWSR1-WT1 fusion gene is deleted.
One of the more common childhood lymphomas, ana-plastic large cell lymphoma (ALCL), was found to have a characteristic gene fusion caused by a translocation t(2;5) that fuses the anaplastic lymphoma kinase 1 (ALK) and nucleophosmin 1 (NPM1) genes and their protein products. Subsequent IHC study of ALCL revealed that ALK expression (Fig. 14) could be used as a surrogate for FISH testing and as a specific marker for exclusion of other lymphomas. 42 However, later studies indicate that ALK expression occurs in other neoplasms, but its use has been refined to test neoplasms as diverse as neuroblastoma 43 and lung cancer, 44 and an anti-ALK monoclonal antibody (MoAb) crizotinib 45 has been developed for the treatment of some of these cancers. The FLI1 gene was discovered early on as a fusion partner with EWSR1 in most Ewing sarcomas, and similar efforts led to development of FLI1 IHC (Fig. 15). 46 However, this marker faces specificity challenges akin to those of Ewing markers such as CD99, and its use as a substitute for FISH has not been substantiated. Fusion gene IHC can be further complicated by alternate fusions that involve one side of a translocation but not the other. Ewing sarcoma supplies a good example of this phenomenon, as ~15% of cases show a EWSR1-ERG fusion rather than EWSR1-FLI1. Happily, ERG IHC has become a reliable marker of vascular neoplasms by virtue of its high expression in endothelial cells. 47 To add to this confusion, Ewing-like sarcomas have recently been shown to contain unrelated fusion genes, such as CIC-DUX4 or BCOR-CCNB3. These new undifferentiated sarcomas share many of the markers expressed by Ewing sarcomas. 48
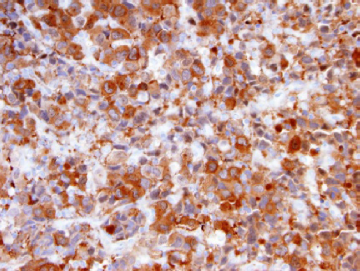
ALK immunostain of ALCL. The positive brown staining indicates the presence of ALK protein. ALK is a receptor tyrosine kinase that is overexpressed in ALCL because of the NPM-ALK fusion. In inflammatory myofibroblastic tumor, ALK is fused with diverse other genes (Table 2).
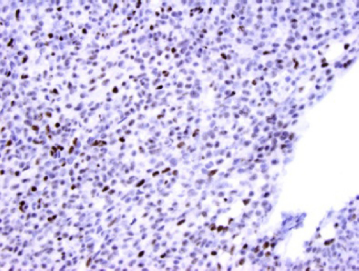
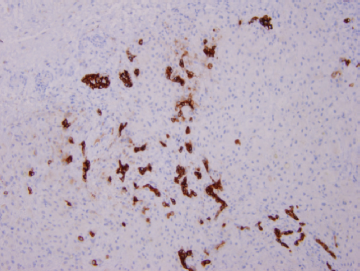
FLI1 immunostain of small round cell tumor. FLI1 is overexpressed in Ewing sarcoma as a result of the EWSR1-FLI1 gene fusion. In this image, the neoplastic cells are negative or faintly staining for FLI1, suggesting that this is not an Ewing sarcoma, but the endothelial cells are positive. FLI1 is normally expressed by endothelial cells. This illustration demonstrates the importance of excluding reactivity by normal cells when evaluating diagnostic IHC.
Another primitive small cell neoplasm has recently been described as characterized by a translocation. In this case, a midline carcinoma affects the head, neck, and mediastinum and contains a characteristic NUT-BRD2 fusion. NUT expression as detected by IHC may be used to confirm the diagnosis, particularly with cases lacking obvious squamous differentiation but expressing cytokeratin. 49 Doubtlessly, fusion proteins will continue to generate similar IHC reagents.
The fusion genes named for the chromosome involved, SSX1-SS18 and SSX2-SS18, have not been used to generate an IHC marker to date. However, gene array studies revealed a downstream protein marker, TLE1, which can be used as a surrogate means of synovial sarcoma diagnosis. 50 Molecular studies of the SSX-SS18 and TLE1 interaction resulted in a remarkable discovery that TLE1 and the fusion protein provided a bridge linking the fusion site with ATF1, a transcription factor, and histone deacetylation factors. 51 This “bridge to badness” thus causes dysregulation of the cellular machinery responsible for gene activation and tumor cell transformation. TLE1 has become a standard component of sarcoma evaluation, and it may be used as a screening test for further genetic evaluation. However, as with many markers, it is not completely specific, as other sarcomas may show a partial weak expression that overlaps with synovial sarcomas. 52
Similar gene array studies have been performed with PAX3-FOXO1 and PAX7-FOXO1, the fusions found in the majority of rhabdomyosarcomas with alveolar histology. Array studies indicated that fusion-negative alveolar rhabdomyosarcomas (ARMS) have no distinct biological or clinical differences from embryonal rhabdomyosarcoma (ERMS). However, the histological distinction between ERMS and ARMS can be a subtle one, even for experienced observers. Gene expression patterns however indicated that a panel of IHC tests, including myogenin, AP2β (Fig. 16), and HMGA2, may discriminate between fusion-positive ARMS and fusion-negative ARMS and ERMS. 53 This may lead to more selective screening in future studies, particularly in locations lacking facilities for FISH or reverse transcription polymerase chain reaction testing.
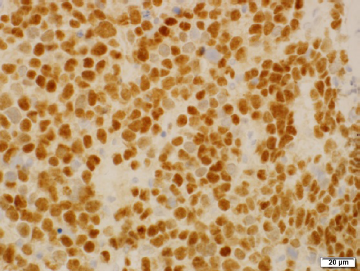
AP2β in rhabdomyosarcoma. PAX-FOXO1 fusion-positive rhabdomyosarcomas generally show strong, diffuse nuclear expression of AP2β and myogenin, as in this image.
Therapeutic Applications of IHC
The use of IHC has also taken a very interesting turn in potential applications and now is used not just for diagnosis and prognostication, but for specific therapy decisions. This application first became a standard practice with breast cancer, with quantitation of estrogen and progesterone receptors. The use of the antiestrogen receptor tamoxifen became a game changer for the outlook of receptor-positive breast cancer, and its use depends on the ability of pathologists to accurately quantitate receptor expression. However, variability in staining may be from factors such as over- or under-fixation, leading the College of American Pathologists to regulate methodology and reporting. Nevertheless, conflicting data on the importance of fixation time have appeared.54,55 Following discovery of the anti-HER2/neu MoAb, transtuximab, IHC determination of expression of the protooncogene HER2/neu was added to the breast tumor panel, leading to similarly dramatic outcomes of patients with positive tumors. So-called triple-negative breast cancers lacking estrogen receptor, progesterone receptor, or HER2/neu expression continue to be the research focus on new strategies for this pervasive neoplasm. 56 The role of HER2/neu has now been expanded to include treatment of gastric adenocarcinoma, particularly in patients with metastasis. 57
A similar therapeutic approach came with CD20, a marker of B-cells and their derivative lymphomas. MoAbs developed against CD have resulted in an effective antilymphoma therapy, rituximab, which has dramatically changed the way oncologists treat B-cell lymphomas. Interestingly, rituximab resistance can be recognized by the lack of CD20 expression, which allows emergent clones to escape immune surveillance. However, these resistant neoplasms continue to express other markers of B-cell differentiation, such as CD19 and CD79a. Thus, CD20 IHC may be useful as both a marker of susceptibility to rituximab and the emergence of chemotherapy resistance. 58
This concept of cell marker-specific antineoplastic therapy currently is limited to plasma membrane proteins, as we lack an effective and safe transport mechanism for intracellular and intranuclear drug delivery. However, therapy can still be designed against molecules interacting with surface receptors, such as tyrosine kinases. This has led to a host of new tailor-made pharmaceuticals that suppress tumorigenic signals from proteins such as c-kit, ALK, c-fos, vascular endothelial growth factor (VEGF), and insulin-like growth factor 2. Perhaps the most successful to date has been imatinib, which is directed against c-kit. Tumors with high c-kit expression resulting from mutations may be recognized by IHC using anti-CD117 (Fig. 17), an MoAb against c-kit. The best known neoplasm with this mutation is the gastrointestinal stromal tumor (GIST). GISTs contain activating mutations that lead to high c-kit expression and a strong CD117 IHC staining pattern. 59
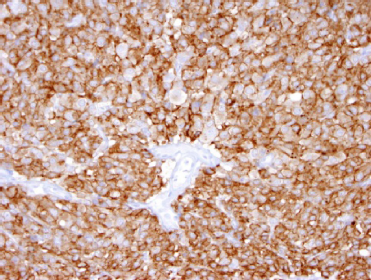
CD117 immunostain in GIST. GISTs overexpress CD117 (c-kit) as a result of an activating mutation. As a result, therapy with an anti-GIST MoAb pharmaceutical, imatinib, is often successful in treating these lesions.
Therapeutic MoAbs such as bevacizumab have been developed against VEGF and have proven efficacy in tumors expressing this growth signal. As a result of the cross-reactivity of this antibody to normal endothelial cells, cardiovascular complications may develop. 60 Some MoAb pharmaceuticals, such as sorafenib, 61 target multiple tyrosine kinase growth factor receptors, including VEGF receptors, Raf kinase, platelet-derived growth factor receptor β, FMS-like tyrosine kinase 3, c-KIT, and RET-receptor tyrosine kinase, and display therapeutic and cytotoxic properties similar to bevizamab.
Recognition of ALK as a tumor driver has led to the development of an anti-ALK MoAb, crizotinib, as targeted therapy. Crizotinib also targets ROS-1 and MET, tyrosine kinases similar to ALK. This has been used for the therapy of ALK-driven tumors, such as inflammatory myofibroblastic tumor, and some forms of lung cancer. 62 However, acquired tumor resistance may develop. Use of these agents highlights the potential utility of ALK IHC in not only diagnosis but also therapy and recognition of resistant tumor clones.
Conclusion
After decades of clinical usage, IHC survives as a commonly used technique for ancillary diagnosis, and its potential for assistance in recognition and management of targeted therapies is still being developed. However, because of issues with nonspecificity and sensitivity, pathologists must avoid becoming overreliant on IHC for primary diagnosis, and we should keep an open mind when dealing with discrepant results, although unexpected results may offer rewards in redirected studies and successful diagnoses.
New targeted antitumor therapies against driver mutations also hold promise for even greater utility of IHC in oncologic treatment and monitoring. However, caveats similar to those above may threaten success, and additional studies will be necessary as this therapeutic approach matures. Correlation with other technologies such as array hybridization and deep sequencing will doubtlessly be forthcoming. This promises opportunities for diagnostic pathologists as part of the healthcare team, but full participation will require continuing education with regular surveys and careful evaluation of prospective literature.
Author Contributions
Wrote the first draft of the manuscript: DP. Made critical revisions and approved final version: DP. Author reviewed and approved of the final manuscript.
