Abstract
A new chemosensor has been used to monitor atmospheric nitrogen oxides [NO + NO2] at parts per billion (ppb) level. It is based on the catalytic reaction of nitrogen oxides with rhodamine B hydrazide (RBH) to produce a colored compound through the hydrolysis of the amide bond of the molecule. A simple colorimeter has been used to monitor atmospheric nitrogen dioxide at ppb level. The air samples were purged through a sampling cuvette containing RBH solution using peristaltic pump. The proposed method has been successfully applied to monitor the ambient nitrogen dioxide levels at traffic junction points within the city limits and the results obtained are compared with the standard Griess-Ilosvay method.
Introduction
Nitrogen oxides play a key role in the atmospheric processes. 1 Among the oxides of nitrogen, two of them, that is, nitric oxide and nitrogen dioxide are generally represented as NO x [NO + NO2] and their presence in the atmosphere can cause a severe impact on the quality of ambient air. Nitric oxide even at 1 ppm concentration level is relatively harmless, but nitrogen dioxide at higher concentration can cause respiratory distress. 2 It can also react with other compounds in the air to form inorganic and organic nitrates such as nitric acid and peroxyacetyl nitrate, respectively, which are mainly responsible for the photochemical smog formation. 3
Nitrogen oxides are the precursors of acid rain and ozone formation, which can damage human health, vegetation, and aquatic ecosystems.4,5 Several major reviews of the health effects of NO x have been reported in the literature in recent years.6,7 Nitrogen dioxide damages lungs in three ways: (i) its conversion to nitric and nitrous acids in the distal airways, which directly damage certain structural and functional lung cells; (ii) it initiates free radical generation which results in protein oxidation, lipid peroxidation, and cell membrane damage; and (iii) it reduces resistance to infection by altering macrophage and immune function. 8 The immediate symptoms of exposure to nitrogen oxide vapors may include coughing, fatigue, nausea, choking, headache, abdominal pain, and difficulty in breathing. 9 A symptom-free period of 3-30 hours may then be followed by the onset of pulmonary edema with anxiety, mental confusion, lethargy, and loss of consciousness. 10 The primary site of toxicity is the lower respiratory tract. Low concentrations may initially cause mild shortness of breath and cough, and then after a period of hours to days, victims may suffer bronchospasm and pulmonary edema. Inhalation of ambient air containing very high concentration of nitrogen oxides can cause burns, spasms, tissue swelling in throat, and upper airway obstruction. 11 Children may be more vulnerable to corrosive agents than adults because of the relatively smaller diameter of their airways. Absorption of nitrogen oxides can lead to a weak rapid pulse, dilated heart, chest congestion, and circulatory collapse. 12 High-dosage exposure may convert Fe2+ in hemoglobin to Fe3+ by virtue of the presence of nitric oxide (NO) causing methemoglobinemia and impaired oxygen transport. 13
Increased NO x inputs into terrestrial and wetland systems can lead to changes in plant species composition and diversity. 14 Nitrogen inputs to aquatic ecosystems such as those found in estuarine and coastal waters can lead to eutrophication (a condition that promotes excessive algae growth, which can lead to a severe depletion of dissolved oxygen and increased levels of toxins which are harmful to aquatic life). 15 Nitrogen alone or in acid rain form can acidify soils and surface waters. It causes the loss of essential plant nutrients with the increase in levels of soluble aluminum that are toxic to plants, and acidification of surface water creates conditions of low pH and high levels of aluminum that are toxic to fish and other aquatic organism. 16
Nitrogen dioxide is a reddish brown gas with a characteristic pungent odor, and is corrosive and highly oxidizing in nature. 17 It is formed naturally in the atmosphere in substantial quantities and is also a by-product of human activity. Natural sources include oxidation of atmospheric nitrogen by lightning, forest fires, grass fires, bush fires, and oxidation of ammonia releases from the soil. 18 Automobiles and other mobile sources contribute about half of the NO x that is emitted into atmosphere. Electric power plant boilers produce about 40% of the NO x emissions from the stationary sources. 19 Substantial emissions are also added by such anthropogenic sources such as industrial boilers, incinerators, gas turbines, reciprocating spark ignition, and diesel engines. 20 Among stationary sources, iron and steel mills, cement manufacturing, glass manufacture, petroleum refineries, and nitric acid manufacturing units are significants. 21 However, the major sources of nitrogen oxides in urban areas are mainly from human activities. In the atmosphere, nitric oxide oxidizes slowly to nitrogen dioxide and the process is enhanced in outdoors in the presence of ozone.
Several methods for the measurement of NO x concentration from atmospheric air have been reported in the literature. 22 Most of these methods are based on instrumental techniques like spectrophotometry, chromatography and chemiluminescence processes. Chemiluminescence methods are widely used in NO x measurement that are mainly based on the catalytic or photolytic reduction of NO2 to NO and its subsequent gas-phase reaction with ozone. In recent years, flow injection-based methods have been developed for the continuous measurement of nitrogen dioxide from ambient air and nitrite/nitrate levels from industrial effluents. NO x is usually absorbed in a suitable absorber solution initially and fixed as nitrite ion followed by its determination by various methods.23,24 In recent years, flow injection analysis based on diazo coupling reaction and the color intensity measurement gained a lot of emphasis due to their simplicity, low cost, and easy adaptability even under modest facility conditions. Rhodamine and its derivatives have been used as chemosensors in the quantification of several anions and cations due to their excellent optical properties as well as high sensitivity. Rhodamine B hydrazide (RBH) quantitatively reacts with nitrite at pH 3 to generate pink colored rhodamine. 25 The intensity of the pink color produced has been correlated to nitrite concentration. In this report, a simple flow injection analysis based on the reaction of NO x with RBH and its catalytic activity to open the spirolactam ring in the molecule has been described. The catalytic activity of NO x with RBH has been used to monitor ambient nitrogen oxides (NO x ) in the atmosphere. The method is based on the suction of atmospheric air through the sampling cuvette containing RBH solution at pH 3 and its color intensity measurement at 563 nm. A sensitive flow injection protocol using a conventional colorimeter and a peristaltic pump for the continuous measurement of ambient nitrogen oxides at trace level has been described. The proposed method can be used in industrial locations to monitor ambient NO x levels continuously and it can be automated for measuring the variation of NO x concentrations; thereby its emissions can be regulated at the source itself.
Experimental
Apparatus
Absorbance measurements were made using Thermo Scientific UV-Vis Spectrophotometer (model: Helios Zeta V 8.01) with 1-cm quartz cuvettes. Air sampling was carried out using Peristaltic pump (Miclins, Chennai, model-PP 30) with suitable suction devices. Control Dynamics (Mumbai) digital pH meter (model APX 175 E/C) was used for all pH measurements. Portable colorimeter was used for ambient air sampling study at city traffic junction points.
Reagents and Solutions
Standard sodium nitrite stock solution (1000 μg mL−1) (SD Fine Chem. Mumbai, India): It has been prepared by dissolving 0.15 g of pre-dried sodium nitrite (at 105 ± 5°C for an hour) in distilled water and diluted to 100 mL. Working standards were prepared by diluting the appropriate volumes of stock solution on the day of use.
RBH (0.02 M) (Sigma-Aldrich, Bangalore, India): It has been prepared by dissolving 0.0913 g of RBH in small volume of acetonitrile and diluted to 100 mL with distilled water.
Griess-Ilosvay reagent (Sigma-Aldrich): It is commercially available reagent and it is used as a standard reagent for the quantification of nitrite or oxides of nitrogen from ambient air. It is a mixture of sulphanilic acid and α-naphthylamine in acetic acid medium. This reagent is used as a standard reagent and it is well accepted by the American Water Works Association. The commercially available Griess-Ilosvay reagent has been recognized as a standard reagent in the estimation of nitrite from liquid samples as well as nitrogen dioxide from gaseous samples after purging the sample gas through this reagent.
Formaldehyde (SD Fine Chem): Used directly as received Sodium bisulfite (SD Fine Chem): It was used as source in the generation of sulfur dioxide. The concentration of generated sulfur dioxide was standardized using standard protocol.
Hydrogen sulfide: Sodium sulfide (Na2S·9H2O–-Sigma-Aldrich) has been used as source in the generation of gaseous hydrogen sulfide. Its concentration was calculated based on standard protocols.
Bromine: Liquid bromine (SD Fine Chem) was used after dilution to the required concentration.
Acetate buffer (pH: 3): Prepared by mixing 0.1 M sodium acetate (SD Fine Chem) and 0.1 M acetic acid (SD Fine Chem) by adjusting the pH to 3. The solution was diluted to 100 mL with distilled water.
Nitric acid (SD Fine Chem) (1:1): It was prepared by diluting equal volumes of conc. HNO3 and distilled water.
Recommended Procedures
Proposed method: Known volumes of ambient air samples containing oxides of nitrogen were purged through a sampling cuvette fitted in a portable colorimeter containing RBH solution buffered at pH 3 after adjusting the air flow rate using peristaltic pump. The intensity of the pink color formation in the cuvette has been measured at 563 nm and the concentration of oxides of nitrogen was calculated with reference to the standard calibration curve constructed using standard nitrite solutions.
Standard method: Air samples containing oxides of nitrogen were purged through a sampling cuvette containing Griess-Ilosvay reagent fitted in a portable colorimeter after optimizing the gas flow rate using peristaltic pump. The color intensity of the resulting solution was measured at 560 nm and the concentration of oxides of nitrogen was measured similar to the proposed procedure.
Laboratory Setup Used for Nitrogen Dioxide Generation and Its measurement
A portable and conventional spectrophotometer was used to measure the absorbance values of the pink colored rhodamine B formed from the catalytic reaction of NO2 with RBH. A glass Carboy containing known mass of high purity metallic copper (Anchor cables, Bangalore, India) wire turnings have been used as NO2 generator. Addition of 1:1 nitric acid into container generates nitrogen dioxide. The generated nitrogen dioxide was purged through a sampling cuvette containing RBH solution buffered at pH 3 using peristaltic pump with an optimum flow rate. The gas flow rate was adjusted in such a way that the gas bubble residential time in the reactant solution should be high enough so that the gaseous nitrogen dioxide catalyzes the reagent solution to form pink colored rhodamine B. The color intensity of the compound has been measured at 563 nm and it is correlated to NO2 concentration with reference to the standard calibration curve. All tubing connections were given using Teflon tubes of different diameters as shown in Figure 1.

Schematic of laboratory setup used for nitrogen dioxide generation and its measurement using flow type colorimeter.
Experimental Setup Used for the Measurement of Ambient Nitrogen Dioxide at Traffic Junction point
A portable colorimeter and a peristaltic pump combination have been used as a field sampling experimental setup for the measurement of ambient nitrogen oxides. The atmospheric air was purged using peristaltic pump at controlled flow rate through the portable colorimeter fitted with a sampling cuvette containing RBH solution buffered at pH 3. As the ambient air passes through the solution, the nitrogen oxides present in the atmospheric air catalyzes the reagent to convert colorless RBH into a pink colored rhodamine. The color intensity of the solution has been continuously measured and the absorbance values were correlated to the concentration of nitrogen oxides present in the ambient air in comparison with standard calibration curve. The flow injection setup was used for monitoring the NO x levels in the ambient air as shown in Figure 2.

Experimental setup used for the measurement of ambient nitrogen oxides [NO + NO2] at traffic junction point within city limits.
Discussions
Nitrite or nitrogen oxides quantitatively react with RBH solution in acidic medium to give pink colored rhodamine B. 24 Hence, an attempt has been made to explore the catalytic reaction of nitrogen oxides [NO + NO2] with colorless leuco form of RBH to produce pink colored rhodamine B as a simple flow injection procedure. The intensity of pink colored compound produced in the reaction has been correlated to the NO x concentration and it has been applied to measure the ambient nitrogen dioxide from atmospheric air.
Initial studies were carried out using standard nitrite and its catalytic reaction on RBH to produce pink colored compound. Standard aliquots of nitrite samples were treated with colorless RBH to generate pink colored rhodamine B. The color intensity of the generated rhodamine B was measured at 563 nm and correlated to nitrite concentration.
Species Responsible for color
The catalytic reaction of nitrogen oxides with colorless RBH in acidic medium to form pink colored rhodamine B. This transformation from colorless leuco form to colored rhodamine B was induced by the catalytic effect of NO x through spirolactam ring opening process and the absorbance values were measured at 563 nm. The species responsible for color in the reaction is shown in Scheme 1.
The catalytic reaction of nitrogen oxides with colorless RBH in acidic medium to form pink colored rhodamine B.
Optimization Study
All the parameters influencing the color development were optimized using standard nitrite solutions and the optimized conditions were incorporated in the recommended procedure. During optimization study, standard nitrite solutions were added in place of nitrogen dioxide to the RBH solution. Hence, all the reaction variables have been optimized using stagnant solution in a 10 mL volumetric flask containing RBH solution buffered at pH 3 using acetic acid–acetate buffer. Similarly, a reagent blank was also prepared using 2 mL of RBH and 2 mL of acetate buffer (pH 3) solution in 10 mL standard flask and diluted to mark with distilled water. All absorbance values during optimization study were measured against the reagent blank at 563 nm.
Effect of Rbh concentration
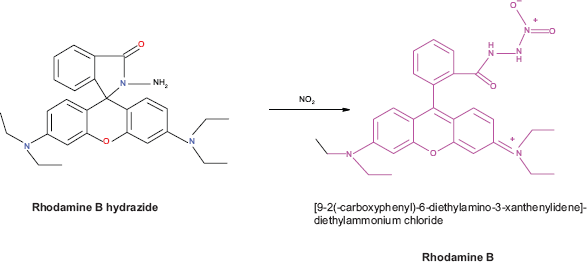
The effect of RBH concentration was varied in order to establish the optimum concentration of RBH required for maximum absorbance by varying its concentration in the volume range 0.5-5 mL. Different volumes of RBH were taken in a series of 10 mL volumetric flasks containing 2 mL of acetate buffer (pH 3). To these solutions, 2 μg mL−1 of nitrite was added and allowed to stand for 5 minutes, and then the absorbance values were measured at 563 nm. It is evident from the graph that the maximum absorbance for the sample was obtained when the RBH concentration is in the range 2.5-4.5 mL. Hence, 3 mL of 0.02 M RBH solution was used as an optimum condition in all further studies (Fig. 3).
Effect of rhodamine B hydrazide.
Effect of pH
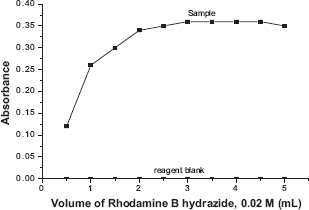
The effect of pH for the catalytic reaction of nitrite on RBH to produce pink colored rhodamine B compound was studied. In alkaline condition, nitrite did not catalyze RBH to produce pink colored rhodamine. Hence, the effect of pH was studied only in acidic medium by varying the buffer pH. In these experiments, various buffer solutions of 1 mL each in the pH range 0.5-7.0 were added to maintain the constant pH in 10 mL volumetric flasks containing 3 mL of 0.02 M RBH along with 2 μg mL−1 of nitrite. The solutions were allowed to stand for 5 minutes and the absorbance values were measured at 563 nm. The maximum absorbance for the sample was observed in the pH range 1-5. Hence, an optimum pH of 3 was fixed and it was maintained by the addition of 3 mL of acetate buffer of pH 3 in all further studies (Fig. 4).
Effect of pH.
Effect of Reaction time
The optimum time period required for the quantitative catalytic reaction of nitrite on RBH was next investigated by allowing the solutions to stand for different time intervals before absorbance measurement. The color development is instantaneous; hence, the effect of time on the catalytic reaction of nitrite on RBH was not required.
The instantaneous reaction between RBH and nitrite to form pink colored dye has inspired us to develop a simple flow injection analysis for the quantification of nitrogen oxides from ambient air using RBH as a single reagent utilizing simple instrumental setup, that is, portable spectrophotometer for absorbance measurement and a peristaltic pump for purging the air samples from atmosphere.
Calibration Plot
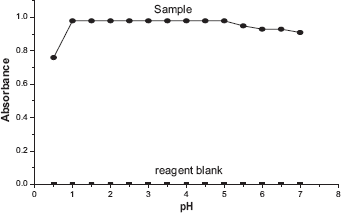
Five-milliliter aliquots of standard nitrite solutions containing 0-10 μg mL−1 nitrite were added into a series of 10 mL volumetric flasks containing 1 mL of 0.05% sulphanilic acid and 1 mL of 2N HCl. The contents were mixed well and allowed to stand for 2 minutes. Then 1 mL of 0.05% α-naphthyl amine was added and diluted to 10 mL with distilled water and the absorbance values were measured at 560 nm using 1-cm quartz cuvette (Fig. 5).
Calibration plot.
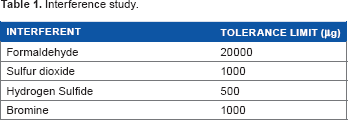
Interference study
In order to evaluate the suitability of the proposed method for the determination of nitrogen dioxide in air, the effect of interference of several species in the determination was examined. Initially, the effect of common atmospheric air pollutants like formaldehyde, sulfur dioxide, and hydrogen sulfide in the determination of nitrogen dioxide was studied. These species were introduced in the form of their respective anions. Formaldehyde did not interfere up to 20,000 μg level in the proposed method, whereas the sulfur dioxide at concentrations above 1000 μg interfered causing decrease in the absorbance values. Hydrogen sulfide up to 500 μg did not interfere but at higher concentrations it i nterfered by decreasing the absorbance values. Bromine did not interfere up to 1000 μg level in the proposed method (Table 1).
Interference study.
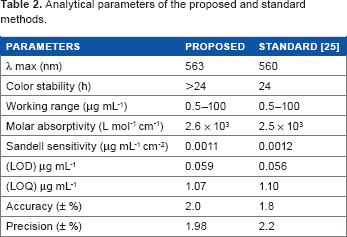
Method Validation
The proposed method has been validated by comparing the results with standard method. The analytical merits of the proposed method as well as standard methods are listed in Table 2.
Analytical parameters of the proposed and standard methods.
Naked Eye sensor
The proposed method based on the catalytic reaction of nitrogen oxides with colorless RBH in acidic medium to generate pink colored rhodamine B can be used as a naked eye sensor. The colorless RBH in leuco form to colored rhodamine B was induced by the catalytic effect of NO x through spirolactam ring opening process and the change in color was measured by naked eye. This method can be applied in the quantification of oxides of nitrogen from various environmental and biological samples as shown in Figure 6.

Schematic representation of reaction between NO2 and RBH.
Application Study
In order to check the suitability of the proposed method for monitoring ambient nitrogen oxides, known volumes of air samples were purged through the RBH solution and the results were compared with the results obtained by the standard Griess-Ilosvay method. 25
Determination of Nitrogen Oxides [NO2] from Ambient air
Air samples were drawn directly from atmospheric air using peristaltic pump at a flow rate of 0.3 L min−1. The ambient air was purged into the sampling cuvette containing RBH solution buffered at pH 3. The absorbance values were measured continuously at 563 nm for different time intervals on different days. The measured concentrations of NO x are given in Table 3. The values of nitrogen oxides measured by the proposed method at a traffic junction point within the city limit have varied and are in agreement with the values measured by the standard method as well as reported by the Karnataka State Pollution Control Board, a statutory body of pollution prevention agency of the local area. The proposed method can be used as a simple flow injection analytical procedure for the measurement of NO x in industrial locations.
Determination of nitrogen dioxide [NO2] from ambient air. * Flow rate: 0.2 L min−1.
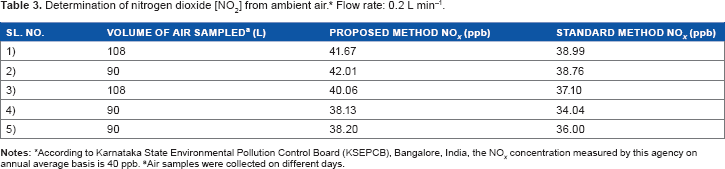
According to Karnataka State Environmental Pollution Control Board (KSEPCB), Bangalore, India, the NO x concentration measured by this agency on annual average basis is 40 ppb.
Air samples were collected on different days.
Conclusions
A simple protocol has been proposed based on the catalytic reaction of nitrogen dioxide with colorless RBH to form pink colored rhodamine B. A simple flow injection analysis for the quantification of oxides of nitrogen [NO x ] from ambient air has been developed using a simple instrumental setup, which consists of portable spectrophotometer and a peristaltic pump. The atmospheric oxides of nitrogen can be measured by purging the ambient air through the sampling cuvette containing RBH solution buffered at pH 3 using peristaltic pump. The developed method has been applied for the continuous measurement of ambient nitrogen oxides present at ppb level at traffic junction points within the city limits. The results obtained are in good agreement with the standard method. The proposed method can be automated and used as a flow injection analytical procedure for continuous measurement of both NO and NO2 which are present in the atmospheric air. This method serves as an alternative one in place of existing methods at ppb level for NO x measurement in ambient air or industrial atmospheres where oxides of nitrogen are the major flue-gas emissions. The method can be automated and it can be used as a warning signal for detecting NO x concentrations under specified industrial atmospheres.
Author Contributions
Conceived and designed the experiments: PM and VY. Analyzed the data: PM and VY. Wrote the first draft of the manuscript: PM and VY. Contributed to the writing of the manuscript: PM and VY. Agree with manuscript results and conclusions: PM and VY. Jointly developed the structure and arguments for the paper: PM and VY. Made critical revisions and approved final version: PM and VY. Two authors reviewed and approved of the final manuscript.
