Abstract
Systemic sclerosis is manifested by the massive deposition of collagen within various organ systems. Endothelin-1 has taken a pivotal role as not only a potent vasoconstrictor, but also as a profibrotic intermediary. Bosentan is proving to be a viable therapeutic option in alleviating manifestations of systemic sclerosis other than pulmonary hypertension. There are many reports in the literature profiling bosentan's safety, efficacy and tolerability in patients. In addition, it has greatly improved patients’ quality of life, reduced the number of hospitalizations and increased the time to clinical worsening of the disease. The clinical indications for use of bosentan in a wide array of patients has served as a valuable asset in the medical management of systemic sclerosis.
Introduction
Systemic sclerosis (SSc) is a heterogeneous autoimmune disease that is manifested by the massive deposition of collagen and altered extracellular matrix turnover. Multiple vital organ systems can be involved, including the skin, blood vessels, heart, lungs and kidneys. The rate and degree of these various organ involvement and symptoms vary. Worldwide, approximately 240 per 1,000,000 people per year are affected by scleroderma. It is more commonly found in the United States 1 . 75% of patients are females typically in their fourth decade of life without a predilection in race. The diagnostic criteria classified by the American College of Rheumatology was instituted due to the many different presentations observed. The following criteria for diagnosis include the presence of;
Typical sclerodermatous skin changes; cutaneous fibrosis
Other skin manifestations attributable to systemic sclerosis, such as digital scars, bilateral finger or hand edema, abnormal skin pigmentations, Raynaud's phenomenon
Visceral manifestations such as pulmonary fibrosis, pulmonary hypertension, myocardial fibrosis, esophageal dysphagia or dysmotility or colonic sacculations. 2
Systemic sclerosis is typically divided into either the diffuse or limited disease form. Even though patients with the diffuse form will have thicker skin over a broader degree of their body, the limited form predisposes people to abnormalities of the systemic and pulmonary circulation. Without therapy, 50% of patients will succumb to this disease within 5 years. When treatment is provided, the 5-year survival is over 80%. The overall therapy for systemic sclerosis is focused on reducing the level of inflammation and preventing fibrosis. Corticosteroids and DMARDS have been routinely used for symptomatic management. Many factors are involved in the pathogenesis of systemic sclerosis, including genetic, environmental and autoimmunological processes.
Despite the aforementioned therapeutic options, accumulating evidence suggests that endothelin-1 in not only a potent vasoconstrictor, but also a strong profibrotic mediator integrally involved in systemic sclerosis. Although approved by the Food and Drug Administration only for scleroderma associated pulmonary hypertension, bosentan is proving to be useful in other various aspects of this disease. In the remainder of this manuscript we will focus on both the on-label and off-label use of bosentan on systemic sclerosis. It should be aforementioned that some of the currently available information regarding various manifestations of systemic sclerosis are based on small studies in abstract form.
Review of pharmacology, mode of action, pharmacokinetics of bosentan
Dosage/administration
Bosentan should be initially administered orally at 62.5 mg twice daily for 4 weeks, then titrated up to the maintenance dose of 125 mg twice daily. In individuals who develop transaminitis, the dose should be adjusted accordingly.
Mode of action
Bosentan is an orally administrated antagonist of both endothelin receptors (ETA and ETB). It has a marginally higher affinity for the ETA receptor. Essentially, bosentan competitively inhibits endothelin-1 functions.
Pharmacokinetics
Peak plasma concentrations are reached within 5 hours after oral administration of 125 milligrams of bosentan. The bioavailability is approximately 50% and 98% protein bound (mainly albumin) and is unaffected by food. Bosentan's half-life is approximately 5 hours, with steady state concentrations being achieved within 3-5 days after multiple doses. It is primarily metabolized by the hepatic system via the cytochrome P450 enzyme pathway by CYP2C9 and CYP3A4 and eliminated through biliary excretion.
Warnings
Although bosentan can be safely used in mild hepatic failure (Child-Pugh class A), it should be avoided in moderate and severe forms (Child-Pugh B and C). In individual with renal failure, bosentan can be safely administered when creatinine clearance is greater than 30 mL/min. Further renal impairment (less than 30 mL/min) can increase metabolites by two-fold. Co-administration of bosentan with other medications should be evaluated. Bosentan decreases levels of cyclosporine, glyburide, simvastatin, and warfarin by up to 50% due to induction of CYP3A4 and/or CYP2C9 pathways. The co-administration of bosentan and cyclosporine are additionally contraindicated due to resulting in increased plasma levels of bosentan.
It is advisable that women taking bosentan not attempt becoming pregnant. Bosentan taken while pregnant is highly likely to cause fetal harm and has a pregnancy-risk category × warning.
Efficacy and comparative studies
Bosentan has been evaluated in many forms of systemic sclerosis. We have divided the use of bosentan to specific organ involvement to assess its clinical benefit.
Cutaneous fibrosis
Skin thickening is a characteristic feature of systemic sclerosis. More extensive skin thickening coincides with more severe internal organ manifestations, a poorer prognosis and increased morbidity. Measuring dermal skin thickness is validated with the use of the modified Rodnan skin score. Domsic presented data predictors of outcome in 689 patients with systemic sclerosis with diffuse cutaneous involvement. 3 The rapid skin thickness progression rate by the use of modified Rodnan method was determined to be a predictor of internal organ involvement and 5-year mortality. Rapid, intermediate and slow STRP cumulative 5-year survival was 72%, 81% and 82% respectively. The three significant predictors of internal organ involvement at one year included being of male gender having the presence of anti-topo I antibody and a rapid STRP.
A study on Bosentan therapy administered to 11systemic sclerosis patients with either form of the disease for various duration from 11 to 18 months evaluated the long-term efficacy of this drug. 4 All the patients experienced improvement in cutaneous fibrosis. Early changes were observed within the first month of therapy, irrespective of the duration of having the disease process and/or baseline skin scores. A gradual improvement was noted in the skin pigmentation, hypertichosis and hyperhidrosis, but interestingly, there were increases in the development in number and size of telangiectasias following bosentan therapy for over 12 months. Larger studies are warranted to confirm these findings.
In addition to treatment advantages, the same authors investigated the role of bosentan for the prevention of cutaneous fibrosis in addition to the therapeutic advantages. 5 Rodnan skin score was used for CF assessment. This assessment was always done by the same Rheumatologist to prevent inter-observer variability. 8 patients with systemic sclerosis were enrolled in this trial.
The earliest changes were noted on the face, chest, abdomen and back and then spreading distally. Again, gradual improvement in the skin pigmentation was observed. Despite a well described improvement with therapy, it was not determined that bosentan can be used for the prevention of cutaneous fibrosis.
Digital ulcers
Digital ulcers cause substantial impairment of daily living activities and have the potential to become infected.6,7 Ulcers are defined as loss of surface epithelialization and do not fissure or crack in the skin or areas of extrusion from calcinosis cutis. Bosentan is able to reduce the number of digital ulcers. Poor digit perfusion due to Raynaud's phenomenon, sclerodactyly, calcinosis, and skin trauma may contribute to ulcer initiation. Calcium channel blockers improve Raynaud's, but are ineffective against digital ulcers.
RAPIDS-1 study, a double-blind, placebo-controlled conducted in the U.S. Canada and Europe had patients randomized into 2 parallel groups using a 2:1 bosentan-to-placebo ratio. 8 It was the first placebo, controlled trial to evaluate the prevention of ulcer formation in 122 patients. The patients were treated for 16 weeks to examine whether bosentan would be an effective therapeutic agent for the prevention of digital ulcers in patients with systemic scleroderma. All patients enrolled had to have a history of documented digital ulcers within the previous 1 year. Patients received bosentan 62.5 mg twice daily or placebo for 4 weeks, then 125 mg bosentan twice daily or placebo for 12 weeks. 79 were randomized into the bosentan arm and 43 into the placebo arm. Baseline characteristics were similar.
A protective effect was seen with the development of new ulcers in patients administered bosentan compared to placebo (48% reduction), most significantly in patients who had ulcers at baseline and with the diffuse form of systemic scleroderma. Unfortunately, no improvement in ulcer healing was observed.
RAPIDS-2 trial evaluated prevention and healing of digital ulcers in 188 patients with a more severe form of the disease. 9 The treatment period was longer. Patients were initiated on bosentan 62.5 mg twice daily for 4 weeks, then increased to 125 mg twice daily for 20 to 32 weeks compared to placebo. The bosentan group had a 30% less incidence in the development of digital ulcers. This finding was more pronounced in patients with greater than 3 ulcers at the time of enrollment.
Launay analyzed the effects of bosentan for active digital ulcers in 9 French patients. 10 All but one patient had the 62.5 mg twice daily bosentan increased to 125 mg twice daily after 4 weeks of therapy and indefinitely continued. The mean follow-up period was 24.3 months. In eight of the nine patients, rapid healing and resolution of ulcers was observed. Another notable observation was the absence of recurrent digital ulcer development.
A 12-week open-label extension to a 16-week study of bosentan use for determining the number of new digital ulcer development was performed by Black. 14 88 patients with systemic sclerosis were enrolled. 53 patients enrolled had active digital ulcers. At the conclusion of the study, only 7 patients (8%) developed new ulcers. Additionally, hand function scores suggested improvement in disability measures.
Even though the digital ulcers may improve with therapy, there still remains the concern for recurrence of ulcer development. García de la Peóa-Lefebvre reported an observational non-controlled, prospective study of the off-label use of bosentan in 15 patients with systemic sclerosis. 12 Ten patients had the limited-cutaneous form, while 5 patients had diffuse cutaneous systemic sclerosis. The aim was to assess the long-term efficacy and tolerability of the treatment modality. Follow-up time after bosentan therapy ranged from 4 months to 36 months. Measurements of healed ulcers, frequency and duration of Raynaud's phenomenon and overall disease assessments were conducted at 6 months (13 patients), 12 months (13 patients), 18 months (12 patients), 24 months (10 patients), 30 months (5 patients) and 36 months (1 patient). At 24 months of therapy there was a statistically significant reduction in digital ulcers and the frequency/duration of Raynaud's phenomenon, but only a trend towards overall disease improvement was evident. No statistical analysis was reported after 24 months. Two patients died during the study due to clinical deterioration from pulmonary disease, 1 patient permanently discontinued bosentan due to jaundice and 2 patients required temporary discontinuation due to transaminitis and were included in the outcome results once therapy was resumed.
Bosentan is currently approved in Europe for prophylaxis of digital ulcerations. It is not used for treatment purposes due to the lack of any observable improvements in the healing of existing ulcers.
Raynaud's phenomenon
The etiology of Raynaud's phenomenon remains elusive. The severe manifestation of the vasospasm from Raynaud's phenomenon leads to the development of digital ulcers and may require hospitalization. 13 Abnormalities in neuronal regulation and the peripheral vasculature diameter secondary to cold exposure are proposed contributing factors. Upon inspection, the small vessels of the digits have intimal thickening. Again, endothelin-1's role in the mechanistic injury as alterations in the vasoregulation of Raynaud's have been proposed. Observational studies, case series and pilot studies using bosentan for Raynaud's phenomenon have been conducted. Ramos-Casals shared their experience of four patients with severe Raynaud's phenomenon and the use of bosentan. All four experienced great improvement in this manifestation. A couple had significant improvement in as little time as 4-5 months. 14 Alegre-Sancheo reported that patients with systemic sclerosis can experience improvement in intensity of Raynaud's episodes after 12 months of oral bosentan therapy. 15 The long-term efficacy was seen in 2 of the 8 patients even after 18 months of follow-up. Also 3 patients while on bosentan were able to completely withdraw themselves from their vasodilator agents.
Raynaud's phenomenon is a disease strongly associated with scleroderma. Circulating endothelial cells serve as a marker of vascular injury in scleroderma. 16 This can indirectly be measured by the circulating levels of CD 146 cells, a population of predominantly endothelial cells. Dunne shared their experience of a patient with systemic sclerosis with severe Raynaud's phenomenon leading to gangrenous lesions. 17 Calcium channel blocker use in this patient was unsuccessful, so bosentan was administered. The patient had a dramatic clinical improvement within 1 month. Laser Doppler fluximetry utilized showed an improvement in small vessel blood flow. The circulating CD146 cells measured over time had drastically tapered down. Concomitant measurements of stem cells containing angioblastic activity (CD 34 cells) within the circulation increased with the signs of clinical improvement. Selenko-Gebauer investigated the benefits of bosentan in 3 patients with Raynaud's phenomenon and either pre-scleroderma or limited sclerosis. 18 These three patients received 16 weeks of bosentan therapy. The primary outcome was to evaluate Raynaud's event activity and severity of pain. This was done using the modified Scleroderma Health Assessment Questionnaire and a visual analogue scale at the time of entry into the study and on monthly visits. A peripheral thermoregulation to assess thermography of both hands was assessed at baseline and at week 16. In all three patients, similar experiences were found at week 16; the number of daily Raynaud's events decreased by half relative to number of baseline events as did the pain severity (Fig. 1). The thermography of both hands showed significant improvement in the skin temperature recovery 20 minutes after cold challenge for only one patient.
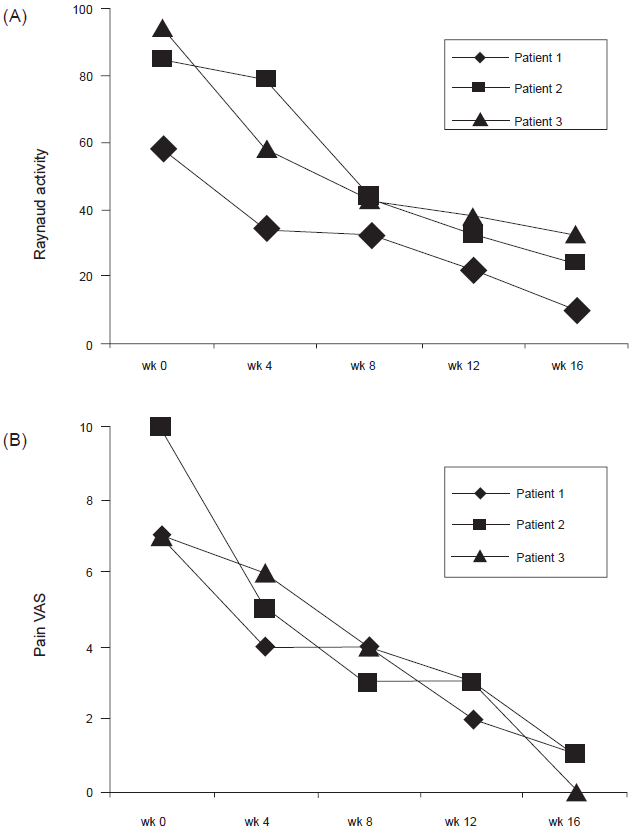
A) Raynaud's activity assessed by a modified SHAQ. Patients were evaluated every 4 weeks during the 16 weeks treatment period. B) Pain severity determined by a visual analogue scale (range 0-10) during bosentan treatment period assessed every 4 weeks. 18
Bosentan was recently evaluated in 15 Japanese patients with connective tissue disease (2 mixed connective tissue disease and 13 systemic sclerosis) with associated pulmonary hypertension, digital ulcers, dermal sclerosis and Raynaud's phenome-non. 19 Patients were treated for the duration of 40 to 96 weeks. Not only did these patients improve in their hemodynamic parameters and exercise capacity, but they also had improvement of Raynaud's after 8 weeks, digital ulcers after 12 weeks and dermal sclerosis after 24 months of therapy.
Cardiac involvement
Cardiac involvement is generally occult, but estimated to occur in 100% of patients when sensitive tools are utilized.20,21 Irreversible interstitial fibrotic lesions and degeneration of myocardial fibers predominantly surrounding blood vessels lead to conduction abnormalities and focal ischemia. Myocardial perfusion and function by cardiac MRI and tissue Doppler ECHO in 18 systemic sclerosis patients without clinical heart failure or pulmonary arterial hypertension were compared to control subjects. After only 4 weeks of bosentan therapy, an increase in myocardial function and perfusion was seen through an evaluation using cardiac MRI and tissue Doppler ECHO. This is in stark contrast to the lack of benefit with bosentan in patients with severe heart failure.22,23
Pulmonary fibrosis
Scleroderma associated ILD, a non-specific interstitial pneumonia (NSIP) is due to fibroblast proliferation and deposition of excessive levels of extracellular matrix proteins in the alveolar membranes. ET-1 is a strong mediator that can enhance the proliferation of pulmonary mesenchymal cells. Endothelin-1's pivotal role in the development of pulmonary fibrosis has been established in rat models. 24 In humans, approximately 80% of all patients with systemic sclerosis will develop some degree of pulmonary fibrosis. With a 42% 10-year mortality rate in patients with more severe fibrosis, therapy for scleroderma has been a major focus in the last few years.
An international, multicenter, prospective, double-blind, randomized, placebo-controlled, parallel group study (BUILD-1 Trial) evaluating the effects of bosentan versus placebo on exercise capacity and time to disease progression in 158 patients with IPF failed to show any beneficial effect with bosentan. 25 The BUILD-2 trial was specifically designed to assess bosentan therapeutic affects in patients with scleroderma-induced interstitial lung disease. 26 It was a double-blinded, randomized, placebo-controlled study on 132 patients. The primary endpoint was to determine changes in six-minute walk test from baseline to 12 months. Secondary endpoints included time to worsening in pulmonary function test or death. Unfortunately, bosentan again failed to show significance between the two groups either in the primary outcome (mean change 6MWD: -9 meters in placebo arm vs. -12 meters in bosentan arm) or the secondary outcomes. Further trials are warranted to see any clinical benefits in pulmonary fibrosis.
Pulmonary hypertension
Renal crisis was previously the major cause of mortality, but with the advent use of angiotensin converting enzyme inhibitors this has now significantly declined. Instead, pulmonary complications have emerged as the major contributing factor for death, particularly pulmonary arterial hypertension.
As patients present with symptoms of dyspnea, followed by fatigue and chest pain, the clinician should be highly suspicious for pulmonary arterial hypertension. 27 These symptoms may be present even two years prior to the actual diagnosis. The reported annual incidence of pulmonary arterial hypertension in systemic scleroderma is between 10% to 50%.28,29 The prevalence of SSc-induced pulmonary arterial hypertension is reported to range between 8%-12% when evaluated by pulmonary artery catheter hemodynamic parameters.30,31 Once pulmonary arterial hypertension has developed, a rapid functional decline is generally experienced if left untreated. 32 In comparison to primary pulmonary arterial hypertension, the risk of death is three-fold with the 1-year patient survival approaching 50% in individuals without medical (Fig. 2).32,33
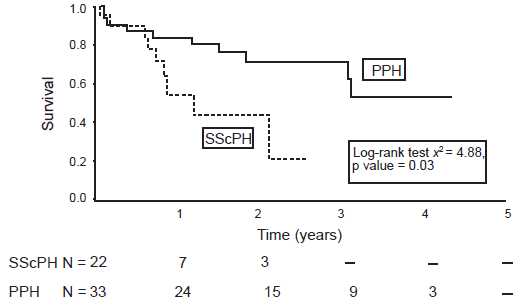
Kaplan-Meier survival estimates of patients with SScPH and PPH. 33
Multiple pathogeneses have been implicated in the development of this ominous disease process. The combined overproduction of endothelin-1 and the reduction of nitric oxide and prostacyclin levels provoke a sustained pulmonary vasoconstriction, adventitial fibrosis and hypertrophy of the pulmonary arteries, vascular remodeling and in situ thrombosis. This eventually leads to the obliteration of the pulmonary vasculature and cause the familiar hemodynamic parameter findings of elevated pulmonary artery pressures (PAP) and pulmonary vascular resistance (PVR) with a normal pulmonary artery occlusion pressure (PAOP).
Normally, the pulmonary vasculature is very compliant. Therapeutic options for PAH are instituted to reduce the PAP and PVR in order to reduce the risk of right heart strain and overt failure. The aforementioned options include the use of oxygen supplementation, diuretic agents, calcium-channel blockers when vasoreactivity is present, inotropic support, prostacyclin analogues, phosphodiesterase inhibitors and endothelin receptor blockers, such as bosentan. In this manuscript, we are primarily concentrating on the role of bosentan in SSc-induced pulmonary arterial hypertension.
As previously mentioned, elevated concentrations of endothelin-1 (ET-1), a potent neurohormone produced by the vascular endothelium, is implicated in contributing elevated PAP and PVR. An inverse correlation has been observed between ET-1 levels and pulmonary blood flow and cardiac output. Two smooth muscle endothelian-1 receptors play an integral role; ETA causes vasoconstriction while ETB leads to fibrosis, hypertrophy, and vascular remodeling. As with all pulmonary arterial hypertension therapies, measures are taken to restore the hemodynamic parameters and prevent the risk of peripheral organ hypoperfusion.
Bosentan, a non-selective endothelian-1 receptor antagonist, has been approved by the Food and Drug Administration for use in patients with World Health Organization (WHO) functional class III and IV pulmonary hypertension. The pharmacological intervention with bosentan produces vasodilation of the pulmonary vasculature and has been shown to improve both hemodynamic parameters and functional outcomes.
Patients with pulmonary arterial hypertension are clinically monitored for progression of disease or clinical improvement with the symptomology assessment tools, oxygen saturation, exercise tolerance (six-minute walk distance) and clinical findings related to right heart failure.
Bosentan is an effective pharmacological agent in reducing pulmonary vascular resistance leading to an increase in cardiac output thereby improving exercise capacity and symptoms. Two pivotal studies have been published evaluating the efficacy of bosentan in pulmonary hypertension in systemic sclerosis. In 2001, Channick published a randomized double-blind placebo controlled trial in Lancet. 34 Thirty-two patients with pulmonary hypertension (primary or scleroderma) WHO Class III-IV were randomly assigned to take bosentan (62.5 mg twice daily for four weeks then 125 mg twice daily) or placebo for twelve weeks. Twenty-one patients in the bosentan group had an average increase in 6 minute walk distance by 70 meters compared to placebo, which decreased by 6 meters (Fig. 3). There was also a significant decrease in pulmonary vascular resistance in the bosentan group leading to an improvement in the cardiac index. Patients in the treatment group additionally reported an improvement in the Borg Dyspnea Scale by 1.6 at twelve weeks compared to those given placebo. 34 The BREATHE-1 study was a larger double blind, placebo control trial published in 2002 in which 213 patients with pulmonary hypertension (primary or connective tissue disease) were randomized to receive placebo or bosentan 62.5 mg twice daily for 4 weeks followed by an increase in bosentan (125 mg or 250 mg twice daily) for a minimum of twelve weeks. 35 Once again, there was an improvement in 6-minute walk distance by 44 meters in the bosentan group, an improvement in the Borg Dyspnea Index and an increase in the time to clinical worsening. STUDY 351 reported similar findings when assessing a subgroup with connective tissue disease. 36 Recently, a published report in the European Journal of Clinical Investigation, evaluated the efficacy of bosentan for severe pulmonary arterial hypertension related to systemic sclerosis with interstitial lung disease. 37 There was an improvement in the 6-minute walk distance from 71.9 m (+/- 54.7) to 202.6 m (+/- 108.1) after 6 months of therapy. Many of the studies with endothelin-1 receptor antagonists have a six-month follow up. In 2003, a one-year follow up study was published which enrolled 29 patients with PAH (primary or scleroderma) who received a twice daily 125 mg bosentan or placebo for a one-year duration. At six months, the patients in the bosentan group maintained their improvement in the 6-minute walk distance and hemodynamics. 38
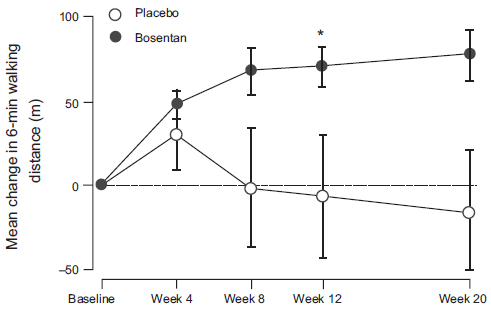
Change in 6 minute walk distance from baseline to week 20. Points = means, bars = SE. Patients who did not complete week-20. Assessments (bosentan n = 1, placebo 4) had their last observed value carried forward. *p < 0.05 versus baseline, p = 0.021 versus placebo. 34
In addition to the 6MWD test, N-terminal probrain natriuetic peptide (NT-proBNP) has been investigated as a noninvasive marker in patients with systemic sclerosis induced pulmonary hypertension. 39 The ten patients (9 WHO class III, 1 WHO class IV) included in the study had 6MWD and NT-proBNP levels drawn at baseline and after 20 weeks of bosentan. In addition to improvements in the 6MWD by 50 meters and an escalation to WHO class II, the median NT-proBNP plasma levels decreased to half the baseline values (474 fmol/mL to 238 fmol/mL). Larger trials are needed to further investigate if the change in NT-proBNP levels correlate with PH disease specific therapy administration.
Additionally, ET-1 has been reported to regulate matrix metalloproteinases (MMPs) in the remodeling various diseases. 40 MMP-2 and MMP-9 have been implicated in the course of systemic sclerosis and altered extracellular matrix deposition. Giannelli investigated differences in serum concentration of MMP-2 and MM-9 in 35 systemic sclerosis patients with and without therapy with bosentan. Twelve of these enrolled patients were diagnosed with WHO class II–III SSc-induced PAH patients. Eight of them received bosentan 62.5 mg twice daily for 4 weeks, then 125 mg twice daily for 50 weeks. 41 At baseline, MMP-9 levels had inversely correlated to the presence of PAH, but not so for the serum MMP-2 levels. Subsequent to the administration of bosentan, MMP-9 serum concentration significantly increased and correlated with clinical outcome measurements. Further investigations are warranted for intermediary mediators causing pulmonary arterial hypertension.
Safety and tolerability
Postmarketing surveillence has shown that bosentan is safe and well tolerated agent.
A pilot study by Bshouty, showed that the use of bosentan in connective tissue disease related pulmonary arterial hypertension and pulmonary fibrosis appeared to be safe. 42 Additional studies followed suit. Within 30 months of bosentan use, safety profile was evaluated in approximately 5,000 patients in a European database of a postmarketing surveillance system. 43 4,623 of the patients were naïve to bosentan. A crude incidence of 7.6% was observed to have transaminitis. This is particularly important when there is co-administration of bosentan and glyburide. Of those individuals with transaminitis, 3.2% were required to fully discontinue the medication. Transaminase levels are generally reversed within 9 weeks of either discontinuation or reduction of bosentan dosage. Spontaneous resolution has also been observed without medication alterations. Additionally, no significant sequalae has been reported due to any degree of transaminitis.
The incidence of other common adverse events reported in clinical trials compared to placebo include; headache (22% vs. 20%), nasopharyngitis (11% vs. 8%), flushing (9% vs. 5%), hypotension (7% vs. 4%), palpitations (5% vs. 1%), edema (4% vs. 3%), fatigue (4% vs. 1%), dyspepsia (4% vs. 0%).
Patient focused perspectives: quality of life
There is very limited information on quality of life issues and methods to quantify the assessments with the use of bosentan. A comparison between the medical outcomes study 36-item short form health survey (SF-36), the Australian Quality of Life (AQoL) and Minnesota Living with Heart Failure (MLwHF) questionnaire showed that both the MLwHF and SF-36 are useful tools to assess quality of life in patients with pulmonary hypertension. 44 Keogh reported that patients diseased with pulmonary artery hypertension have quality of life improvement in 59% after 3 months and 41% after 6 months of bosentan therapy. 45 A retrospective analysis of 69 patients in an open-label mulitcenter trial by the same group reported quality of life and six-minute walk distance data improvement for at least 6 months with the administration of bosentan. 46 Denton investigated changes in health-related quality of life and how it corresponded with survival over a 48-week period. 47 The SF-36 and a disease-specific (scleroderma) modified Health Assessment Questionnaire (HAQ) were used for assessment. This multicenter, prospective singlearm trial with bosentan assessed 53 moderate-to-severely disabled patients at weeks 14 and 48 for WHO classification, quality of life, clinical worsening and survival. At the end of the 48-week period the overall survival was 92%, the WHO class improved in 27% of patients, while the quality of life changes were minimal in all patients.
Conclusions, place in therapy
Systemic sclerosis is a rare disease that causes degenerative changes, vascular abnormalities and progressive fibrosis in the skin, blood vessels, heart, lungs. The most common initial symptoms include Raynaud's phenomenon and progressive cutaneous fibrosis. Visceral organ involvement subsequently progress in an indolent fashion. This disease has an associated 50% mortality rate at 5 years in untreated patients. The underlying pathophysiology of this disease is an alteration of endothelial cells leading to a cascade of profibrotic changes inducing an excessive collagen deposition in various organs. Endothelin-1 has been determined to play a major role.
There are a number of novel technologies and improved screening methods that allow physicians to identify and treat the various manifestations of this debilitating condition. Different treatment strategies for systemic sclerosis exist for patients. This review is not to imply that bosentan should be used as a first line agent or the sole therapy for various manifestations of systemic sclerosis. We have; however, provided literature review for the off-label use of bosentan as an effective therapeutic agent for systemic sclerosis-induced cutaneous fibrosis, digital ulcers, Raynaud's phenomenon, and possibly heart involvement. However, it has not been shown to be effective in pulmonary fibrosis at this time.
Bosentan is best known for its use in patients with WHO class III and IV pulmonary hypertension. It has lead to the improvement in hemodynamics parameters, clinical improvements, the time to clinical worsening and transplantation requirements and most importantly impacted patients’ quality of life.
We have referenced many studies that have proven bosentan a safe and efficacious drug that is well tolerated by patients. The most common adverse drug event experienced is transaminitis, but this is easily reversible. To date, there have been no reports of severe hepatic injury related to bosentan. With close monitoring of liver function test bosentan can be a life altering therapeutic option for patients with systemic sclerosis and various manifestations.
Disclosure
The authors report no conflicts of interest.
